CHEMISTRY THE CENTRAL SCIENCE
22 CHEMISTRY OF THE NONMETALS
22.4 GROUP 7A: THE HALOGENS
The elements of group 7A, the halogens, have the outer-electron configuration ns2np5, where n ranges from 2 through 6. The halogens have large negative electron affinities ![]() (Section 7.5), and they most often achieve a noble-gas configuration by gaining an electron, which results in a – 1 oxidation state. Fluorine, being the most electronegative element, exists in compounds only in the –1 state. The other halogens exhibit positive oxidation states up to +7 in combination with more electronegative atoms such as O. In the positive oxidation states, the halogens tend to be good oxidizing agents, readily accepting electrons.
(Section 7.5), and they most often achieve a noble-gas configuration by gaining an electron, which results in a – 1 oxidation state. Fluorine, being the most electronegative element, exists in compounds only in the –1 state. The other halogens exhibit positive oxidation states up to +7 in combination with more electronegative atoms such as O. In the positive oxidation states, the halogens tend to be good oxidizing agents, readily accepting electrons.
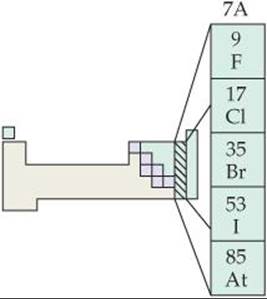
Chlorine, bromine, and iodine are found as the halides in seawater and in salt deposits. Fluorine occurs in the minerals fluorspar (CaF2), cryolite (Na3AlF6), and fluorapatite [Ca5(PO4)3F].* Only fluorspar is an important commercial source of fluorine.
All isotopes of astatine are radioactive. The longest-lived isotope is astatine-210, which has a half-life of 8.1 hr and decays mainly by electron capture. Because astatine is so unstable, very little is known about its chemistry.
Properties and Production of the Halogens
Most properties of the halogens vary in a regular fashion as we go from fluorine to iodine (![]() TABLE 22.2).
TABLE 22.2).
Under ordinary conditions the halogens exist as diatomic molecules. The molecules are held together in the solid and liquid states by dispersion forces. ![]() (Section 11.2) Because I2 is the largest and most polarizable halogen molecule, the intermolecular forces between I2 molecules are the strongest. Thus, I2 has the highest melting point and boiling point. At room temperature and 1 atm, I2 is a purple solid, Br2 is a red-brown liquid, and Cl2 and F2 are gases.
(Section 11.2) Because I2 is the largest and most polarizable halogen molecule, the intermolecular forces between I2 molecules are the strongest. Thus, I2 has the highest melting point and boiling point. At room temperature and 1 atm, I2 is a purple solid, Br2 is a red-brown liquid, and Cl2 and F2 are gases. ![]() (Figure 7.27) Chlorine readily liquefies upon compression at room temperature and is normally stored and handled in liquid form under pressure in steel containers.
(Figure 7.27) Chlorine readily liquefies upon compression at room temperature and is normally stored and handled in liquid form under pressure in steel containers.
The comparatively low bond enthalpy of F2 (155 kJ/mol) accounts in part for the extreme reactivity of elemental fluorine. Because of its high reactivity, F2 is difficult to work with. Certain metals, such as copper and nickel, can be used to contain F2 because their surfaces form a protective coating of metal fluoride. Chlorine and the heavier halogens are also reactive, although less so than fluorine.
Because of their high electronegativities, the halogens tend to gain electrons from other substances and thereby serve as oxidizing agents. The oxidizing ability of the halogens, indicated by their standard reduction potentials, decreases going down the group. As a result, a given halogen is able to oxidize the halide anions below it. For example, Cl2 oxidizes Br– and I– but not F–, as seen in ![]() FIGURE 22.8.
FIGURE 22.8.
![]() GO FIGURE
GO FIGURE
Do Br2 and I2 appear to be more or less soluble in CCl4 than in H2O?
