CHEMISTRY THE CENTRAL SCIENCE
24 THE CHEMISTRY OF LIFE: ORGANIC AND BIOLOGICAL CHEMISTRY
24.2 INTRODUCTION TO HYDROCARBONS
Because carbon compounds are so numerous, it is convenient to organize them into families that have structural similarities. The simplest class of organic compounds is the hydrocarbons, compounds composed of only carbon and hydrogen. The key structural feature of hydrocarbons (and of most other organic substances) is the presence of stable carbon-carbon bonds. Carbon is the only element capable of forming stable, extended chains of atoms bonded through single, double, or triple bonds.
Hydrocarbons can be divided into four types, depending on the kinds of carbon-carbon bonds in their molecules. ![]() TABLE 24.1 shows an example of each type.
TABLE 24.1 shows an example of each type.
Alkanes contain only single bonds. Alkenes, also known as olefins, contain at least one C≡C double bond, and alkynes contain at least one C═C triple bond. In aromatic hydrocarbons the carbon atoms are connected in a planar ring structure, joined by both σ and delocalized π bonds between carbon atoms. Benzene (C6H6) is the best-known example of an aromatic hydrocarbon.
Each type of hydrocarbon exhibits different chemical behaviors, as we will see shortly. The physical properties of all four types, however, are similar in many ways. Because hydrocarbon molecules are relatively nonpolar, they are almost completely insoluble in water but dissolve readily in nonpolar solvents. Their melting points and boiling points are determined by dispersion forces. ![]() (Section 11.2) As a result, hydrocarbons of very low molecular weight, such as C2H6(bp = –89 °C), are gases at room temperature; those of moderate molecular weight, such as C6H14(bp = 69 °C), are liquids; and those of high molecular weight, such as C22H46(mp = 44 °C), are solids.
(Section 11.2) As a result, hydrocarbons of very low molecular weight, such as C2H6(bp = –89 °C), are gases at room temperature; those of moderate molecular weight, such as C6H14(bp = 69 °C), are liquids; and those of high molecular weight, such as C22H46(mp = 44 °C), are solids.
![]() TABLE 24.2 lists the ten simplest alkanes. Many of these substances are familiar because they are used so widely. Methane is a major component of natural gas and is used for home heating and in gas stoves and water heaters. Propane is the major component of bottled gas used for home heating and cooking in areas where natural gas is not available. Butane is used in disposable lighters and in fuel canisters for gas camping stoves and lanterns. Alkanes with from 5 to 12 carbon atoms per molecule are used to make gasoline. Notice that each succeeding compound inTable 24.2 has an additional CH2 unit.
TABLE 24.2 lists the ten simplest alkanes. Many of these substances are familiar because they are used so widely. Methane is a major component of natural gas and is used for home heating and in gas stoves and water heaters. Propane is the major component of bottled gas used for home heating and cooking in areas where natural gas is not available. Butane is used in disposable lighters and in fuel canisters for gas camping stoves and lanterns. Alkanes with from 5 to 12 carbon atoms per molecule are used to make gasoline. Notice that each succeeding compound inTable 24.2 has an additional CH2 unit.
