CHEMISTRY THE CENTRAL SCIENCE
20 ELECTRO-CHEMISTRY
20.6 CELL POTENTIALS UNDER NONS TANDARD CO NDITIONS
We have seen how to calculate the emf of a cell when the reactants and products are under standard conditions. As a voltaic cell is discharged, however, reactants are consumed and products are generated, so concentrations change. The emf progressively drops until E = 0, at which point we say the cell is “dead.” In this section we examine how the emf generated under nonstandard conditions can be calculated by using an equation first derived by Walther Nernst (1864–1941), a German chemist who established many of the theoretical foundations of electrochemistry.
The Nernst Equation
The effect of concentration on cell emf can be obtained from the effect of concentration on free-energy change. ![]() (Section 19.7) Recall that the free-energy change for any chemical reaction, ΔG, is related to the standard free-energy change for the reaction, ΔG°:
(Section 19.7) Recall that the free-energy change for any chemical reaction, ΔG, is related to the standard free-energy change for the reaction, ΔG°:
![]()
The quantity Q is the reaction quotient, which has the form of the equilibrium-constant expression except that the concentrations are those that exist in the reaction mixture at a given moment. ![]() (Section 15.6)
(Section 15.6)
Substituting ΔG = –nFE (Equation 20.11) into Equation 20.15 gives
![]()
Solving this equation for E gives the Nernst equation:

This equation is customarily expressed in terms of the base-10 logarithm:

At T = 298 K the quantity 2.303 RT/F equals 0.0592, with units of volts, and so the Nernst equation simplifies to
![]()
We can use this equation to find the emf E produced by a cell under nonstandard conditions or to determine the concentration of a reactant or product by measuring E for the cell. For example, consider the following reaction:
![]()
In this case n = 2 (two electrons are transferred from Zn to Cu2+), and the standard emf is + 1.10 V (Section 20.4). Thus, at 298 K the Nernst equation gives
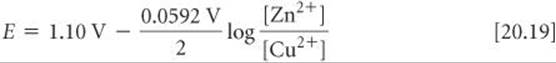
Recall that pure solids are excluded from the expression for Q. ![]() (Section 15.6) According to Equation 20.19, the emf increases as [Cu2+] increases and as [Zn2+] decreases. For example, when [Cu2+] is 5.0 M and [Zn2+] is 0.050 M, we have
(Section 15.6) According to Equation 20.19, the emf increases as [Cu2+] increases and as [Zn2+] decreases. For example, when [Cu2+] is 5.0 M and [Zn2+] is 0.050 M, we have
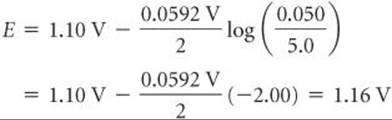
Thus, increasing the concentration of reactant Cu2+ and decreasing the concentration of product Zn2+ relative to standard conditions increases the emf of the cell relative to standard conditions (E° = +1.10 V).
The Nernst equation helps us understand why the emf of a voltaic cell drops as the cell discharges. As reactants are converted to products, the value of Q increases, so the value of E decreases, eventually reaching E = 0. Because ΔG = –nFE (Equation 20.11), it follows that ΔG = 0 whenE = 0. Recall that a system is at equilibrium when ΔG = 0. ![]() (Section 19.7) Thus, when E = 0, the cell reaction has reached equilibrium, and no net reaction occurs.
(Section 19.7) Thus, when E = 0, the cell reaction has reached equilibrium, and no net reaction occurs.
In general, increasing the concentration of reactants or decreasing the concentration of products increases the driving force for the reaction, resulting in a higher emf. Conversely, decreasing the concentration of reactants or increasing the concentration of products causes the emf to decrease from its value under standard conditions.
SAMPLE EXERCISE 20.11 Cell Potential under Nonstandard Conditions
Calculate the emf at 298 K generated by a voltaic cell in which the reaction is
![]()
![]()
SOLUTION
Analyze We are given a chemical equation for a voltaic cell and the concentrations of reac-tants and products under which it operates. We are asked to calculate the emf of the cell under these nonstandard conditions.
Plan To calculate the emf of a cell under nonstandard conditions, we use the Nernst equation in the form of Equation 20.18.
Solve We calculate E° for the cell from standard reduction potentials (Table 20.1 or Appendix E). The standard emf for this reaction was calculated in Sample Exercise 20.6: E° = 0.79 V. As that exercise shows, six electrons are transferred from reducing agent to oxidizing agent, so n = 6. The reaction quotient, Q, is
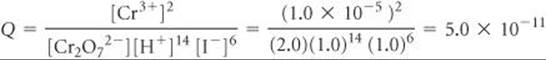
Using Equation 20.18, we have
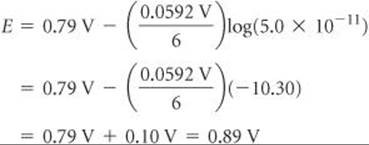
Check This result is qualitatively what we expect: Because the concentration of Cr2O72– (a reactant) is greater than 1 M and the concentration of Cr3+ (a product) is less than 1 M, the emf is greater than E°. Because Q is about 10–10, log Q is about –10. Thus, the correction to E° is about 0.06 × 10/6, which is 0.1, in agreement with the more detailed calculation.
PRACTICE EXERCISE
Calculate the emf generated by the cell described in the practice exercise accompanying Sample Exercise 20.6 when [Al3+] = 4.0 × 10–3 and [I–] = 0.010 M.
Answer: E = +2.36 V
SAMPLE EXERCISE 20.12 Calculating Concentrations in a Voltaic Cell
If the potential of a Zn-H+ cell (like that in Figure 20.9) is 0.45 V at 25 °C when [Zn2+] = 1.0 M and PH2 = 1.0 atm, what is the H+ concentration?
SOLUTION
Analyze We are given a description of a voltaic cell, its emf, and the concentration of Zn2+ and the partial pressure of H2 (both products in the cell reaction). We are asked to calculate the concentration of H+, a reactant.
Plan We write the equation for the cell reaction and use standard reduction potentials to calculate E° for the reaction. After determining the value of n from our reaction equation, we solve the Nernst equation, Equation 20.18, for Q. Finally, we use the equation for the cell reaction to write an expression for Q that contains [H+] to determine [H+].
Solve The cell reaction is
![]()
The standard emf is

