CHEMISTRY THE CENTRAL SCIENCE
21 NUCLEAR CHEMISTRY

THE REMNANT OF THE SUPERNOVA CASSIOPEIA A as viewed from the Chandra X-ray Observatory.
WHAT'S AHEAD
21.1 RADIOACTIVITY
In this chapter we learn how to describe nuclear reactions by equations analogous to chemical equations, in which the nuclear charges and masses of reactants and products are in balance. Radioactive nuclei most commonly decay by emission of alpha, beta, or gamma radiation.
21.2 PATTERNS OF NUCLEAR STABILITY
We recognize that nuclear stability is determined largely by the neutron-to-proton ratio. For stable nuclei, this ratio increases with increasing atomic number. All nuclei with 84 or more protons are radioactive. Heavy nuclei gain stability by a series of nuclear disintegrations leading to stable nuclei.
21.3 NUCLEAR TRANSMUTATIONS
We study nuclear transmutations, which are nuclear reactions induced by bombardment of a nucleus by a neutron or an accelerated charged particle.
21.4 RATES OF RADIOACTIVE DECAY
We learn that radioisotope decays are first-order kinetic processes with characteristic half-lives. Decay rates can be used to determine the age of ancient artifacts and geological formations.
21.5 DETECTION OF RADIOACTIVITY
We see that the radiation emitted by a radioactive substance can be detected by dosimeters, Geiger counters, and scintillation counters.
21.6 ENERGY CHANGES IN NUCLEAR REACTIONS
We recognize that energy changes in nuclear reactions are related to mass changes via Einstein's equation, E = mc2. The nuclear binding energy of a nucleus is the difference between the mass of the nucleus and the sum of the masses of its nucleons.
21.7 NUCLEAR POWER: FISSION
We learn that in nuclear fission a heavy nucleus splits to form two or more product nuclei. This type of nuclear reaction is the energy source for nuclear power plants, and we look at the operating principles of these plants.
21.8 NUCLEAR POWER: FUSION
We learn that in nuclear fusion two light nuclei are fused together to form a more stable, heavier nucleus.
21.9 RADIATION IN THE ENVIRONMENT AND LIVING SYSTEMS
We discover that naturally occurring radioisotopes bathe our planet—and us—with low levels of radiation. The radiation emitted in nuclear reactions can damage living cells but also has diagnostic and therapeutic applications.
ULTIMATELY THE CHEMICAL ENERGY that fuels life on Earth comes from sunlight. Plants convert the radiant energy of the Sun into chemical energy through photosynthesis, producing oxygen and carbohydrates. Life on Earth could not exist without energy from the Sun, but where does the Sun get its energy? Stars, including our sun, use nuclear reactions that involve changes in atomic nuclei to generate their energy. For example, the Sun produces energy by fusing hydrogen atoms to form helium, releasing vast amounts of energy in the process.
The fusion of hydrogen to form helium is the dominant nuclear reaction for most of a star's lifetime. Toward the end of its life, the hydrogen in the star's core is exhausted and the helium atoms fuse to form progressively heavier elements. A select few stars end their lives in dramatic supernova explosions such as the one shown in the chapter opening photograph. The nuclear reactions that occur when a star goes supernova are responsible for the existence of all naturally occurring elements heavier than nickel.
Nuclear chemistry is the study of nuclear reactions, with an emphasis on their uses in chemistry and their effects on biological systems. Nuclear chemistry affects our lives in many ways, particularly in energy and medical applications. In radiation therapy, for example, gamma rays from a radioactive substance such as cobalt-60 are directed to cancerous tumors to destroy them. Positron emission tomography (PET) is one example of a medical diagnostic tool that relies on decay of a radioactive element injected into the body. Radioactivity is also used to help determine the mechanisms of chemical reactions, to trace the movement of atoms in biological systems and the environment, and to date historical artifacts.
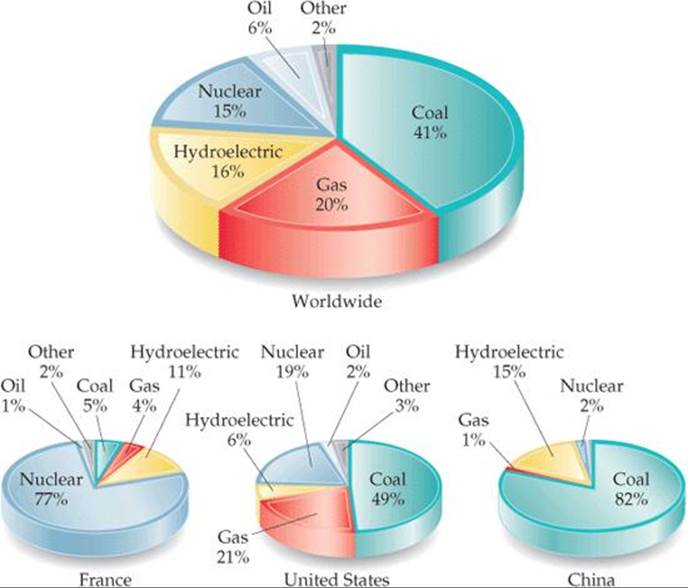
![]() FIGURE 21.1 Sources of electricity generation, worldwide and for select countries.