TABLE 24.1 • The Four Hydrocarbon Types
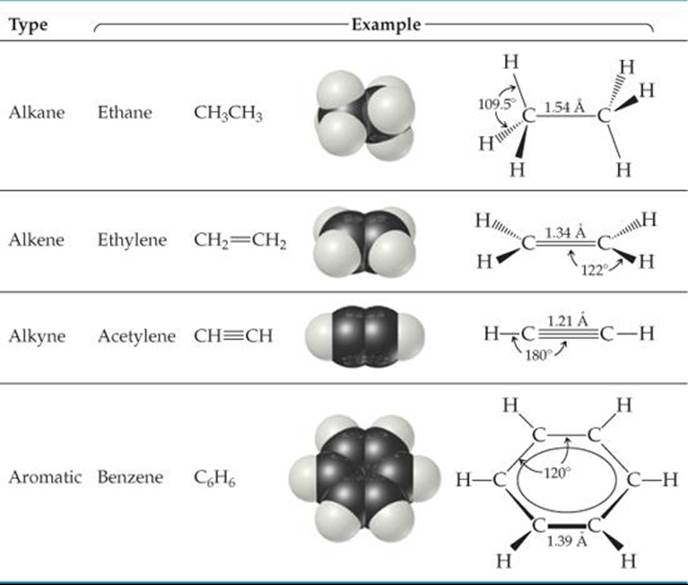
TABLE 24.2 • First Ten Members of the Straight-Chain Alkane Series
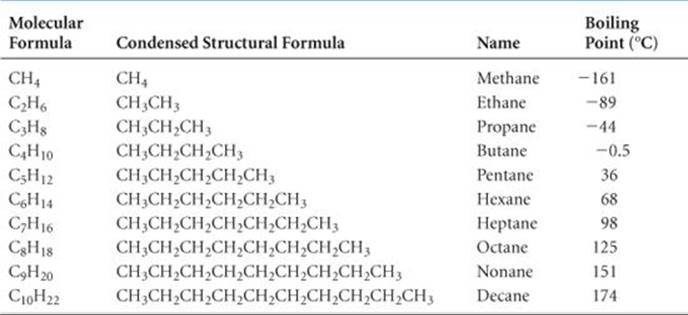
The formulas for the alkanes given in Table 24.2 are written in a notation called condensed structural formulas. This notation reveals the way in which atoms are bonded to one another but does not require drawing in all the bonds. For example, the structural formula and the condensed structural formulas for butane (C4H10) are

![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
How many C—H and C—C bonds are formed by the middle carbon atom of propane?
Structures of Alkanes
According to the VSEPR model, the molecular geometry about each carbon atom in an alkane is tetrahedral. ![]() (Section 9.2) The bonding may be described as involving sp3 -hybridized orbitals on the carbon, as pictured in
(Section 9.2) The bonding may be described as involving sp3 -hybridized orbitals on the carbon, as pictured in ![]() FIGURE 24.3 for methane.
FIGURE 24.3 for methane. ![]() (Section 9.5)
(Section 9.5)
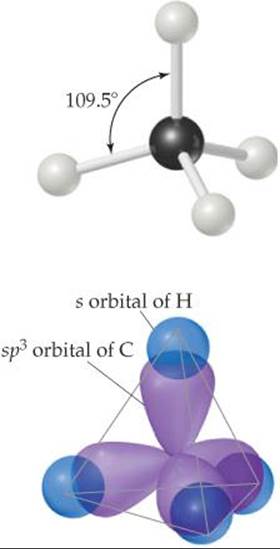
![]() FIGURE 24.3 Bonds about carbon in methane. This tetrahedral molecular geometry is found around all carbons in alkanes.
FIGURE 24.3 Bonds about carbon in methane. This tetrahedral molecular geometry is found around all carbons in alkanes.
Rotation about a carbon-carbon single bond is relatively easy and occurs rapidly at room temperature. To visualize such rotation, imagine grasping either methyl group of the propane molecule in ![]() FIGURE 24.4 and rotating the group relative to the rest of the molecule. Because motion of this sort occurs rapidly in alkanes, a long-chain alkane molecule is constantly undergoing motions that cause it to change its shape, something like a length of chain that is being shaken.
FIGURE 24.4 and rotating the group relative to the rest of the molecule. Because motion of this sort occurs rapidly in alkanes, a long-chain alkane molecule is constantly undergoing motions that cause it to change its shape, something like a length of chain that is being shaken.
