CHEMISTRY THE CENTRAL SCIENCE
ANSWERS TO GO FIGURE
CHAPTER 1
Figure 1.1 9
Figure 1.4 Vapor (gas)
Figure 1.5 Molecules of a compound are composed of more than one type of atom, and molecules of an element are composed of only one type of atom.
Figure 1.6 Earth is rich in silicon and aluminum; the human body is rich in carbon and hydrogen
Figure 1.7 They are the same; there are twice as many hydrogen molecules as oxygen molecules, and the hydrogen gas takes up twice the volume of the oxygen gas.
Figure 1.17 True
Figure 1.18 1000
Figure 1.23 The darts would be scattered widely (poor precision) but their average position would be at the center (good accuracy).
CHAPTER 2
Figure 2.3 We know the rays travel from the cathode because of the way the magnetic field diverts the path (b).
Figure 2.4 The electron beam would be deflected downward because of repulsion by the negative plate and attraction toward the positive plate.
Figure 2.8 The beta rays, whose path is diverted away from the negative plate and toward the positive plate, consist of electrons. Because the electrons are much less massive than the alpha particles, their motion is affected more strongly by the electric field.
Figure 2.10 The beam consists of alpha particles, which carry a +2 charge.
Figure 2.14 Based on the periodic trend, we expect that elements that precede a nonreactive gas, as F does, will also be reactive nonmetals. The elements fitting this pattern are H and Cl.
Figure 2.19 The ball-and-stick model more clearly shows the connections between atoms, so we can see the angles at which the atoms are attached in the molecule.
Figure 2.20 The elements are in the following groups: Ag+ is 1B, Zn2+ is 2B, and Sc3+ is 3B. Sc3+ has the same number of electrons as Ar (element 18).
Figure 2.24 Removing one O atom from the perbromate ion gives the bromate ion, BrO3–.
CHAPTER 3
Figure 3.3 The formula CO2 represents one molecule containing one C and two O atoms, whereas 2 CO represents two molecules, each containing one C atom and one O atom for a total of two C and two O atoms.
Figure 3.8 Both figures show combustion reactions in which the fuel is a hydrocarbon (CH4 in Figure 3.4 and C3H8 in Figure 3.8). In both cases the reactants are the hydrocarbon and O2, and the products are CO2 and H2O.
Figure 3.9 As shown, 18.0 g H2O = 1 mol H2O = 6.02 × 1023 molecules H2O. Thus, 9.00 g H2O = 0.500 mol H2O = 3.01 × 1023 molecules H2O.
Figure 3.12 (a) The molar mass of CH4, 16.0 g CH4/1 mol CH4. (b) Avogadro's number, 1 mol CH4/6.02 × 1023 formula units CH4, where a formula unit in this case is a molecule.
Figure 3.13 The mole ratio is obtained by dividing the molecular weight by the empirical formula weight, Equation 3.11.
Figure 3.17 There are 7 mol O2, and each mol O2 yields 2 mol H2O. Thus, 14 mol H2O would have formed.
CHAPTER 4
Figure 4.3 NaCl(aq)
Figure 4.4 K+ and NO3–
Figure 4.9 Two moles of hydrochloric acid are needed to react with each mole of Mg(OH)2.
Figure 4.19 The volume needed to reach the end point if Ba(OH)2(aq) were used would be one-half the volume needed for titration with NaOH(aq).
CHAPTER 5
Figure 5.1 In the act of throwing, the pitcher transfers energy to the ball, which then becomes kinetic energy of the ball. For a given amount of energy E transferred to the ball, Equation 5.1 tells us that the speed of the ball is ![]() where m is the mass of the ball. Because a baseball has less mass than a bowling bowl, it will have a higher speed for a given amount of energy transferred.
where m is the mass of the ball. Because a baseball has less mass than a bowling bowl, it will have a higher speed for a given amount of energy transferred.
Figure 5.2 When she starts going uphill, kinetic energy is converted to potential energy and her speed decreases.
Figure 5.3 The electrostatic potential energy of two oppositely charged particles is negative (Equation 5.2). As the particles become closer, the electrostatic potential energy becomes even more negative—that is, it decreases.
Figure 5.4 Yes, the system is still closed—matter can't escape the system to the surroundings unless the piston is pulled completely out of the cylinder.