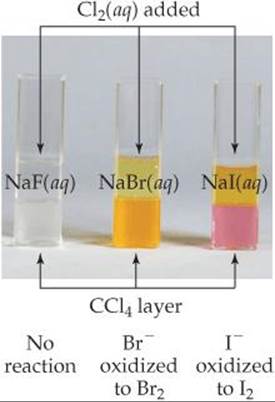
![]() FIGURE 22.8 Reaction of Cl2 with aqueous solutions of NaF, NaBr, and NaI. The top liquid layer is water; the bottom liquid layer is carbon tetrachloride.
FIGURE 22.8 Reaction of Cl2 with aqueous solutions of NaF, NaBr, and NaI. The top liquid layer is water; the bottom liquid layer is carbon tetrachloride.
TABLE 22.2 • Some Properties of the Halogens
SAMPLE EXERCISE 22.4 Predicting Chemical Reactions among the Halogens
Write the balanced equation for the reaction, if any, between (a) I–(aq) and Br2(l), (b) Cl–(aq) and I2(s).
SOLUTION
Analyze We are asked to determine whether a reaction occurs when a particular halide and halogen are combined.
Plan A given halogen is able to oxidize anions of the halogens below it in the periodic table. Thus, in each pair the halogen having the smaller atomic number ends up as the halide ion. If the halogen with the smaller atomic number is already the halide, there is no reaction. Thus, the key to determining whether a reaction occurs is locating the elements in the periodic table.
Solve
(a) Br2 can oxidize (remove electrons from) the anions of the halogens below it in the periodic table. Thus, it oxidizes I–:
![]()
(b) Cl– is the anion of a halogen above iodine in the periodic table. Thus, I2 cannot oxidize Cl–; there is no reaction.
PRACTICE EXERCISE
Write the balanced chemical equation for the reaction between Br–(aq) and Cl2(aq).
Answer: ![]()
Notice in Table 22.2 that the standard reduction potential of F2 is exceptionally high. Fluorine gas readily oxidizes water:
![]()
Fluorine cannot be prepared by electrolytic oxidation of aqueous solutions of fluoride salts because water is oxidized more readily than F–. ![]() (Section 20.9) In practice, the element is formed by electrolytic oxidation of a solution of KF in anhydrous HF.
(Section 20.9) In practice, the element is formed by electrolytic oxidation of a solution of KF in anhydrous HF.
Chlorine is produced mainly by electrolysis of either molten or aqueous sodium chloride. Both bromine and iodine are obtained commercially from brines containing the halide ions; the reaction used is oxidation with Cl2.
Uses of the Halogens
Fluorine is used to prepare fluorocarbons—very stable carbon-fluorine compounds used as refrigerants, lubricants, and plastics. Teflon® (![]() FIGURE 22.9) is a polymeric fluorocarbon noted for its high thermal stability and lack of chemical reactivity.
FIGURE 22.9) is a polymeric fluorocarbon noted for its high thermal stability and lack of chemical reactivity.
![]() GO FIGURE
GO FIGURE
What is the repeating unit in this polymer?
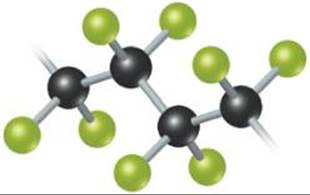
![]() FIGURE 22.9 Structure of Teflon®, a fluorocarbon polymer.
FIGURE 22.9 Structure of Teflon®, a fluorocarbon polymer.
Chlorine is by far the most commercially important halogen. About 1 × 1010 kg (10 million tons) of Cl2 is produced annually in the United States. In addition, hydrogen chloride production is about 4.0 × 109 kg (4.4 million tons) annually. About half of this chlorine finds its way eventually into the manufacture of chlorine-containing organic compounds, such as the vinyl chloride (C2H3Cl) used in making polyvinyl chloride (PVC) plastics. ![]() (Section 12.8) Much of the remainder is used as a bleaching agent in the paper and textile industries. When Cl2 dissolves in cold dilute base, it converts into Cl– and hypochlorite, ClO–:
(Section 12.8) Much of the remainder is used as a bleaching agent in the paper and textile industries. When Cl2 dissolves in cold dilute base, it converts into Cl– and hypochlorite, ClO–:
![]()
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What is the oxidation state of Cl in each Cl species in Equation 22.19?