Structural Isomers
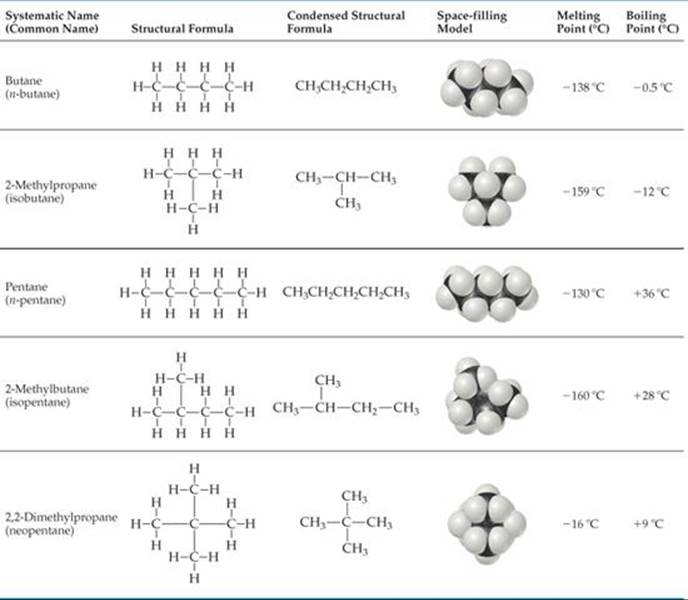
The alkanes in Table 24.2 are called straight-chain hydrocarbons because all the carbon atoms are joined in a continuous chain. Alkanes consisting of four or more carbon atoms can also form branched chains, and when they do, they are called branched-chain hydrocarbons. (The branches in organic molecules are often called side chains.) ![]() TABLE 24.3, for example, shows all the straight-chain and branched-chain alkanes containing four and five carbon atoms.
TABLE 24.3, for example, shows all the straight-chain and branched-chain alkanes containing four and five carbon atoms.
Compounds that have the same molecular formula but different bonding arrangements (and hence different structures) are called structural isomers. Thus, C4H10 has two structural isomers and C5H12 has three. The structural isomers of a given alkane differ slightly from one another in physical properties, as the melting and boiling points in Table 24.3 indicate.
The number of possible structural isomers increases rapidly with the number of carbon atoms in the alkane. There are 18 isomers with the molecular formula C8H18, for example, and 75 with the molecular formula C10H22.
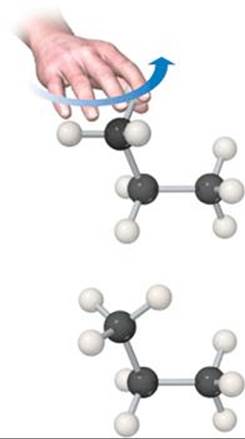
![]() FIGURE 24.4 Rotation about a C—C bond occurs easily and rapidly in all alkanes.
FIGURE 24.4 Rotation about a C—C bond occurs easily and rapidly in all alkanes.
TABLE 24.3 • Isomers of C4H10 and C5H12
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What evidence can you cite to support the fact that although isomers have the same molecular formula they are in fact different compounds?
Nomenclature of Alkanes
In the first column of Table 24.3, the names in parentheses are called the common names. The common name of the isomer with no branches begins with the letter n (indicating the “normal” structure). When one CH3 group branches off the major chain, the common name of the isomer begins with iso-, and when two CH3 groups branch off, the common name begins with neo-. As the number of isomers grows, however, it becomes impossible to find a suitable prefix to denote each isomer by a common name. The need for a systematic means of naming organic compounds was recognized as early as 1892, when an organization called the International Union of Chemistry met in Geneva to formulate rules for naming organic substances. Since that time the task of updating the rules for naming compounds has fallen to the International Union of Pure and Applied Chemistry (IUPAC). Chemists everywhere, regardless of their nationality, subscribe to a common system for naming compounds.
The IUPAC names for the isomers of butane and pentane are the ones given first in Table 24.3. These systematic names, as well as those of other organic compounds, have three parts to them:
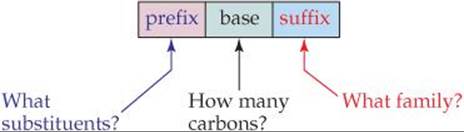
The following steps summarize the procedures used to name alkanes, which all have names ending with -ane. We use a similar approach to write the names of other organic compounds.