Figure 5.5 If Efinal = Einitial, thenΔ E = 0
Figure 5.6
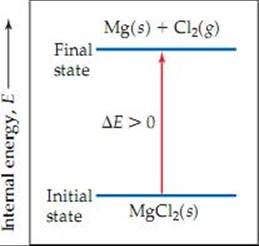
Figure 5.7 No. The sign on w is positive and the sign on q is negative. We need to know the magnitudes of q and w to determine whether Δ E = q + w is positive or negative.
Figure 5.10 The battery is doing work on the surroundings, so w < 0.
Figure 5.11 We need to know whether Zn(s) or HCl(aq) is the limiting reagent of the reaction. If it is Zn(s), then the addition of more Zn will lead to the generation of more H2(g) and more work will be done.
Figure 5.17 Endothermic—heat is being added to the system to raise the temperature of the water.
Figure 5.18 Two cups provide more thermal insulation so less heat will escape the system.
Figure 5.19 The stirrer ensures that all of the water in the bomb is at the same temperature.
Figure 5.21 The condensation of 2 H2O(g) to 2 H2O(l)
Figure 5.22 Yes, ΔH3 would remain the same as it is the enthalpy change for the process CO(g) + ![]() O2(g)
O2(g) ![]() CO2(g).
CO2(g).
Figure 5.24 Grams of fat
CHAPTER 6
Figure 6.3 The wavelength of (a) is twice that of (b) and the frequency of (a) is consequently half that of (b). Thus, the wavelength of (b) is 0.50 m and its frequency is 6.0 × 108 cycles/s.
Figure 6.4 The X-ray has a shorter wavelength and, consequently, higher frequency than the red light.
Figure 6.5 The hottest area is the white or yellowish white area in the center.
Figure 6.7 If the tube is not evacuated, the electrons that are freed from the metal surface will strike gas molecules near that surface. As a result, they will become attached to the gas molecules and never arrive at the positive terminal.
Figure 6.12 The n = 2 to n = 1 transition involves a larger energy change than the n = 3 to n = 2 transition. (Compare the space differences between the states in the figure.) If the n = 2 to n = 1 transition produces visible light, the n = 3 to n = 2 transition must produce radiation of lower energy. The infrared radiation has lower frequency and, hence, lower energy than visible light, whereas the ultraviolet has greater frequency and greater energy. Thus, the n = 2 to n = 1 transition will produce ultraviolet radiation.
Figure 6.16 The region of highest electron density is where the density of dots is highest, which is near the nucleus.
Figure 6.17 The fourth shell (n = 4) would contain four subshells, labeled 4s, 4p, 4d, and 4f.
Figure 6.18 There would be four maxima and three nodes.
Figure 6.22 (a) The intensity of the color indicates that the probability of finding the electron is greater at the interior of the lobes than on the edges. (b) 2px.
Figure 6.24 The 4d and 4f subshells are not shown.
CHAPTER 7
Figure 7.1 Row 7—these elements are generally radioactive and not stable.
Figure 7.3 2s
Figure 7.6 Bottom and left
Figure 7.7 They get larger, just like the atoms do.
Figure 7.9 Ar; it has a larger Zeff.
Figure 7.10 There is more electron–electron repulsion in the case of oxygen because two electrons have to occupy the same orbital.
Figure 7.11 The halogens (group 7A); it does make sense because we know that they are very stable as anions.
Figure 7.12 Ionization energy—lower ionization energy is correlated with increasing metallic character.
Figure 7.14 Anions are above the lines; cations are below the line.
Figure 7.22 Lilac
CHAPTER 8
Figure 8.1 Covalent
Figure 8.2 Yes, the same sort of reaction should occur between any of the alkali metals and any of the elemental halogens.
Figure 8.3 Cations have a smaller radius than their neutral atoms and anions have a larger radius. Because Na and Cl are in the same row of the periodic table, we would expect Na+ to have a smaller radius than Cl–, so we would guess that the larger green spheres represent Cl–.
Figure 8.4 The distance between ions in KF should be larger than that in NaF and smaller than that in KCl. We would thus expect the lattice energy of KF to be between 701 and 910 kJ/mol.
Figure 8.6 The repulsions between the nuclei would decrease, the attractions between the nuclei and the electrons would decrease, and the repulsions between the electrons would be unaffected.
Figure 8.7 The electronegativity decreases with increasing atomic number.
Figure 8.9 μ will decrease
Figure 8.10 The bonds are not polar enough to cause enough excess electron density on the halogen atom to lead to a red shading.
