CHEMISTRY THE CENTRAL SCIENCE
21 NUCLEAR CHEMISTRY
21.5 DETECTION OF RADIOACTIVITY
A variety of methods have been devised to detect emissions from radioactive substances. Henri Becquerel discovered radioactivity because radiation caused fogging of photographic plates, and since that time photographic plates and film have been used to detect radioactivity. The radiation affects photographic film in much the same way as X-rays do. The greater the extent of exposure to radiation, the darker the area of the developed negative. People who work with radioactive substances carry film badges to record the extent of their exposure to radiation (![]() FIGURE 21.8).
FIGURE 21.8).
Radioactivity can also be detected and measured by a Geiger counter. The operation of this device is based on the fact that radiation is able to ionize matter. The ions and electrons produced by the ionizing radiation permit conduction of an electrical current. The basic design of a Geiger counter is shown in ![]() FIGURE 21.9. A current pulse between the anode and the metal cylinder occurs whenever entering radiation produces ions. Each pulse is counted in order to estimate the amount of radiation.
FIGURE 21.9. A current pulse between the anode and the metal cylinder occurs whenever entering radiation produces ions. Each pulse is counted in order to estimate the amount of radiation.

![]() FIGURE 21.8 Badge dosimeters monitor the extent to which the individual has been exposed to high-energy radiation. The radiation dose is determined from the extent of darkening of the film in the dosimeter.
FIGURE 21.8 Badge dosimeters monitor the extent to which the individual has been exposed to high-energy radiation. The radiation dose is determined from the extent of darkening of the film in the dosimeter.
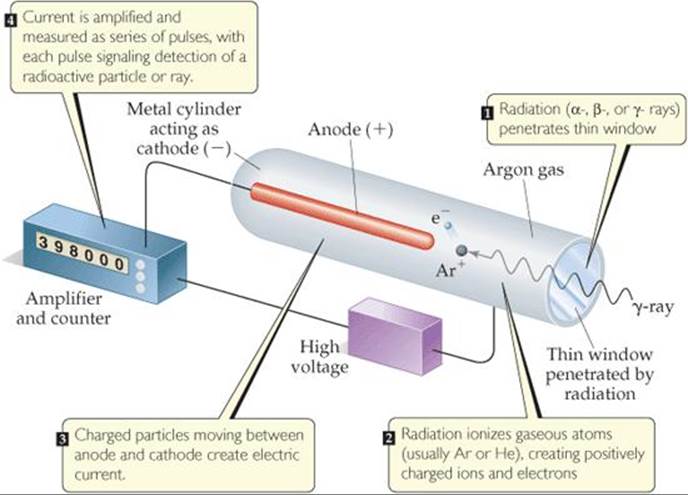
![]() FIGURE 21.9 Schematic drawing of a Geiger counter.
FIGURE 21.9 Schematic drawing of a Geiger counter.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Will alpha, beta, and gamma rays pass through the window of a Geiger counter with equal efficiency?
Substances that are electronically excited by radiation can also be used to detect and measure radiation. For example, some substances excited by radiation give off light as electrons return to their lower-energy states. These substances are called phosphors. Different substances respond to different particles. Zinc sulfide, for example, responds to alpha particles. An instrument called a scintillation counter is used to detect and measure radiation, based on the tiny flashes of light produced when radiation strikes a suitable phosphor. The flashes of light are magnified electronically and counted to measure the amount of radiation.
Radiotracers
Because radioisotopes can be detected readily, they can be used to follow an element through its chemical reactions. The incorporation of carbon atoms from CO2 into glucose during photosynthesis, for example, has been studied using CO2 enriched in carbon-14:
![]()
The use of the carbon-14 label provides direct experimental evidence that carbon dioxide in the environment is chemically converted to glucose in plants. Analogous labeling experiments using oxygen-18 show that the O2 produced during photosynthesis comes from water, not carbon dioxide. When it is possible to isolate and purify intermediates and products from reactions, detection devices such as scintillation counters can be used to “follow” the radioisotope as it moves from starting material through intermediates to final product. These types of experiments are useful for identifying elementary steps in a reaction mechanism. ![]() (Section 14.6)
(Section 14.6)
The use of radioisotopes is possible because all isotopes of an element have essentially identical chemical properties. When a small quantity of a radioisotope is mixed with the naturally occurring stable isotopes of the same element, all the isotopes go through the same reactions together. The element's path is revealed by the radioactivity of the radioisotope. Because the radioisotope can be used to trace the path of the element, it is called a radiotracer.
 CHEMISTRY AND LIFE
CHEMISTRY AND LIFE
MEDICAL APPLICATIONS OF RADIOTRACERS
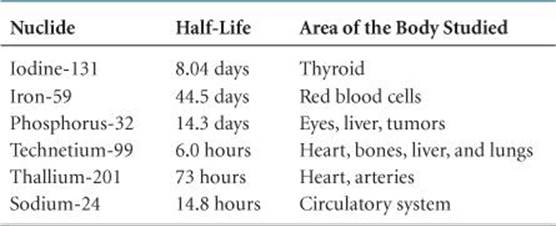
Radiotracers have found wide use as diagnostic tools in medicine. ![]() TABLE 21.6 lists some radiotracers and their uses. These radioisotopes are incorporated into a compound that is administered to the patient, usually intravenously. The diagnostic use of these isotopes is based on the ability of the radioactive compound to localize and concentrate in the organ or tissue under investigation. Iodine-131, for example, has been used to test the activity of the thyroid gland. This gland is the only important user of iodine in the body. The patient drinks a solution of NaI containing iodine-131. Only a very small amount is used so that the patient does not receive a harmful dose of radioactivity. A Geiger counter placed close to the thyroid, in the neck region, determines the ability of the thyroid to take up the iodine. A normal thyroid will absorb about 12% of the iodine within a few hours.