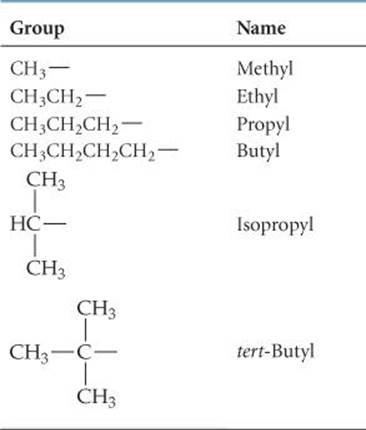
1. Find the longest continuous chain of carbon atoms, and use the name of this chain (given in Table 24.2) as the base name. Be careful in this step because the longest chain may not be written in a straight line, as in the following structure:
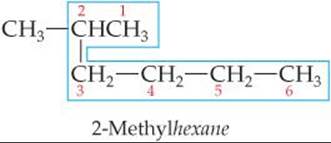
Because the longest continuous chain contains six C atoms, this isomer is named as a substituted hexane. Groups attached to the main chain are called substituents because they are substituted in place of an H atom on the main chain. In this molecule the CH3 group not enclosed by the blue outline is the only substituent in the molecule.
2. Number the carbon atoms in the longest chain, beginning with the end nearest a substituent. In our example, we number the C atoms beginning at the upper right because that places the CH3 substituent on C2 of the chain. (If we had numbered from the lower right, the CH3would be on C5.) The chain is numbered from the end that gives the lower number to the substituent position.
3. Name each substituent. A substituent formed by removing an H atom from an alkane is called an alkyl group. Alkyl groups are named by replacing the -one ending of the alkane name with -yl. The methyl group (CH3), for example, is derived from methane (CH4) and the ethyl group (C2H5) is derived from ethane (C2H6). ![]() TABLE 24.4 lists six common alkyl groups.
TABLE 24.4 lists six common alkyl groups.
4. Begin the name with the number or numbers of the carbon or carbons to which each substituent is bonded. For our compound, the name 2-methylhexane indicates the presence of a methyl group on C2 of a hexane (six-carbon) chain.
5. When two or more substituents are present, list them in alphabetical order. The presence of two or more of the same substituent is indicated by the prefixes di- (two), tri- (three), tetra- (four), penta- (five), and so forth. The prefixes are ignored in determining the alphabetical order of the substituents:
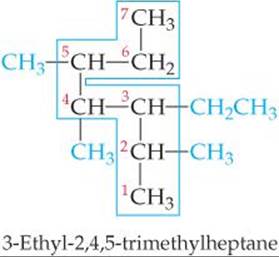
TABLE 24.4 • Condensed Structural Formulas and Common Names for Several Alkyl Groups
SAMPLE EXERCISE 24.1 Naming Alkanes
Give the systematic name for the following alkane:
SOLUTION
Analyze We are given the condensed structural formula of an alkane and asked to give its name.
Plan Because the hydrocarbon is an alkane, its name ends in -ane. The name of the parent hydrocarbon is based on the longest continuous chain of carbon atoms. Branches are alkyl groups, named after the number of C atoms in the branch and located by counting C atoms along the longest continuous chain.
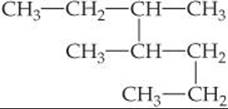
Solve The longest continuous chain of C atoms extends from the upper left CH3 group to the lower left CH3 group and is seven C atoms long:
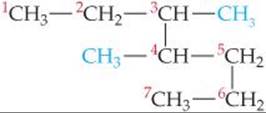
The parent compound is thus heptane. There are two methyl groups branching off the main chain. Hence, this compound is a dimethylheptane. To specify the location of the two methyl groups, we must number the C atoms from the end that gives the lower two numbers to the carbons bearing side chains. This means that we should start numbering at the upper left carbon. There is a methyl group on C3 and one on C4. The compound is thus 3,4-dimethylheptane.
PRACTICE EXERCISE
Name the following alkane:
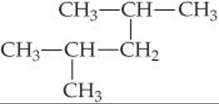
Answer: 2,4-dimethylpentane
SAMPLE EXERCISE 24.2 Writing Condensed Structural Formulas
Write the condensed structural formula for 3-ethyl-2-methylpentane.
SOLUTION
Analyze We are given the systematic name for a hydrocarbon and asked to write its condensed structural formula.
Plan Because the name ends in -ane, the compound is an alkane, meaning that all the carbon–carbon bonds are single bonds. The parent hydrocarbon is pentane, indicating five C atoms (Table 24.2). There are two alkyl groups specified, an ethyl group (two carbon atoms, C2H5) and a methyl group (one carbon atom, CH3). Counting from left to right along the five-carbon chain, the name tells us that the ethyl group is attached to C3 and the methyl group is attached to C2.