Figure 8.12 The lengths of the bonds of the outer O atoms to the inner O atom are the same.
Figure 8.13 Yes. The electron densities on the left and right parts of the molecule are the same, indicating that resonance has made the two O — O bonds equivalent to one another.
Figure 8.14 The dashed bonds represent the “half bonds” that result when the two resonance structures are averaged.
Figure 8.15 Exothermic
Figure 8.17 As the bond gets longer, it gets weaker. We would therefore expect a plot of bond enthalpy versus bond length to have a negative slope.
CHAPTER 9
Figure 9.1 The atomic radii (Figure 7.7)
Figure 9.3 Octahedral
Figure 9.7 The electron pair in the bonding domain is attracted toward two nuclear centers, whereas the nonbonding pair is attracted toward just one.
Figure 9.8 90°
Figure 9.9 The nonbonding electron pairs exert a greater repulsive force than the bonding electron pairs.
Figure 9.10 The heads of the arrows point toward regions of highest electron density, as indicated by the red color.
Figure 9.14 As the internuclear distance decreases, nucleus–nucleus repulsion becomes a dominant component of the potential energy.
Figure 9.16 The small lobes of the sp hybrid orbitals are very much smaller in spatial extent and, therefore, provide very little overlap with the F orbitals.
Figure 9.17 Three: one s and two p orbitals
Figure 9.23 The two p orbitals that form the π bond must align, and each of them is perpendicular to the plane of the sp2 hybrid orbitals.
Figure 9.24 Acetylene, because it has two C — C π bonds, whereas ethylene has one π bond
Figure 9.26 C — H and C — C
Figure 9.33 The ![]()
Figure 9.34 The two electrons in the σ1s MO
Figure 9.35 The 1 s orbitals of Li are small in spatial extent because they experience a strong nuclear attraction. In addition, both the bonding and antibonding MOs formed from them are occupied, so that there is no significant net bonding.
Figure 9.36 Nodal planes between the atoms are found in antibonding MOs.
Figure 9.42 The σ2p and π2p orbitals. Because the σ2p orbital mixes with the σ2s, it is pushed to higher energy and the σ2s is moved to lower energy. The σ2p orbital thus rises above the π2p in energy.
Figure 9.43 F2 contains four more electrons than N2. These electrons go into the antibonding π*2p orbitals, thus lowering the bond order.
Figure 9.45 Because N2 has no unpaired electrons, it is diamagnetic. Therefore, it would simply flow down with no tendency to remain in the magnetic field.
Figure 9.46 11. All the electrons in the n = 2 level are valence-shell electrons.
CHAPTER 10
Figure 10.2 It will increase.
Figure 10.5 Decrease
Figure 10.6 1520 torr
Figure 10.7 Linear
Figure 10.10 one
Figure 10.11 It is small and inert.
Figure 10.17 About a third
Figure 10.18 Higher speeds are correlated with smaller molar masses (assuming constant T).
Figure 10.20 n, moles of gas
Figure 10.22 Not really—CO2 is least ideal and does have the largest molar mass, but H2, the lightest gas, deviates more from the ideal line than the heavier N2.
Figure 10.23 True
Figure 10.25 It would increase.
CHAPTER 11
Figure 11.2 The density in a liquid is much closer to a solid than it is to a gas.
Figure 11.9 Both compounds are nonpolar and incapable of forming hydrogen bonds. Therefore, the boiling point is determined by the dispersion forces, which are stronger for the larger, heavier SnH4.
Figure 11.10 The non-hydrogen atom must possess a nonbonding electron pair.
Figure 11.11 There are four electron pairs surrounding oxygen in a water molecule. Two of the electron pairs are used to make covalent bonds to hydrogen within the H2O molecule, while the other two are available to make hydrogen bonds to neighboring molecules. Because the electron-pair geometry is tetrahedral (four electron domains around the central atom), the H — O … H bond angle is approximately 109°.
Figure 11.19 Wax is a hydrocarbon that cannot form hydrogen bonds.
Therefore, coating the inside of tube with wax will dramatically decrease the adhesive forces between water and the tube and change the shape of the water meniscus to an inverted U-shape. Neither wax nor glass can form metallic bonds with mercury so the shape of the mercury meniscus will be qualitatively the same, an inverted U-shape.
