CHEMISTRY THE CENTRAL SCIENCE
21 NUCLEAR CHEMISTRY
21.3 NUCLEAR TRANSMUTATIONS
Thus far we have examined nuclear reactions in which a nucleus decays spontaneously. A nucleus can also change identity if it is struck by a neutron or by another nucleus. Nuclear reactions induced in this way are known as nuclear transmutations.
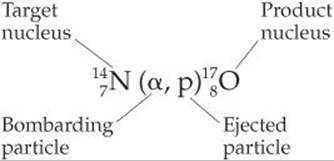
In 1919, Ernest Rutherford performed the first conversion of one nucleus into another, using alpha particles emitted by radium to convert nitrogen-14 into oxygen-17:
![]()
Such reactions have allowed scientists to synthesize hundreds of radioisotopes in the laboratory.
Nuclear transmutations are sometimes represented by listing, in order, the target nucleus, the bombarding particle, the ejected particle, and the product nucleus. Using this condensed notation, Equation 21.8 becomes
SAMPLE EXERCISE 21.5 Writing a Balanced Nuclear Equation
Write the balanced nuclear equation for the process summarized as ![]() .
.
SOLUTION
Analyze We must go from the condensed descriptive form of the reaction to the balanced nuclear equation.
Plan We arrive at the balanced equation by writing n and α, each with its associated subscripts and superscripts.
Solve The n is the abbreviation for a neutron ![]() and α represents an alpha particle
and α represents an alpha particle ![]() . The neutron is the bombarding particle, and the alpha particle is a product. Therefore, the nuclear equation is
. The neutron is the bombarding particle, and the alpha particle is a product. Therefore, the nuclear equation is
![]()
PRACTICE EXERCISE
Write the condensed version of the nuclear reaction
![]()
Answer:![]()
Accelerating Charged Particles
Alpha particles and other positively charged particles must move very fast to overcome the electrostatic repulsion between them and the target nucleus. The higher the nuclear charge on either the bombarding particle or the target nucleus, the faster the bombarding particle must move to bring about a nuclear reaction. Many methods have been devised to accelerate charged particles, using strong magnetic and electrostatic fields. These particle accelerators, popularly called “atom smashers,” bear such names as cyclotron and synchrotron.
A common theme in all particle accelerators is the need to create charged particles so that they can be manipulated by electrical and magnetic fields. The tubes through which the particles move must be kept at high vacuum so that the particles do not inadvertently collide with any gas-phase molecules.
![]() FIGURE 21.5 shows the Relativistic Heavy Ion Collider (RHIC) located at Brookhaven National Laboratory. This facility and the Large Hadron Collider (LHC) at CERN (Conseil Européen pour la Recherche Nucléaire) near Geneva are two of the largest particle accelerators in the world. Both LHC and RHIC are capable of accelerating protons, as well as heavy ions such as gold and lead, to speeds approaching the speed of light. Scientists study the outcomes of collisions involving these ultra-high-energy particles. These experiments are used to investigate the fundamental structure of matter and ultimately answer questions about the beginning of the universe.
FIGURE 21.5 shows the Relativistic Heavy Ion Collider (RHIC) located at Brookhaven National Laboratory. This facility and the Large Hadron Collider (LHC) at CERN (Conseil Européen pour la Recherche Nucléaire) near Geneva are two of the largest particle accelerators in the world. Both LHC and RHIC are capable of accelerating protons, as well as heavy ions such as gold and lead, to speeds approaching the speed of light. Scientists study the outcomes of collisions involving these ultra-high-energy particles. These experiments are used to investigate the fundamental structure of matter and ultimately answer questions about the beginning of the universe.

![]() FIGURE 21.5 The Relativistic Heavy Ion Collider. This particle accelerator is located at Brookhaven National Laboratory on Long Island, New York.
FIGURE 21.5 The Relativistic Heavy Ion Collider. This particle accelerator is located at Brookhaven National Laboratory on Long Island, New York.
Reactions Involving Neutrons
Most synthetic isotopes used in medicine and scientific research are made using neutrons as the bombarding particles. Because neutrons are neutral, they are not repelled by the nucleus. Consequently, they do not need to be accelerated to cause nuclear reactions. The neutrons are produced in nuclear reactors. For example, cobalt-60, which is used in cancer radiation therapy, is produced by neutron capture. Iron-58 is placed in a nuclear reactor and bombarded by neutrons to trigger the reactions sequence
![]()
![]()
![]()
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Can an electrostatic or magnetic field be used to accelerate neutrons in a particle accelerator? Why or why not?
Transuranium Elements
Nuclear transmutations have been used to produce the elements with atomic number above 92, collectively known as the transuranium elements because they follow uranium in the periodic table. Elements 93 (neptunium, Np) and 94 (plutonium, Pu) were produced in 1940 by bombarding uranium-238 with neutrons:
![]()
![]()
Elements with still larger atomic numbers are normally formed in small quantities in particle accelerators. Curium-242, for example, is formed when a plutonium-239 target is bombarded with accelerated alpha particles:
![]()
In 1996 a team of European scientists based in Germany synthesized element 112, copernicium, Cn, by bombarding a lead target continuously for three weeks with a beam of zinc atoms:
![]()
Amazingly, their discovery was based on the detection of only one atom of the new element, which decays after roughly 100 μs by alpha decay to form darmstadtium-273 (element 110). Within one minute, another five alpha decays take place producing fermium-253 (element 100). The finding has been verified in both Japan and Russia. More recently, scientists have reported the synthesis of elements 113 through 118. These results have yet to be confirmed by the International Union for Pure and Applied Chemistry, although the results look promising. Names and symbols have not yet been chosen for these new elements.