Because each Zn atom loses two electrons,
![]()
Using Equation 20.18, we can solve for Q:
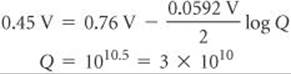
Q has the form of the equilibrium constant for the reaction:
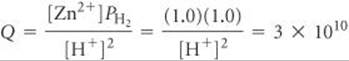
Solving for [H+], we have
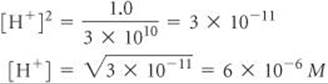
Comment A voltaic cell whose cell reaction involves H+ can be used to measure [H+] or pH. A pH meter is a specially designed voltaic cell with a voltmeter calibrated to read pH directly. ![]() (Section 16.4)
(Section 16.4)
PRACTICE EXERCISE
What is the pH of the solution in the cathode half-cell in Figure 20.9 when PH2 = 1.0 atm, [Zn2+] in the anode half-cell is 0.10 M, and the cell emf is 0.542 V?
Answer: pH = 4.23 (using data from Appendix E to obtain E° to three significant figures)
Concentration Cells
In the voltaic cells we have looked at thus far, the reactive species at the anode has been different from the reactive species at the cathode. Cell emf depends on concentration, however, so a voltaic cell can be constructed using the same species in both half-cells as long as the concentrations are different. A cell based solely on the emf generated because of a difference in a concentration is called a concentration cell.
An example of a concentration cell is diagrammed in ![]() FIGURE 20.15(a). One half-cell consists of a strip of nickel metal immersed in a 1.00 × 10–3M solution of Ni2+(aq). The other half-cell also has an Ni(s) electrode, but it is immersed in a 1.00 M solution of Ni2+(aq). The two half-cells are connected by a salt bridge and by an external wire running through a voltmeter. The half-cell reactions are the reverse of each other:
FIGURE 20.15(a). One half-cell consists of a strip of nickel metal immersed in a 1.00 × 10–3M solution of Ni2+(aq). The other half-cell also has an Ni(s) electrode, but it is immersed in a 1.00 M solution of Ni2+(aq). The two half-cells are connected by a salt bridge and by an external wire running through a voltmeter. The half-cell reactions are the reverse of each other:

Although the standard emf for this cell is zero,
![]()
the cell operates under nonstandard conditions because the concentration of Ni2+(aq) is not 1 M in both half-cells. In fact, the cell operates until [Ni2+]anode = [Ni2+]cathode. Oxidation of Ni(s) occurs in the half-cell containing the more dilute solution, which means this is the anode of the cell. Reduction of Ni2+(aq) occurs in the half-cell containing the more concentrated solution, making it the cathode. The overall cell reaction is therefore

We can calculate the emf of a concentration cell by using the Nernst equation. For this particular cell, we see that n = 2. The expression for the reaction quotient for the overall reaction is Q = [Ni2+]dilute/[Ni2+]concentrated. Thus, the emf at 298 K is

![]() GO FIGURE
GO FIGURE
Assuming that the solutions are made from Ni(NO3)2, how do the ions migrate as the cell operates?
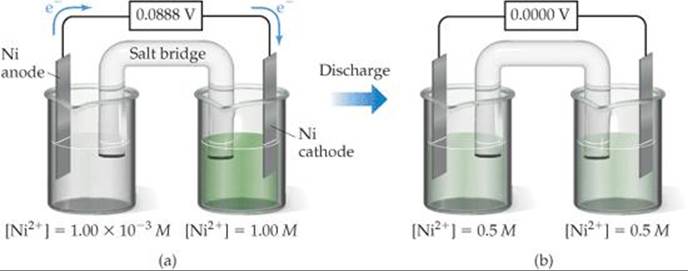
![]() FIGURE 20.15 Concentration cell based on the Ni2+-Ni cell reaction. (a) Concentrations of Ni2+(aq) in the two half-cells are unequal, and the cell generates an electrical current. (b) The cell operates until [Ni2+(ag)] is the same in the two half-cells, at which point the cell has reached equilibrium and is “dead.”
FIGURE 20.15 Concentration cell based on the Ni2+-Ni cell reaction. (a) Concentrations of Ni2+(aq) in the two half-cells are unequal, and the cell generates an electrical current. (b) The cell operates until [Ni2+(ag)] is the same in the two half-cells, at which point the cell has reached equilibrium and is “dead.”
This concentration cell generates an emf of nearly 0.09 V even though E° = 0. The difference in concentration provides the driving force for the cell. When the concentrations in the two half-cells become the same, Q = 1 and E = 0.
The idea of generating a potential by a concentration difference is the basis for the operation of pH meters. It is also a critical aspect in biology. For example, nerve cells in the brain generate a potential across the cell membrane by having different concentrations of ions on the two sides of the membrane. The regulation of the heartbeat in mammals, as discussed in the following “Chemistry and Life” box, is another example of the importance of electrochemistry to living organisms.
 CHEMISTRY AND LIFE
CHEMISTRY AND LIFE
HEARTBEATS AND ELECTROCARDIOGRAPHY
The human heart is a marvel of efficiency and dependability. In a typical day an adult's heart pumps more than 7000 L of blood through the circulatory system, usually with no maintenance required beyond a sensible diet and lifestyle. We generally think of the heart as a mechanical device, a muscle that circulates blood via regularly spaced muscular contractions. However, more than two centuries ago, two pioneers in electricity, Luigi Galvani (1729–1787) and Alessan-dro Volta (1745–1827), discovered that the contractions of the heart are controlled by electrical phenomena, as are nerve impulses throughout the body. The pulses of electricity that cause the heart to beat result from a remarkable combination of electrochemistry and the properties of semipermeable membranes. ![]() (Section 13.5)
(Section 13.5)
Cell walls are membranes with variable permeability with re-spect to a number of physiologically important ions (especially Na+, K+, and Ca2+). The concentrations of these ions are different for the fluids inside the cells (the intracellular fluid, or ICF) and outside the cells (theextracellular fluid, or ECF). In cardiac muscle cells, for example, the concentrations of K+ in the ICF and ECF are typically about 135 millimolar (mM) and 4 mM, respectively. For Na+, however, the concentration difference between the ICF and ECF is opposite that for K+; typically, [Na+]ICF = 10 mM and [Na+]ECF = 145 mM.
The cell membrane is initially permeable to K+ ions but is much less so to Na+ and Ca+. The difference in concentration of K+ ions between the ICF and ECF generates a concentration cell: Even though the same ions are present on both sides of the membrane, there is a potential difference between the two fluids that we can calculate using the Nernst equation with E° = 0. At physiological temperature (37°C) the potential in millivolts for moving K+ from the ECF to the ICF is
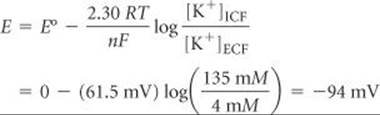
In essence, the interior of the cell and the ECF together serve as a voltaic cell. The negative sign for the potential indicates that work is required to move K+ into the intracellular fluid.
Changes in the relative concentrations of the ions in the ECF and ICF lead to changes in the emf of the voltaic cell. The cells of the heart that govern the rate of heart contraction are called the pacemaker cells. The membranes of the cells regulate the concentrations of ions in the ICF, allowing them to change in a systematic way. The concentration changes cause the emf to change in a cyclic fashion, as shown in ![]() FIGURE 20.16. The emf cycle determines the rate at which the heart beats. If the pacemaker cells malfunction because of disease or injury, an artificial pacemaker can be surgically implanted. The artificial pacemaker contains a small battery that generates the electrical pulses needed to trigger the contractions of the heart.