Figure 11.21 Because we are dealing with a state function, the energy of going straight from a solid to a gas must be the same as going from a solid to a gas through an intermediate liquid state. Therefore, the heat of sublimation must be equal to the sum of the heat of fusion and the heat of vaporization: ΔHsub = ΔHfus + ΔHvap.
Figure 11.24 Increases, because the molecules have more kinetic energy as the temperature increases and can escape more easily
Figure 11.25 All liquids including ethylene glycol reach their normal boiling point when their vapor pressure is equal to atmospheric pressure, 760 torr.
Figure 11.27 Freezing, because for most substances the solid phase is denser than the liquid phase and increasing the pressure will eventually drive a phase transition from the liquid to the solid state (provided the temperature is below the critical temperature)
CHAPTER 12
Figure 12.13 A hexagonal lattice
Figure 12.15 The solvent is the majority component and the solute the minority component. Therefore, there will be more solvent atoms than solute atoms.
Figure 12.17 The samarium atoms sit on the corners of the unit cell so there is only 8 × (1/8) = 1 Sm atom per unit cell. Eight of the nine cobalt atoms sit on faces of the unit cell, and the other sits in the middle of the unit cell so there are 8 × (1/2) + 1 = 5 Co atoms per unit cell.
Figure 12.19 The atoms are randomly arranged in red gold, which is a substitutional alloy. Purple gold is an intermetallic compound in which the atoms are arranged in a specific ordered pattern.
Figure 12.20 By drawing Lewis structures you can show that there are three (chlorine), two (sulfur), one (phosphorus), and zero (silicon) nonbonding electron pairs per atom.
Figure 12.22 In the fourth period, vanadium and chromium have very similar melting points. Molybdenum and tungsten have the highest melting points in the fifth and sixth periods, respectively. All of these elements are located near the middle of the period where the bonding orbitals are mostly filled and the antibonding orbitals mostly empty.
Figure 12.23 The molecular orbitals become more closely spaced in energy.
Figure 12.24 Potassium has only one valence electron per atom. If we fill the 4s band halfway probably a small amount of electron density will leak over and start to fill the 3d orbitals as well. The 4p orbitals should be empty.
Figure 12.25 Ionic substances cleave because the nearest neighbor interactions switch from attractive to repulsive if the atoms slide so that ions of like charge (cation–cation and anion–anion) touch each other. Metals don't cleave because the atoms are attracted to all other atoms in the crystal through metallic bonding.
Figure 12.26 No, ions of like charge do not touch in an ionic compound because they are repelled from one another. In an ionic compound the cations touch the anions.
Figure 12.28 In NaF there are four Na+ ions (12 × 1/4) and four F– ions (8 × 1/8 + 6 × 1/2) per unit cell. In MgF2 there are two Mg2+ ions (8 × 1/8 + 1) and four F– ions (4 × 1/2 + 2) per unit cell. In ScF3 there is one Sc3+ ion (8 × 1/8) and three F– ions (12 × 1/4) per unit cell.
Figure 12.29 The intermolecular forces are stronger in toluene, as shown by its higher boiling point. The molecules pack more efficiently in benzene, which explains its higher melting point, even though the intermolecular forces are weaker.
Figure 12.44 Decrease. As the quantum dots get smaller, the band gap increases and the emitted light shifts to shorter wavelength.
Figure 12.47 Each carbon atom in C60 is bonded to three neighboring carbon atoms through covalent bonds. Thus, the bonding is more like graphite, where carbon atoms also bond to three neighbors, than diamond, where carbon atoms bond to four neighbors.
CHAPTER 13
Figure 13.2 Opposite charges attract. The electron-rich O atom of the H2O molecule, which is the negative end of the dipole, is attracted to the positive Na+ ion.
Figure 13.3 The negative end of the water dipole (the O) is attracted to the positive Na+ ion, whereas the positive end of the dipole (the H) is attracted to the negative Cl– ion.
Figure 13.4 For exothermic solution processes the magnitude of ΔHmix will be larger than the magnitude of ΔHsolute + ΔHsolvent
Figure 13.8 The dissolving of the crystal and the crystallization by which ions in solution become reattached to the solid
Figure 13.9 If the solution wasn't supersaturated, solute would not crystallize from it.
Figure 13.14 If the partial pressure of a gas over a solution is doubled, the concentration of gas in the solution would double.