Sodium hypochlorite (NaClO) is the active ingredient in many liquid bleaches. Chlorine is also used in water treatment to oxidize and thereby destroy bacteria. ![]() (Section 18.4)
(Section 18.4)
A common use of iodine is as KI in table salt. Iodized salt provides the small amount of iodine necessary in our diets; it is essential for the formation of thyroxin, a hormone secreted by the thyroid gland. Lack of iodine in the diet results in an enlarged thyroid gland, a condition calledgoiter.
The Hydrogen Halides
All the halogens form stable diatomic molecules with hydrogen. Aqueous solutions of HCl, HBr, and HI are strong acids. The hydrogen halides can be formed by direct reaction of the elements. The most important means of preparing HF and HCl, however, is by reacting a salt of the halide with a strong nonvolatile acid, as in the reaction
![]()
Neither HBr nor HI can be prepared in this way, however, because H2SO4 oxidizes Br– and I– (![]() FIGURE 22.10). This difference in reactivity reflects the greater ease of oxidation of Br– and I– relative to F– and Cl–. These undesirable oxidations are avoided by using a nonvolatile acid, such as H3PO4, that is a weaker oxidizing agent than H2SO4.
FIGURE 22.10). This difference in reactivity reflects the greater ease of oxidation of Br– and I– relative to F– and Cl–. These undesirable oxidations are avoided by using a nonvolatile acid, such as H3PO4, that is a weaker oxidizing agent than H2SO4.
SAMPLE EXERCISE 22.5 Writing a Balanced Chemical Equation
Write a balanced equation for the formation of hydrogen bromide gas from the reaction of solid sodium bromide with phosphoric acid.
SOLUTION
Analyze We are asked to write a balanced equation for the reaction between NaBr and H3PO4 to form HBr and another product.
Plan As in Equation 22.20, a metathesis reaction takes place. ![]() (Section 4.2) Let's assume that only one H in H3PO4 reacts. (The actual number depends on the reaction conditions.) The H2PO4– and Na+ will form NaH2PO4 as one product.
(Section 4.2) Let's assume that only one H in H3PO4 reacts. (The actual number depends on the reaction conditions.) The H2PO4– and Na+ will form NaH2PO4 as one product.
Solve The balanced equation is
![]()
PRACTICE EXERCISE
Write the balanced equation for the preparation of HI from NaI and H3PO4.
Answer: ![]()
![]() GO FIGURE
GO FIGURE
Are these reactions acid-base reactions or oxidation-reduction reactions?

![]() FIGURE 22.10 Reaction of H2SO4 with NaI and NaBr.
FIGURE 22.10 Reaction of H2SO4 with NaI and NaBr.
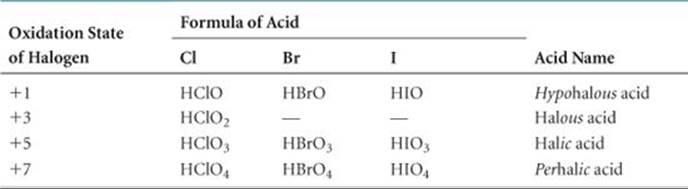
TABLE 22.3 • The Stable Oxyacids of the Halogens
The hydrogen halides form hydrohalic acid solutions when dissolved in water. These solutions have the characteristic properties of acids, such as reactions with active metals to produce hydrogen gas. ![]() (Section 4.4) Hydrofluoric acid also reacts readily with silica (SiO2) and with silicates to form hexafluorosilicic acid (H2SiF6):
(Section 4.4) Hydrofluoric acid also reacts readily with silica (SiO2) and with silicates to form hexafluorosilicic acid (H2SiF6):
![]()
Interhalogen Compounds
Because the halogens exist as diatomic molecules, diatomic molecules made up of two different halogen atoms exist. These compounds are the simplest examples of interhalogens, compounds, such as ClF and IF5, formed between two halogen elements.