FIGURE 20.16. The emf cycle determines the rate at which the heart beats. If the pacemaker cells malfunction because of disease or injury, an artificial pacemaker can be surgically implanted. The artificial pacemaker contains a small battery that generates the electrical pulses needed to trigger the contractions of the heart.
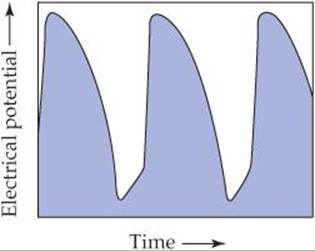
![]() FIGURE 20.16 Changes in electrical potential in the human heart. Variation of the electrical potential caused by changes of ion concentrations in the pacemaker cells of the heart.
FIGURE 20.16 Changes in electrical potential in the human heart. Variation of the electrical potential caused by changes of ion concentrations in the pacemaker cells of the heart.
During the late 1800s, scientists discovered that the electrical impulses that cause the contraction of the heart muscle are strong enough to be detected at the surface of the body. This observation formed the basis for electrocardiography, noninvasive monitoring of the heart by using a complex array of electrodes on the skin to measure voltage changes during heartbeats. A typical electrocardiogram is shown in ![]() FIGURE 20.17. It is quite striking that, although the heart's major function is the mechanical pumping of blood, it is most easily monitored by using theelectrical impulses generated by tiny voltaic cells.
FIGURE 20.17. It is quite striking that, although the heart's major function is the mechanical pumping of blood, it is most easily monitored by using theelectrical impulses generated by tiny voltaic cells.
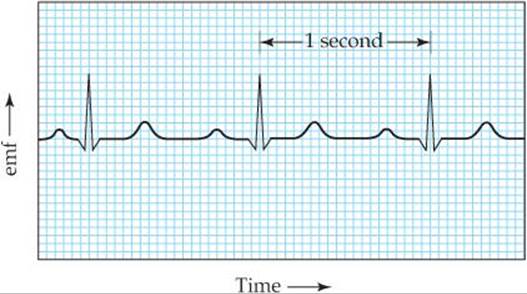
![]() FIGURE 20.17 A typical electrocardiogram. The printout records the electrical events monitored by electrodes attached to the body surface.
FIGURE 20.17 A typical electrocardiogram. The printout records the electrical events monitored by electrodes attached to the body surface.
SAMPLE EXERCISE 20.13 Determining pH Using a Concentration Cell
A voltaic cell is constructed with two hydrogen electrodes. Electrode 1 has PH2 = 1.00 atm and an unknown concentration of H+(aq). Electrode 2 is a standard hydrogen electrode (PH2 = 1.00 atm, [H+] = 1.00 M). At 298 K the measured cell potential is 0.211 V, and the electrical current is observed to flow from electrode 1 through the external circuit to electrode 2. Calculate [H+] for the solution at electrode 1. What is the pH of the solution?
SOLUTION
Analyze We are given the potential of a concentration cell and the direction in which the current flows. We also have the concentrations or partial pressures of all reactants and products except for [H+] in half-cell 1, which is our unknown.
Plan We can use the Nernst equation to determine Q and then use Q to calculate the unknown concentration. Because this is a concentration cell, ![]() = 0 V.
= 0 V.
Solve Using the Nernst equation, we have
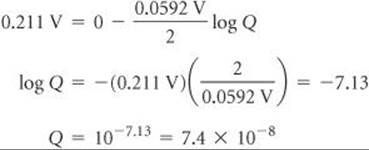
Because electrons flow from electrode 1 to electrode 2, electrode 1 is the anode of the cell and electrode 2 is the cathode. The electrode reactions are therefore as follows, with the concentration of H+(aq) in electrode 1 represented with the unknown x:

Thus,
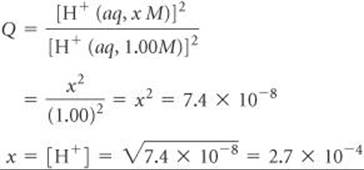
At electrode 1, therefore, the pH of the solution is
![]()
Comment The concentration H+ of at electrode 1 is lower than that in electrode 2, which is why electrode 1 is the anode of the cell: The oxidation of H2 to H+(aq) increases [H+] at electrode 1.
PRACTICE EXERCISE
A concentration cell is constructed with two Zn(s)–Zn2+(aq) half-cells. In one half-cell [Zn2+] = 1.35 M, and in the other [Zn2+] = 3.75 × 10–4M. (a) Which half-cell is the anode? (b) What is the emf of the cell?
Answers: (a) the half-cell in which [Zn2+] = 3.75 × 10–4M, (b) 0.105 V