FIGURE 21.1 Sources of electricity generation, worldwide and for select countries.
Nuclear reactions are also used to generate electricity. Roughly 15% of the electricity generated worldwide comes from nuclear power plants, though the percentage varies from one country to the next, as ![]() FIGURE 21.1 shows.
FIGURE 21.1 shows.
The use of nuclear energy for power generation and the disposal of nuclear wastes from power plants are controversial social and political issues. It is imperative, therefore, that as a citizen with a stake in these matters, you have some understanding of nuclear reactions and the uses of radioactive substances.
21.1 RADIOACTIVITY
To understand nuclear reactions, we must review and develop some ideas introduced in Section 2.3. First, recall that two types of subatomic particles reside in the nucleus: protons and neutrons. We will refer to these particles as nucleons. Recall also that all atoms of a given element have the same number of protons; this number is the element's atomic number. The atoms of a given element can have different numbers of neutrons, however, so they can have different mass numbers; the mass number is the total number of nucleons in the nucleus. Atoms with the same atomic number but different mass numbers are known as isotopes.
The different isotopes of an element are distinguished by their mass numbers. For example, the three naturally occurring isotopes of uranium are uranium-234, uranium-235, and uranium-238, where the numerical suffixes represent the mass numbers. These isotopes are also written ![]() ,
, ![]() , and
, and ![]() , where the superscript is the mass number and the subscript is the atomic number.
, where the superscript is the mass number and the subscript is the atomic number.
Different isotopes of an element have different natural abundances. For example, 99.3% of naturally occurring uranium is uranium-238, 0.7% is uranium-235, and only a trace is uranium-234. Different isotopes of an element also have different stabilities. Indeed, the nuclear properties of any given isotope depend on the number of protons and neutrons in its nucleus.
A nuclide is a nucleus containing a specified number of protons and neutrons. Nuclides that are radioactive are called radionuclides, and atoms containing these nuclei are called radioisotopes.
Nuclear Equations
Most nuclei in nature are stable and remain intact indefinitely. Radionuclides, however, are unstable and spontaneously emit particles and electromagnetic radiation. Emission of radiation is one of the ways in which an unstable nucleus is transformed into a more stable one that has less energy. The emitted radiation is the carrier of the excess energy. Uranium-238, for example, is radioactive, undergoing a nuclear reaction emitting helium-4 nuclei. The helium-4 particles are known as alpha (α) particles, and a stream of them is called alpha radiation. When a ![]() nucleus loses an alpha particle, the remaining fragment has an atomic number of 90 and a mass number of 234. The element with atomic number 90 is Th, thorium. Therefore, the products of uranium-238 decomposition are an alpha particle and a thorium-234 nucleus. We represent this reaction by the nuclear equation
nucleus loses an alpha particle, the remaining fragment has an atomic number of 90 and a mass number of 234. The element with atomic number 90 is Th, thorium. Therefore, the products of uranium-238 decomposition are an alpha particle and a thorium-234 nucleus. We represent this reaction by the nuclear equation
![]()
When a nucleus spontaneously decomposes in this way, it is said either to have decayed or to have undergone radioactive decay. Because an alpha particle is involved in this reaction, scientists also describe the process as alpha decay.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What change in the mass number of a nucleus occurs when the nucleus emits an alpha particle?
In Equation 21.1 the sum of the mass numbers is the same on both sides of the equation (238 = 234 + 4). Likewise, the sum of the atomic numbers on both sides of the equation is equal (92 = 90 + 2). Mass numbers and atomic numbers must be balanced in all nuclear equations.
The radioactive properties of the nucleus in an atom are independent of the chemical state of the atom. In writing nuclear equations, therefore, we are not concerned with the chemical form (element or compound) of the atom in which the nucleus resides.
SAMPLE EXERCISE 21.1 Predicting the Product of a Nuclear Reaction
What product is formed when radium-226 undergoes alpha decay?
SOLUTION
Analyze We are asked to determine the nucleus that results when radium-226 loses an alpha particle.
Plan We can best do this by writing a balanced nuclear reaction for the process.
Solve The periodic table shows that radium has an atomic number of 88. The complete chemical symbol for radium-226 is therefore ![]() . An alpha particle is a helium-4 nucleus, and so its symbol is
. An alpha particle is a helium-4 nucleus, and so its symbol is ![]() (sometimes written as
(sometimes written as ![]() ). The alpha particle is a product of the nuclear reaction, and so the equation is of the form
). The alpha particle is a product of the nuclear reaction, and so the equation is of the form
![]()
where A is the mass number of the product nucleus and Z is its atomic number. Mass numbers and atomic numbers must balance, so
![]()
and
![]()
Hence,
![]()
Again, from the periodic table, the element with Z = 86 is radon (Rn). The product, therefore, is ![]() , and the nuclear equation is
, and the nuclear equation is
![]()
PRACTICE EXERCISE
Which element undergoes alpha decay to form lead-208?