Solve We begin by writing five C atoms attached by single bonds. These represent the backbone of the parent pentane chain:
C—C—C—C—C
We next place a methyl group on the second C and an ethyl group on the third C of the chain. We then add hydrogens to all the other C atoms to make four bonds to each carbon:
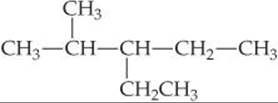
The formula can be written more concisely as
CH3CH(CH3)CH(C2H5)CH2CH3
where the branching alkyl groups are indicated in parentheses.
PRACTICE EXERCISE
Write the condensed structural formula for 2,3-dimethylhexane.
Answer: 
Cycloalkanes
Alkanes that form rings, or cycles, are called cycloalkanes. As ![]() FIGURE 24.5 illustrates, cycloalkane structures are sometimes drawn as line structures, which are polygons in which each corner represents a CH2 group. This method of representation is similar to that used for benzene rings.
FIGURE 24.5 illustrates, cycloalkane structures are sometimes drawn as line structures, which are polygons in which each corner represents a CH2 group. This method of representation is similar to that used for benzene rings.![]() (Section 8.6) (Remember from our benzene discussion that in aromatic structures each vertex represents a CH group, not a CH2 group.)
(Section 8.6) (Remember from our benzene discussion that in aromatic structures each vertex represents a CH group, not a CH2 group.)
Carbon rings containing fewer than five carbon atoms are strained because the C—C—C bond angles must be less than the 109.5° tetrahedral angle. The amount of strain increases as the rings get smaller. In cyclopropane, which has the shape of an equilateral triangle, the angle is only 60°; this molecule is therefore much more reactive than propane, its straight-chain analog.
Reactions of Alkanes
Because they contain only C—C and C—H bonds, most alkanes are relatively unreactive. At room temperature, for example, they do not react with acids, bases, or strong oxidizing agents. Their low chemical reactivity, as noted in Section 24.1, is due primarily to the strength and lack of polarity of C—C and C—H bonds.
Alkanes are not completely inert, however. One of their most commercially important reactions is combustion in air, which is the basis of their use as fuels. ![]() (Section 3.2) For example, the complete combustion of ethane proceeds as follows:
(Section 3.2) For example, the complete combustion of ethane proceeds as follows:
2 C2H6(g) + 7 O2(g) → 4 CO2(g) + 6 H2O(l) ΔH° = –2855 kJ
![]() GO FIGURE
GO FIGURE
The general formula for straight-chain alkanes is CnH2n+2. What is the general formula for cycloalkanes?
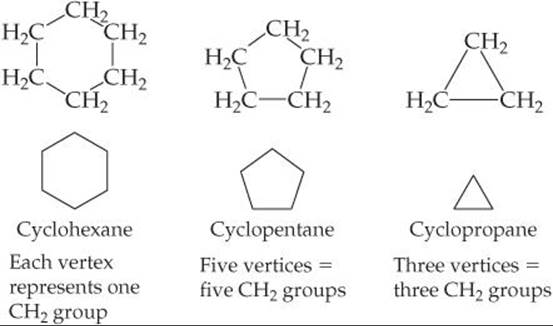
![]() FIGURE 24.5 Condensed structural formulas and line structures for three cycloalkanes.
FIGURE 24.5 Condensed structural formulas and line structures for three cycloalkanes.
![]() CHEMISTRY PUT TO WORK
CHEMISTRY PUT TO WORK
Gasoline
Petroleum, or crude oil, is a mixture of hydrocarbons plus smaller quantities of other organic compounds containing nitrogen, oxygen, or sulfur. The tremendous demand for petroleum to meet the world's energy needs has led to the tapping of oil wells in such forbidding places as the North Sea and northern Alaska.