TABLE 21.6 lists some radiotracers and their uses. These radioisotopes are incorporated into a compound that is administered to the patient, usually intravenously. The diagnostic use of these isotopes is based on the ability of the radioactive compound to localize and concentrate in the organ or tissue under investigation. Iodine-131, for example, has been used to test the activity of the thyroid gland. This gland is the only important user of iodine in the body. The patient drinks a solution of NaI containing iodine-131. Only a very small amount is used so that the patient does not receive a harmful dose of radioactivity. A Geiger counter placed close to the thyroid, in the neck region, determines the ability of the thyroid to take up the iodine. A normal thyroid will absorb about 12% of the iodine within a few hours.
TABLE 21.6 • Some Radionuclides Used as Radiotracers
The medical applications of radiotracers are further illustrated by positron emission tomography (PET). PET is used for clinical diagnosis of many diseases. In this method, compounds containing radionuclides that decay by positron emission are injected into a patient. These compounds are chosen to enable researchers to monitor blood flow, oxygen and glucose metabolic rates, and other biological functions. Some of the most interesting work involves the study of the brain, which depends on glucose for most of its energy. Changes in how this sugar is metabolized or used by the brain may signal a disease such as cancer, epilepsy, Parkinson's disease, or schizophrenia.
The compound to be detected in the patient must be labeled with a radionuclide that is a positron emitter. The most widely used nuclides are carbon-11 (t1/2 = 20.4 min), fluorine-18 (t1/2 = 110 min), oxygen-15 (t1/2 = 2 min), and nitrogen-13 (t1/2 = 10 min). Glucose, for example, can be labeled with carbon-11. Because the half-lives of positron emitters are so short, the chemist must quickly incorporate the radionuclide into the sugar (or other appropriate) molecule and inject the compound immediately. The patient is placed in an instrument that measures the positron emission and constructs a computer-based image of the organ in which the emitting compound is localized. When the element decays, the emitted positron quickly collides with an electron. The positron and electron are annihilated in the collision, producing two gamma rays that move in opposite directions. The gamma rays are detected by an encircling ring of scintillation counters (![]() FIGURE 21.10). Because the rays move in opposite directions but were created in the same place at the same time, it is possible to accurately locate the point in the body where the radioactive isotope decayed. The nature of this image provides clues to the presence of disease or other abnormality and helps medical researchers understand how a particular disease affects the functioning of the brain. For example, the images shown in
FIGURE 21.10). Because the rays move in opposite directions but were created in the same place at the same time, it is possible to accurately locate the point in the body where the radioactive isotope decayed. The nature of this image provides clues to the presence of disease or other abnormality and helps medical researchers understand how a particular disease affects the functioning of the brain. For example, the images shown in ![]() FIGURE 21.11 reveal that levels of activity in brains of patients with Alzheimer's disease are different from the levels in those without the disease.
FIGURE 21.11 reveal that levels of activity in brains of patients with Alzheimer's disease are different from the levels in those without the disease.
RELATED EXERCISES: 21.53, 21.54
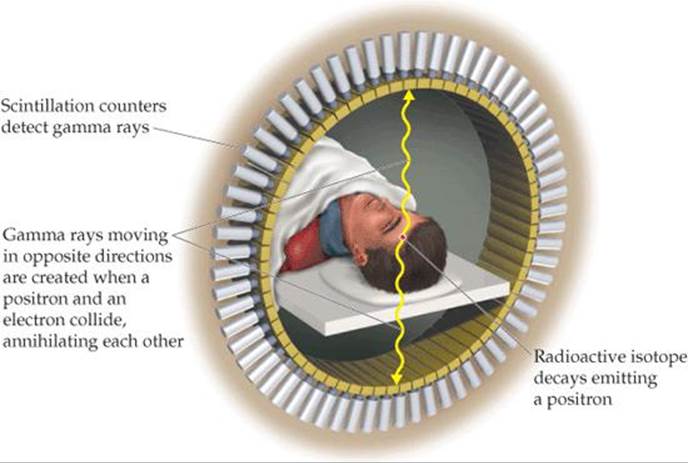
![]() FIGURE 21.10 Schematic representation of a positron emission tomography (PET) scanner.
FIGURE 21.10 Schematic representation of a positron emission tomography (PET) scanner.
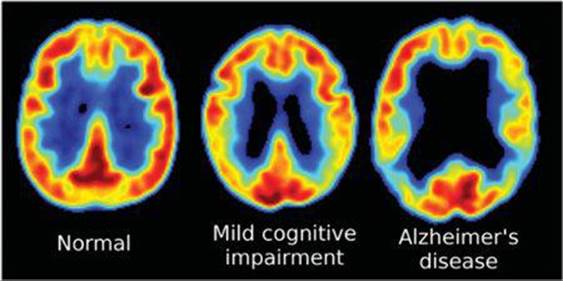
![]() FIGURE 21.11 Positron emission tomography (PET) scans showing glucose metabolism levels in the brain. Red and yellow colors show higher levels of glucose metabolism.
FIGURE 21.11 Positron emission tomography (PET) scans showing glucose metabolism levels in the brain. Red and yellow colors show higher levels of glucose metabolism.