With one exception, the higher interhalogen compounds have a central Cl, Br, or I atom surrounded by 3, 5, or 7 fluorine atoms. The large size of the iodine atom allows the formation of IF3, IF5, and IF7, in which the oxidation state of I is +3, +5, and +7, respectively. With the smaller bromine and chorine atoms, only compounds with 3 or 5 fluorines form. The only higher interhalogen compounds that do not have outer F atoms are ICl3 and ICl5; the large size of the I atom can accommodate 5 Cl atoms, whereas Br is not large enough to allow even BrCl3 to form. All of the interhalogen compounds are powerful oxidizing agents.
Oxyacids and Oxyanions
![]() TABLE 22.3 summarizes the formulas of the known oxyacids of the halogens and the way they are named.*
TABLE 22.3 summarizes the formulas of the known oxyacids of the halogens and the way they are named.* ![]() (Section 2.8) The acid strengths of the oxyacids increase with increasing oxidation state of the central halogen atom.
(Section 2.8) The acid strengths of the oxyacids increase with increasing oxidation state of the central halogen atom. ![]() (Section 16.10) All the oxyacids are strong oxidizing agents. The oxyanions, formed on removal of H+ from the oxyacids, are generally more stable than the oxyacids. Hypochlorite salts are used as bleaches and disinfectants because of the powerful oxidizing capabilities of the ClO– ion. Sodium hypochlorite is used as a bleaching agent. Chlorate salts are similarly very reactive. For example, potassium chlorate is used to make matches and fireworks.
(Section 16.10) All the oxyacids are strong oxidizing agents. The oxyanions, formed on removal of H+ from the oxyacids, are generally more stable than the oxyacids. Hypochlorite salts are used as bleaches and disinfectants because of the powerful oxidizing capabilities of the ClO– ion. Sodium hypochlorite is used as a bleaching agent. Chlorate salts are similarly very reactive. For example, potassium chlorate is used to make matches and fireworks.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Which do you expect to be the stronger oxidizing agent, NaBrO3 or NaClO3?
Perchloric acid and its salts are the most stable oxyacids and oxyanions. Dilute solutions of perchloric acid are quite safe, and many perchlorate salts are stable except when heated with organic materials. When heated, however, perchlorates can become vigorous, even violent, oxidizers. Considerable caution should be exercised, therefore, when handling these substances, and it is crucial to avoid contact between perchlorates and readily oxidized material. The use of ammonium perchlorate (NH4ClO4) as the oxidizer in the solid booster rockets for the Space Shuttle demonstrates the oxidizing power of perchlorates. The solid propellant contains a mixture of NH4ClO4 and powdered aluminum, the reducing agent. Each shuttle launch requires about 6 × 105 kg (700 tons) of NH4ClO4 (![]() FIGURE 22.11).
FIGURE 22.11).

![]() FIGURE 22.11 Launch of the Space Shuttle Columbia from the Kennedy Space Center.
FIGURE 22.11 Launch of the Space Shuttle Columbia from the Kennedy Space Center.
 CHEMISTRY AND LIFE
CHEMISTRY AND LIFE
HOW MUCH PERCHLORATE IS TOO MUCH?
Since the 1950s both NASA and the Pentagon have used ammonium perchlorate, NH4ClO4, as a rocket fuel. The result is that traces of perchlorate ion are found in groundwater in many regions of the United States, with levels ranging from about 4 to 100 ppb.
Perchlorate is known to suppress thyroid hormone levels in humans. However, it is disputed whether the amounts found in drinking water are sufficiently high to cause health problems. The Environmental Protection Agency currently states that a dose of 0.007 mg per kilogram of body mass per day is not expected to cause adverse health effects in humans. For a 70-kg (154-lb.) person drinking 2 L of water per day, that amounts to a concentration of 25 ppb. California has proposed a standard of 6 ppb.
Removal of perchlorate ion from water supplies is not easy. Although perchlorate is an oxidizing agent, the ClO4– ion is quite stable in aqueous solution. One promising avenue is reduction by microorganisms. While research continues on the best means of decreasing perchlorate levels in drinking water, federal agencies continue to explore what level constitutes a safe upper limit.
RELATED EXERCISE: 22.93