Figure 13.15 The slopes increase as the molecular weight increases. The larger the molecular weight, the greater the polarizability of the gas molecules, leading to greater intermolecular attractive forces between gas molecules and water molecules.
Figure 13.18 Looking at where the solubility curves for KCl and NaCl intersect the 80 °C line, we see that the solubility of KCl is about 51 g/100 g H2O, whereas NaCl has a solubility of about 39 g/100 g H2O. Thus, KCl is more soluble than NaCl at this temperature.
Figure 13.19 N2 has the same molecular weight as CO but is nonpolar, so we can predict that its curve will be just below that of CO.
Figure 13.25 The water will move through the semipermeable membrane toward the more concentrated solution. Thus, the liquid level in the left arm will increase.
Figure 13.26 Water will move toward the more concentrated solute solution, which is inside the red blood cells, causing them to undergo hemolysis.
Figure 13.30 The two negatively charged groups both have the composition — CO2–.
Figure 13.32 Recall the rule that likes dissolve likes. The oil drop is composed of nonpolar molecules, which interact with the nonpolar part of the stearate ion with dispersion forces.
CHAPTER 14
Figure 14.3 B
Figure 14.4 It decreases.
Figure 14.8 The reaction is first order in CH3NC.
Figure 14.10 At early times in the reaction; both graphs look linear close to t = 0.
Figure 14.17 The energy needed to overcome the energy barrier (the activation energy) looks about twice as large as the overall energy change for the reaction.
Figure 14.23 For the blue curve: The transition states are at the top of the peaks (2) and the intermediate is in the “valley” between the two peaks. For the red curve: The top of the peak is the transition state; no intermediates are shown.
Figure 14.27 Substrate; if products bound tightly, they would not leave and the active site would not be free.
CHAPTER 15
Figure 15.1 The color in the tube stops changing.
Figure 15.2 No
Figure 15.6 The boxes would be approximately the same size.
Figure 15.7 It will be lower; some CO2 has to react with CaO to make some CaCO3.
Figure 15.9 500 atm and 400 °C
Figure 15.10 Nitrogen (and some of the added hydrogen) is converted into ammonia.
Figure 15.14 About two to three times faster, based on the graph
Figure 15.15 About 5 × 10–4
CHAPTER 16
Figure 16.2 Hydrogen bonds
Figure 16.3 O2– (aq) + H2O(l) ![]() 2 OH–(aq)
2 OH–(aq)
Figure 16.7 Phenolphthalein changes from colorless, for pH values less than 8, to pink for pH values greater than 10. A pink color indicates pH > 10.
Figure 16.8 Bromothymol blue would be most suitable because it changes pH over a range that brackets pH = 7. Methyl red is not sensitive to pH changes when pH > 6, while phenolphthalein is not sensitive to pH changes when pH < 8, so neither changes color at pH = 7.
Figure 16.11 Yes. The equilibrium of interest is H3CCOOH ![]() H+ + H3CCOO–. If the percent dissociation remained constant as the acid concentration increased, the concentration of all three species would increase at the same rate. However, because there are two products and only one reactant, the total concentration of products would increase faster than the concentration of reactants. To offset this effect the percent dissociation decreases as the acid concentration increases.
H+ + H3CCOO–. If the percent dissociation remained constant as the acid concentration increased, the concentration of all three species would increase at the same rate. However, because there are two products and only one reactant, the total concentration of products would increase faster than the concentration of reactants. To offset this effect the percent dissociation decreases as the acid concentration increases.
Figure 16.12 The acidic hydrogens belong to carboxlyate (— COOH) groups, whereas the fourth proton bound to oxygen is part of a hydroxyl (— OH) group. In organic acids, like citric acid, the acidic protons are almost always part of a carboxylate group.
Figure 16.13 The nitrogen atom in hydroxylamine accepts a proton to form NH3OH+. As a general rule, nonbonding electron pairs on nitrogen atoms are more basic than nonbonding electron pairs on oxygen atoms.
CHAPTER 17
Figure 17.6 The pH will increase on addition of the base.
Figure 17.7 25.00 mL. The number of moles of added base needed to reach the equivalence point remains the same. Therefore, by doubling the concentration of added base the volume needed to reach the equivalence point is halved.
Figure 17.9 The volume of base needed to reach the equivalence point would not change because this quantity does not depend on the strength of the acid. However, the pH at the equivalence point, which is greater than 7 for a weak acid–strong base titration, would decrease to 7 because hydrochloric acid is a strong acid.
