CHEMISTRY THE CENTRAL SCIENCE
22 CHEMISTRY OF THE NONMETALS
22.11 BORON
Boron is the only group 3A element that can be considered nonmetallic and thus is our final element in this chapter. The element has an extended network structure with a melting point (2300 °C) that is intermediate between the melting points of carbon (3550 °C) and silicon (1410°C). The electron configuration of boron is [He]2s2 2p1.
In the family of compounds called boranes, the molecules contain only boron and hydrogen. The simplest borane is BH3. This molecule contains only six valence electrons and is therefore an exception to the octet rule. As a result, BH3 reacts with itself to form diborane (B2H6). This reaction can be viewed as a Lewis acid-base reaction in which one B — H bonding pair of electrons in each BH3 molecule is donated to the other. As a result, diborane is an unusual molecule in which hydrogen atoms form a bridge between two B atoms (![]() FIGURE 22.36). Such hydrogens, called bridging hydrogens, exhibit interesting chemical reactivity, which you may learn about in a more advanced chemistry course.
FIGURE 22.36). Such hydrogens, called bridging hydrogens, exhibit interesting chemical reactivity, which you may learn about in a more advanced chemistry course.
![]() FIGURE 22.36 The structure of diborane (B2H6).
FIGURE 22.36 The structure of diborane (B2H6).
Sharing hydrogen atoms between the two boron atoms compensates somewhat for the deficiency in valence electrons around each boron. Nevertheless, diborane is an extremely reactive molecule, spontaneously flammable in air in a highly exothermic reaction:
![]()
Boron and hydrogen form a series of anions called borane anions. Salts of the borohydride ion (BH4–) are widely used as reducing agents. For example, sodium borohydride (NaBH4) is a commonly used reducing agent for certain organic compounds.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Recall that the hydride ion is HT. What is the oxidation state of boron in sodium borohydride?
The only important oxide of boron is boric oxide (B2O3). This substance is the anhydride of boric acid, which we may write as H3BO3 or B(OH)3. Boric acid is so weak an acid (Ka = 5.8 × 10–10) that solutions of H3BO3 are used as an eyewash. Upon heating, boric acid loses water by a condensation reaction similar to that described for phosphorus in Section 22.8:
![]()
The diprotic acid H2B4O7 is tetraboric acid. The hydrated sodium salt Na2B4O7 · 10 H2O, called borax, occurs in dry lake deposits in California and can also be prepared from other borate minerals. Solutions of borax are alkaline, and the substance is used in various laundry and cleaning products.
SAMPLE INTEGRATIVE EXERCISE Putting Concepts Together
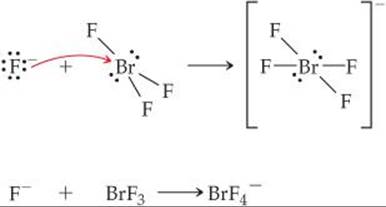
The interhalogen compound BrF3 is a volatile, straw-colored liquid. The compound exhibits appreciable electrical conductivity because of autoionization (“solv” refers to BrF3 as the solvent):
![]()
(a) What are the molecular structures of the BrF2+ and BrF4– ions? (b) The electrical conductivity of BrF3 decreases with increasing temperature. Is the autoionization process exothermic or endothermic? (c) One chemical characteristic of BrF3 is that it acts as a Lewis acid toward fluoride ions. What do we expect will happen when KBr is dissolved in BrF3?
SOLUTION
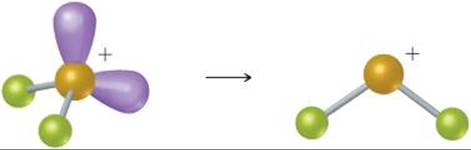
(a) The BrF2+ ion has 7 + 2(7) - 1 = 20 valence-shell electrons. The Lewis structure for the ion is
![]()
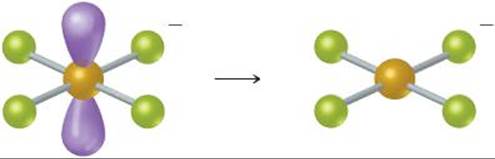
Because there are four electron domains around the central Br atom, the resulting electron domain geometry is tetrahedral. ![]() (Section 9.2) Because bonding pairs of electrons occupy two of these domains, the molecular geometry is nonlinear:
(Section 9.2) Because bonding pairs of electrons occupy two of these domains, the molecular geometry is nonlinear:
The BrF4– ion has 7 + 4(7) + 1 = 36 electrons, leading to the Lewis structure
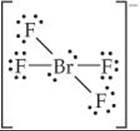
Because there are six electron domains around the central Br atom in this ion, the geometry is octahedral. The two nonbonding pairs of electrons are located opposite each other on the octahedron, leading to a square-planar molecular geometry:
(b) The observation that conductivity decreases as temperature increases indicates that there are fewer ions present in the solution at the higher temperature. Thus, increasing the temperature causes the equilibrium to shift to the left. According to Le Châtelier's principle, this shift indicates that the reaction is exothermic as it proceeds from left to right. ![]() (Section 15.7)
(Section 15.7)
(c) A Lewis acid is an electron-pair acceptor. ![]() (Section 16.11) The fluoride ion has four valence-shell electron pairs and can act as a Lewis base (an electron-pair donor). Thus, we can envision the following reaction occurring:
(Section 16.11) The fluoride ion has four valence-shell electron pairs and can act as a Lewis base (an electron-pair donor). Thus, we can envision the following reaction occurring: