CHEMISTRY THE CENTRAL SCIENCE
24 THE CHEMISTRY OF LIFE: ORGANIC AND BIOLOGICAL CHEMISTRY
24.5 CHIRALITY IN ORGANIC CHEMISTRY
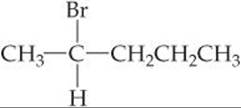
A molecule possessing a nonsuperimposable mirror image is termed chiral (Greek cheir, “hand”). ![]() (Section 23.4) Compounds containing carbon atoms with four different attached groups are inherently chiral. A carbon atom with four different attached groups is called a chiral center. For example, consider 2-bromopentane:
(Section 23.4) Compounds containing carbon atoms with four different attached groups are inherently chiral. A carbon atom with four different attached groups is called a chiral center. For example, consider 2-bromopentane:
![]() GO FIGURE
GO FIGURE
If you replace Br with CH3, will the compound be chiral?
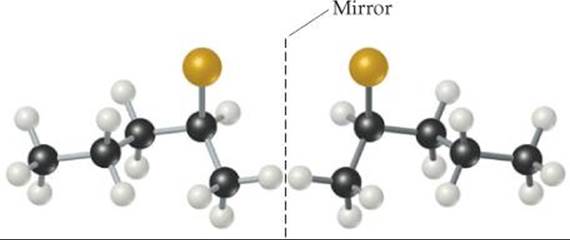
![]() FIGURE 24.15 The two enantiomeric forms of 2-bromopentane. The mirror-image isomers are not superimposable on each other.
FIGURE 24.15 The two enantiomeric forms of 2-bromopentane. The mirror-image isomers are not superimposable on each other.
All four groups attached to C2 are different, making that carbon a chiral center. ![]() FIGURE 24.15 illustrates the nonsuperimposable mirror images of this molecule. Imagine moving the molecule shown to the left of the mirror over to the right of the mirror. If you then turn it in every possible way, you will conclude that it cannot be superimposed on the molecule shown to the right of the mirror. Nonsuperimposable mirror images are called either optical isomers or enantiomers.
FIGURE 24.15 illustrates the nonsuperimposable mirror images of this molecule. Imagine moving the molecule shown to the left of the mirror over to the right of the mirror. If you then turn it in every possible way, you will conclude that it cannot be superimposed on the molecule shown to the right of the mirror. Nonsuperimposable mirror images are called either optical isomers or enantiomers. ![]() (Section 23.4) Organic chemists use the labels R and S to distinguish the two forms. We need not go into the rules for deciding on the labels.
(Section 23.4) Organic chemists use the labels R and S to distinguish the two forms. We need not go into the rules for deciding on the labels.
The two members of an enantiomer pair have identical physical properties and identical chemical properties when they react with nonchiral reagents. Only in a chiral environment do they behave differently from each other. One interesting property of chiral substances is that their solutions may rotate the plane of polarized light, as explained in Section 23.4.
Chirality is common in organic substances. It is not often observed, however, because when a chiral substance is synthesized in a typical reaction, the two enantiomers are formed in precisely the same quantity. The resulting mixture is called a racemic mixture, and it does not rotate the plane of polarized light because the two forms rotate the light to equal extents in opposite directions. ![]() (Section 23.4)
(Section 23.4)
Many drugs are chiral substances. When a drug is administered as a racemic mixture, often only one enantiomer has beneficial results. The other is either inert, or nearly so, or may even have a harmful effect. For example, the drug (R)-albuterol (![]() FIGURE 24.16) is a bronchodilator used to relieve the symptoms of asthma. The enantiomer (S)-albuterol is not only ineffective as a bronchodilator but also actually counters the effects of (R)-albuterol. As another example, the nonsteroidal analgesic ibuprofen is a chiral molecule usually sold as the racemic mixture. However, a preparation consisting of just the more active enantiomer, (S)-ibuprofen (
FIGURE 24.16) is a bronchodilator used to relieve the symptoms of asthma. The enantiomer (S)-albuterol is not only ineffective as a bronchodilator but also actually counters the effects of (R)-albuterol. As another example, the nonsteroidal analgesic ibuprofen is a chiral molecule usually sold as the racemic mixture. However, a preparation consisting of just the more active enantiomer, (S)-ibuprofen (![]() FIGURE 24.17), relieves pain and reduces inflammation more rapidly than the racemic mixture. For this reason, the chiral version of the drug may in time come to replace the racemic one.
FIGURE 24.17), relieves pain and reduces inflammation more rapidly than the racemic mixture. For this reason, the chiral version of the drug may in time come to replace the racemic one.
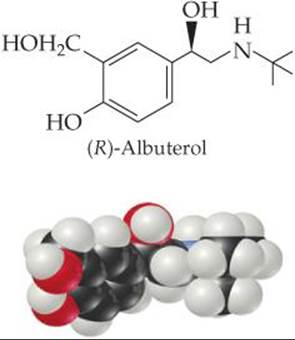
![]() FIGURE 24.16 (R)-Albuterol. This compound, which acts as a bronchodilator in patients with asthma, is one member of an enantiomer pair. The other member, (S)-albuterol, does not have the same physiological effect.
FIGURE 24.16 (R)-Albuterol. This compound, which acts as a bronchodilator in patients with asthma, is one member of an enantiomer pair. The other member, (S)-albuterol, does not have the same physiological effect.
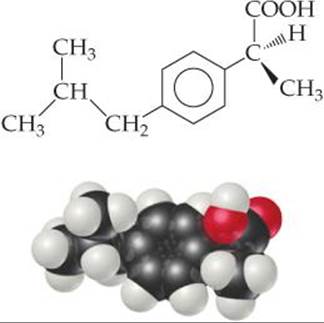
![]() FIGURE 24.17 (S)-Ibuprofen. For relieving pain and reducing inflammation, the ability of this enantiomer far outweighs that of the (R) isomer.
FIGURE 24.17 (S)-Ibuprofen. For relieving pain and reducing inflammation, the ability of this enantiomer far outweighs that of the (R) isomer.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What are the requirements on the four groups attached to a carbon atom in order that it be a chiral center?