Figure 17.11 The pH at the equivalence point increases (becomes more basic) as the acid becomes weaker. The volume of added base needed to reach the equivalence point remains unchanged.
Figure 17.13 Yes. Any indicator that changes color between pH = 3 and pH = 11 could be used for a strong acid–strong base titration. Methyl red changes color between pH values of approximately 4 and 6.
Figure 17.22 ZnS and CuS would both precipitate on addition of H2S, preventing separation of the two ions.
Figure 17.23 Yes. CuS would precipitate in step 2 on addition of H2S to an acidic solution, while the Zn2+ ions remained in solution.
CHAPTER 18
Figure 18.1 About 85 km
Figure 18.3 The atmosphere absorbs a significant fraction of solar radiation.
Figure 18.4 The peak value is about 5 × 1012 molecules per cm3. If we use Avogradro's number to convert molecules to moles, and the conversion factor of 1000 cm3 = 1000 mL = 1 L, we find that the concentration of ozone at the peak is 8 × 10–9 mole/L.
Figure 18.16 This is ambiguous; both temperature and salinity vary with density in similar ways; but temperature seems to parallel density better. Temperature decreases down to 1000 m, then remains relatively constant; density increases down to 1000 m, and then remains relatively constant.
Figure 18.17 The depth of the aquifer; the nature of the intervening layers (how porous or dense they are)
Figure 18.19 Water is the chemical species that is crossing the membrane, not the ions.
CHAPTER 19
Figure 19.1 Yes, the potential energy of the eggs decreases as they fall.
Figure 19.2 Because the final volume would be less than twice the volume of Flask A, the final pressure would be greater than 0.5 atm.
Figure 19.3 The freezing of liquid water to ice is exothermic.
Figure 19.4 To be truly reversible, the temperature change δT must be infinitesimally small.
Figure 19.8 There are two other independent rotational motions of the H2O molecule:

Figure 19.9 Ice, because it is the phase in which the molecules are held most rigidly
Figure 19.11 The decrease in the number of molecules due to the formation of new bonds.
Figure 19.13 During a phase change, the temperature remains constant but the entropy change can be large.
Figure 19.14 Based on the three molecules shown, the addition of each C increases S° by 40–45 J/mol-K. Based on this observation, we would predict that S°(C4H10) would 310–315 J/mol-K. Appendix C confirms that this is a good prediction: S°(C4H10) = 310.0 J/mol-K.
Figure 19.16 Spontaneous
Figure 19.17 If we plot progress of the reaction versus free energy, equilibrium is at a minimum point in free energy, as shown in the figure. In that sense, the reaction runs “downhill” until it reaches that minimum point.
CHAPTER 20
Figure 20.1 (a) The bubbling is caused by the hydrogen gas formed in the reaction. (b) The reaction is exothermic, and the heat causes the formation of steam.
Figure 20.2 The permanganate, MnO4–, is reduced, as the half-reactions in the text show.
Figure 20.3 The blue color is due to Cu2+(aq). As this ion is reduced, forming Cu(s), the blue color fades.
Figure 20.4 The Zn is oxidized and, therefore, serves as the anode of the cell.
Figure 20.5 The electrical balance is maintained in two ways: Anions migrate into the half-cell, and cations migrate out.
Figure 20.9 As the cell operates, H+ is reduced to H2 in the cathode half-cell. As H+ is depleted, the positive Na+ ions are drawn into the half-cell to maintain electrical balance in the solution.
Figure 20.10 The reduction reaction occurs at the cathode. The substance that is reduced most easily is the one with the larger standard reduction potential, ![]() .
.
Figure 20.12 Oxidation is the loss of electrons. An oxidizing agent causes another substance to lose electrons by gaining them itself. A strong oxidizing agent readily gains electrons, meaning that it is easily reduced.
Figure 20.14 The variable n is the number of moles of electrons transferred in the process.
Figure 20.15 The Ni2+(aq) and the cations in the salt bridge migrate toward the cathode. The NO3–(aq) and the anions in the salt bridge migrate toward the anode.
Figure 20.19 The cathode consists of PbO2(s). Because each oxygen has an oxidation state of –2, lead must have an oxidation state of +4 in this compound.
Figure 20.22 The oxidizing agent of O2(g) from the air
CHAPTER 21
Figure 21.2 From Figure 21.2 we see that the belt of stability for a nucleus containing 70 protons lies at approximately 102 neutrons.