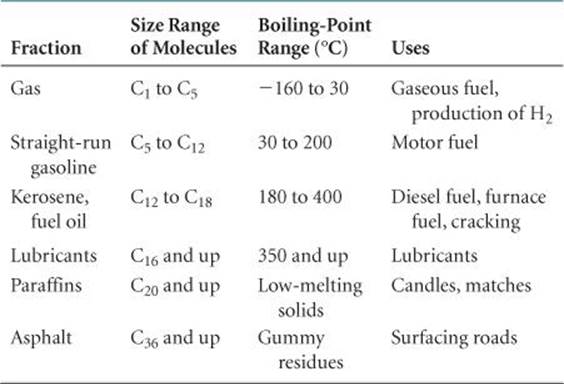
The usual first step in the refining, or processing, of petroleum is to separate it into fractions on the basis of boiling point (![]() TABLE 24.5). Because gasoline is the most commercially important of these fractions, various processes are used to maximize its yield.
TABLE 24.5). Because gasoline is the most commercially important of these fractions, various processes are used to maximize its yield.
Gasoline is a mixture of volatile alkanes and aromatic hydrocarbons. In a traditional automobile engine, a mixture of air and gasoline vapor is compressed by a piston and then ignited by a spark plug. The burning of the gasoline should create a strong, smooth expansion of gas, forcing the piston outward and imparting force along the driveshaft of the engine. If the gas burns too rapidly, the piston receives a single hard slam rather than a strong, smooth push. The result is a “knocking” or “pinging” sound and a reduction in the efficiency with which energy produced by the combustion is converted to work.
The octane number of a gasoline is a measure of its resistance to knocking. Gasolines with high octane numbers burn more smoothly and are thus more effective fuels (![]() FIGURE 24.6). Branched alkanes and aromatic hydrocarbons have higher octane numbers than straight-chain alkanes. The octane number of gasoline is obtained by comparing its knocking characteristics with those of isooctane (2,2,4-trimethylpentane) and heptane. Isooctane is assigned an octane number of 100, and heptane is assigned 0. Gasoline with the same knocking characteristics as a mixture of 91% isooctane and 9% heptane, for instance, is rated as 91 octane.
FIGURE 24.6). Branched alkanes and aromatic hydrocarbons have higher octane numbers than straight-chain alkanes. The octane number of gasoline is obtained by comparing its knocking characteristics with those of isooctane (2,2,4-trimethylpentane) and heptane. Isooctane is assigned an octane number of 100, and heptane is assigned 0. Gasoline with the same knocking characteristics as a mixture of 91% isooctane and 9% heptane, for instance, is rated as 91 octane.
TABLE 24.5 • Hydrocarbon Fractions from Petroleum

![]() FIGURE 24.6 Octane rating. The octane rating of gasoline measures its resistance to knocking when burned in an engine. The octane rating of the gasoline in the foreground is 89.
FIGURE 24.6 Octane rating. The octane rating of gasoline measures its resistance to knocking when burned in an engine. The octane rating of the gasoline in the foreground is 89.
The gasoline obtained by fractionating petroleum (called straight-run gasoline) contains mainly straight-chain hydrocarbons and has an octane number around 50. To increase its octane rating, it is subjected to a process called reforming, which converts the straight-chain alkanes into branched-chain ones.
Cracking is used to produce aromatic hydrocarbons and to convert some of the less-volatile fractions of petroleum into compounds suitable for use as automobile fuel. In cracking, the hydrocarbons are mixed with a catalyst and heated to 400 °C to 500 °C. The catalysts used are either clay minerals or synthetic Al2O3–SiO2 mixtures. In addition to forming molecules more suitable for gasoline, cracking results in the formation of such low-molecular-weight hydrocarbons as ethylene and propene. These substances are used in a variety of reactions to form plastics and other chemicals.
Adding compounds called either antiknock agents or octane enhancers increases the octane rating of gasoline. Until the mid-1970s the principal antiknock agent was tetraethyl lead, (C2H5)4Pb. It is no longer used, however, because of the environmental hazards of lead and because it poisons catalytic converters. ![]() (Section 14.7 “Chemistry Put to Work: Catalytic Converters”) Aromatic compounds such as toluene (C6H5CH3) and oxygenated hydrocarbons such as ethanol (CH3CH2OH) are now generally used as antiknock agents.
(Section 14.7 “Chemistry Put to Work: Catalytic Converters”) Aromatic compounds such as toluene (C6H5CH3) and oxygenated hydrocarbons such as ethanol (CH3CH2OH) are now generally used as antiknock agents.
RELATED EXERCISES: 24.19 and 24.20