CHEMISTRY THE CENTRAL SCIENCE
24 THE CHEMISTRY OF LIFE: ORGANIC AND BIOLOGICAL CHEMISTRY
24.9 LIPIDS
Lipids are a diverse class of nonpolar biological molecules used by organisms for long-term energy storage (fats, oils) and as elements of biological structures (phospholipids, cell membranes, waxes).
Fats
Fats are lipids derived from glyercol and fatty acids. Glycerol is an alcohol with three OH groups. Fatty acids are carboxylic acids (RCOOH) in which R is a hydrocarbon chain, usually 16 to 19 carbon atoms in length. Glycerol and fatty acids undergo condensation reactions to form ester linkages as shown in ![]() FIGURE 24.25. Three fatty acid molecules join to a glycerol. Although the three fatty acids in a fat can be the same, as they are in Figure 24.25, it is also possible that a fat contains three different fatty acids.
FIGURE 24.25. Three fatty acid molecules join to a glycerol. Although the three fatty acids in a fat can be the same, as they are in Figure 24.25, it is also possible that a fat contains three different fatty acids.
Lipids with saturated fatty acids are called saturated fats and are commonly solids at room temperature (such as butter and shortening). Unsaturated fats contain one or more double bonds in their carbon-carbon chains. The cis and trans nomenclature we learned for alkenes applies: Trans fats have H atoms on the opposite sides of the C═C double bond, and cis fats have H atoms on the same sides of the C═C double bond. Unsaturated fats (such as olive oil and peanut oil) are usually liquid at room temperature and are more often found in plants. For example, the major component (approximately 60 to 80%) of olive oil is oleic acid, cis-CH3(CH2)7CH═CH(CH2)7COOH.
Oleic acid is an example of a monounsaturated fatty acid, meaning it has only one carbon-carbon double bond in the chain. In contrast, polyunsaturated fatty acids have more than one carbon-carbon double bond in the chain.
For humans, trans fats are not nutritionally required, which is why some governments are moving to ban them in foods. How, then, do trans fats end up in our food? The process that converts unsaturated fats (such as oils) into saturated fats (such as shortening) is hydrogenation. ![]() (Section 24.3) The by-products of this hydrogenation process include trans fats.
(Section 24.3) The by-products of this hydrogenation process include trans fats.
![]() GO FIGURE
GO FIGURE
What structural features of a fat molecule cause it to be insoluble in water?
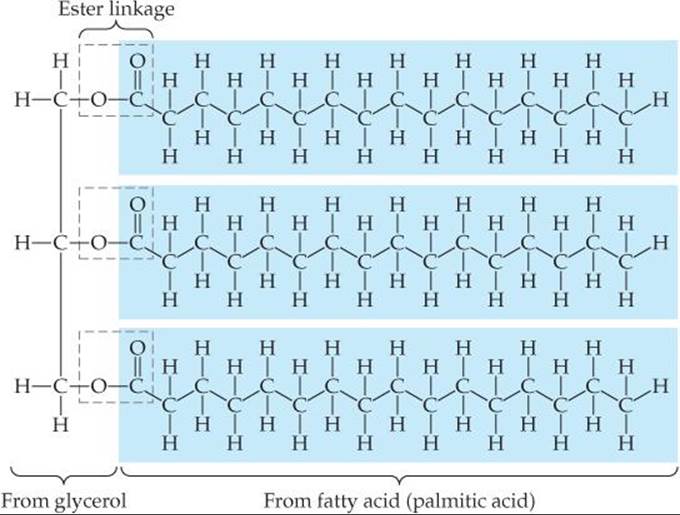
![]() FIGURE 24.25 Structure of a fat.
FIGURE 24.25 Structure of a fat.
Some of the fatty acids essential for human health must be available in our diets because our metabolism cannot synthesize them. These essential fatty acids are ones that have the carbon–carbon double bonds either three carbons or six carbons away from the—CH3 end of the chain. These are called omega-3 and omega-6 fatty acids, where omega refers to the last carbon in the chain (the carboxylic acid carbon is considered the first, or alpha, one).
Phospholipids
Phospholipids are similar in chemical structure to fats but have only two fatty acids attached to a glycerol. The third alcohol group of glycerol is joined to a phosphate group (![]() FIGURE 24.26). The phosphate group can be also attached to a small charged or polar group, such as choline, as shown in the figure. The diversity in phospholipids is based on differences in their fatty acids and in the groups attached to the phosphate group.
FIGURE 24.26). The phosphate group can be also attached to a small charged or polar group, such as choline, as shown in the figure. The diversity in phospholipids is based on differences in their fatty acids and in the groups attached to the phosphate group.
In water, phospholipids cluster together with their charged polar heads facing the water and their nonpolar tails facing inward. The phospholipids thus form a bilayer that is a key component of cell membranes (![]() FIGURE 24.27).
FIGURE 24.27).