Figure 21.4 Only three of the elements with an even number of protons have fewer than three isotopes: He, Be, and C. Note that these three elements are the lightest elements that have an even atomic number. Because they are so light, any change in the number of neutrons will change the neutron/proton ratio significantly. This helps to explain why they do not have more stable isotopes. None of the elements in Figure 21.4 that have an odd number of protons have more than two stable isotopes.
Figure 21.6 6.25 g. After one half-life, the amount of the radioactive material will have dropped to 25.0 g. After two half-lives, it will have dropped to 12.5 g. After three half-lives, it will have dropped to 6.25 g.
Figure 21.19 Because large quantities of water are needed to condense the secondary coolant once it passes through the turbine
Figure 21.21 The United States has the most reactors in operation. China has the most reactors under construction. France generates the largest percentage of its electricity from nuclear power.
Figure 21.24 Alpha rays are less dangerous when outside the body because they cannot penetrate the skin. However, once inside the body they can do great harm to any cells they come in contact with.
CHAPTER 22
Figure 22.5 Beaker on the right is warmer.
Figure 22.6 HF is the most stable, SbH3 the least stable.
Figure 22.8 More soluble in CCl4—the colors are deeper.
Figure 22.9 CF2
Figure 22.10 Redox reactions: The halides are being oxidized.
Figure 22.14 No
Figure 22.16 Based on this structure—yes, it would have a dipole moment. In fact, if you look it up, hydrogen peroxide's dipole moment is larger than water's!
Figure 22.20 They have been converted into water.
Figure 22.21 Formally they could both be +2. If we consider that the central sulfur is like SO42–, however, then the central sulfur would be +6, like SO42–, and then the terminal sulfur would be –2.
Figure 22.22 Nitrite
Figure 22.23 Longer
Figure 22.26 The NO double bond
Figure 22.28 In P4O6 the electron domains about the P atoms are trigonal pyramidal; in P4O10 the electron domains about the P atoms are tetrahedral.
Figure 22.33 The minimum temperature should be the melting point of silicon; the temperature of the heating coil should not be so high that the silicon rod starts to melt outside the zone of the heating coil.
CHAPTER 23
Figure 23.3 Zn (it is colorless)
Figure 23.4 The increase parallels the linear increase in valence electron count.
Figure 23.5 All the electron spins would align with the direction of the magnetic field.
Figure 23.9 109.5 degrees for the tetrahedral Zn complex; 90 degrees for the square-planar Pt complex
Figure 23.13 4 for both (assuming no other ligands come in to bind)
Figure 23.15 In the same place as O2
Figure 23.16 The peak with a maximum at 650 nm, the longest wavelength and lowest energy
Figure 23.21 The cis one
Figure 23.24 Larger, since ammonia can displace water
Figure 23.26 The peak would stay in the same position in terms of wavelength, but its absorbance would decrease.
Figure 23.28 dx2 – y2 and dz2
Figure 23.29 Convert the wavelength of light, 495 nm, into energy in joules using E = hc/λ.
Figure 23.30 It would be to the right of the “yellow” member of the series, but the energy gap between filled and empty d orbitals would be even larger than that of the “yellow” one.
Figure 23.34 That orbital has the lobes that point directly at the ligands.
CHAPTER 24
Figure 24.1 Tetrahedral
Figure 24.2 The OH group is polar whereas the CH3 group is nonpolar. Hence, adding CH3 will (a) reduce the substance's solubility in polar solvents and (b) increase its solubility in nonpolar solvents.
Figure 24.5 CnH2n, because there are no CH3 groups, each carbon has two hydrogens.
Figure 24.7 Just one
Figure 24.9 Intermediates are minima and transition states are maxima on energy profiles.
Figure 24.14 Both lactic acid and citric acid
Figure 24.15 No, because there are not four different groups around any carbon
Figure 24.18 Those labeled “basic amino acids,” which have basic side groups that are protonated at pH 7
Figure 24.25 The long hydrocarbon chains, which are nonpolar
Figure 24.27 The polar parts of the phospholipids seek to interact with water whereas the nonpolar parts seek to interact with other nonpolar substances and to avoid water.
Figure 24.29 Negative charge because of charge on phosphate groups
Figure 24.31 GC because each base has three hydrogen bonding sites, whereas there are only two in AT