Answer: ![]()
TABLE 21.1 • Properties of Alpha, Beta, and Gamma Radiation

Types of Radioactive Decay
The three most common kinds of radiation given off when a radionuclide decays are alpha (α), beta (β), and gamma (γ) radiation. ![]() (Section 2.2)
(Section 2.2) ![]() TABLE 21.1 summarizes some of the important properties of these types of radiation. As just described, alpha radiation consists of a stream of helium-4 nuclei known as alpha particles, which we denote as
TABLE 21.1 summarizes some of the important properties of these types of radiation. As just described, alpha radiation consists of a stream of helium-4 nuclei known as alpha particles, which we denote as ![]() or
or ![]() .
.
Beta radiation consists of streams of beta (β) particles, which are high-speed electrons emitted by an unstable nucleus. Beta particles are represented in nuclear equations by ![]() or sometimes by
or sometimes by ![]() . The superscript 0 indicates that the mass of the electron is exceedingly small relative to the mass of a nucleon. The subscript – 1 represents the negative charge of the beta particle, which is opposite that of the proton.
. The superscript 0 indicates that the mass of the electron is exceedingly small relative to the mass of a nucleon. The subscript – 1 represents the negative charge of the beta particle, which is opposite that of the proton.
Iodine-131 is an isotope that undergoes decay by beta emission:
![]()
You can see from this equation that beta decay causes the atomic number of the reactant to increase from 53 to 54, which means a proton was created. Therefore, beta emission is equivalent to the conversion of a neutron ![]() to a proton
to a proton ![]() :
:
![]()
Just because an electron is emitted from a nucleus in beta decay, we should not think that the nucleus is composed of these particles any more than we consider a match to be composed of sparks simply because it gives them off when struck. The beta-particle electron comes into being only when the nucleus undergoes a nuclear reaction. Furthermore, the speed of the beta particle is sufficiently high that it does not end up in an orbital of the decaying atom.
Gamma (γ) radiation (or gamma rays) consists of high-energy photons (that is, electromagnetic radiation of very short wavelength). It changes neither the atomic number nor the mass number of a nucleus and is represented as either ![]() or merely γ. Gamma radiation usually accompanies other radioactive emission because it represents the energy lost when the nucleons in a nuclear reaction reorganize into more stable arrangements. Generally, gamma rays are not shown when writing nuclear equations.
or merely γ. Gamma radiation usually accompanies other radioactive emission because it represents the energy lost when the nucleons in a nuclear reaction reorganize into more stable arrangements. Generally, gamma rays are not shown when writing nuclear equations.
Two other types of radioactive decay are positron emission and electron capture. A positron, ![]() , is a particle that has the same mass as an electron (thus, we use the letter e and superscript 0 for the mass) but the opposite charge (represented by the +1 subscript).*
, is a particle that has the same mass as an electron (thus, we use the letter e and superscript 0 for the mass) but the opposite charge (represented by the +1 subscript).*
The isotope carbon-11 decays by positron emission:
![]()
Positron emission causes the atomic number of the reactant in this equation to decrease from 6 to 5. In general, positron emission has the effect of converting a proton to a neutron, thereby decreasing the atomic number of the nucleus by 1:
![]()
Electron capture is the capture by the nucleus of an electron from the electron cloud surrounding the nucleus, as in this rubidium-81 decay:
![]()
Because the electron is consumed rather than formed in the process, it is shown on the reactant side of the equation. Electron capture, like positron emission, has the effect of converting a proton to a neutron:
![]()
![]() TABLE 21.2 summarizes the symbols used to represent the particles commonly encountered in nuclear reactions. The various types of radioactive decay are summarized in
TABLE 21.2 summarizes the symbols used to represent the particles commonly encountered in nuclear reactions. The various types of radioactive decay are summarized in ![]() TABLE 21.3.
TABLE 21.3.
TABLE 21.2 • Particles Found in Nuclear Reactions

TABLE 21.3 • Types of Radioactive Decay

![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Which particles in Table 21.2 result in no change in nuclear charge when emitted in nuclear decay?
SAMPLE EXERCISE 21.2 Writing Nuclear Equations
Write nuclear equations for (a) mercury-201 undergoing electron capture; (b) thorium-231 decaying to protactinium-231.
SOLUTION
Analyze We must write balanced nuclear equations in which the masses and charges of reactants and products are equal.
Plan We can begin by writing the complete chemical symbols for the nuclei and decay particles that are given in the problem.
Solve
(a) The information given in the question can be summarized as
![]()
The mass numbers must have the same sum on both sides of the equation:
![]()
Thus, the product nucleus must have a mass number of 201. Similarly, balancing the atomic numbers gives
![]()
Thus, the atomic number of the product nucleus must be 79, which identifies it as gold (Au):
![]()
(b) In this case we must determine what type of particle is emitted in the course of the radioactive decay:
![]()
From 231 = 231 + A and 90 = 91 + Z, we deduce A = 0 and Z = –1. According to Table 21.2, the particle with these characteristics is the beta particle (electron). We therefore write
![]()
PRACTICE EXERCISE
Write a balanced nuclear equation for the reaction in which oxygen-15 undergoes positron emission.
Answer: ![]()