CHEMISTRY THE CENTRAL SCIENCE
3 STOICHIOMETRY: CALCULATIONS WITH CHEMICAL FORMULAS AND EQUATIONS
3.7 LIMITING REACTANTS
Suppose you wish to make several sandwiches using one slice of cheese and two slices of bread for each. Using Bd = bread, Ch = cheese, and Bd2Ch = sandwich, the recipe for making a sandwich can be represented like a chemical equation:
2 Bd + Ch → Bd2Ch
If you have 10 slices of bread and 7 slices of cheese, you can make only 5 sandwiches and will have 2 slices of cheese left over. The amount of bread available limits the number of sandwiches.
An analogous situation occurs in chemical reactions when one reactant is used up before the others. The reaction stops as soon as any reactant is totally consumed, leaving the excess reactants as leftovers. Suppose, for example, we have a mixture of 10 mol H2 and 7 mol O2, which react to form water:
2 H2(g) + O2(g) → 2 H2O(g)
Because 2 mol H2![]() 1 mol O2, the number of moles of O2 needed to react with all the H2 is
1 mol O2, the number of moles of O2 needed to react with all the H2 is
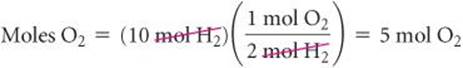
Because 7 mol O2 is available at the start of the reaction, 7 mol O2 − 5 mol O2 = 2 mol O2 is present when all the H2 is consumed.
The reactant that is completely consumed in a reaction is called the limiting reactant because it determines, or limits, the amount of product formed. The other reactants are sometimes called excess reactants. In our example, shown in ![]() FIGURE 3.17, H2 is the limiting reactant, which means that once all the H2 has been consumed, the reaction stops. The excess reactant is O2; some is left over when the reaction stops.
FIGURE 3.17, H2 is the limiting reactant, which means that once all the H2 has been consumed, the reaction stops. The excess reactant is O2; some is left over when the reaction stops.
There are no restrictions on the starting amounts of reactants in any reaction. Indeed, many reactions are carried out using an excess of one reactant. The quantities of reactants consumed and products formed, however, are restricted by the quantity of the limiting reactant. For example, when a combustion reaction takes place in the open air, oxygen is plentiful and is therefore the excess reactant. If you run out of gasoline while driving, the car stops because the gasoline is the limiting reactant in the combustion reaction that moves the car.
![]() GO FIGURE
GO FIGURE
If O2 had been the limiting reactant, how many moles of H2O would have formed?
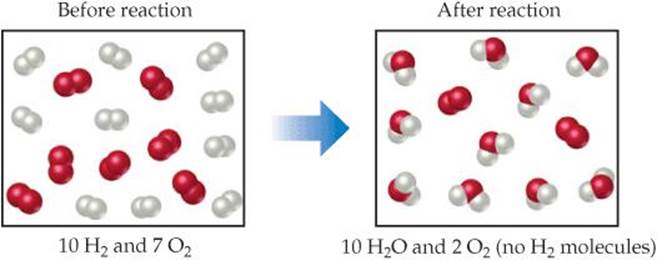
![]() FIGURE 3.17 Limiting reactant. Because H2 is completely consumed, it is the limiting reactant. Because some O2 is left over after the reaction is complete, O2 is the excess reactant. The amount of H2O formed depends on the amount of limiting reactant, H2.
FIGURE 3.17 Limiting reactant. Because H2 is completely consumed, it is the limiting reactant. Because some O2 is left over after the reaction is complete, O2 is the excess reactant. The amount of H2O formed depends on the amount of limiting reactant, H2.
Before we leave the example illustrated in Figure 3.17, let's summarize the data:

The second line in the table (Change) summarizes the amounts of reactants consumed (where this consumption is indicated by the minus signs) and the amount of the product formed (indicated by the plus sign). These quantities are restricted by the quantity of the limiting reactant and depend on the coefficients in the balanced equation. The mole ratio H2:O2:H2O = 10:5:10 conforms to the ratio of the coefficients in the balanced equation, 2:1:2. The final quantities, which depend on the initial quantities and their changes, are found by adding the initial quantity and change quantity for each column. None of the limiting reactant (H2) remains at the end of the reaction. What remains is 2 mol O2 (excess reactant) and 10 mol H2O (product).
SAMPLE EXERCISE 3.18 Calculating the Amount of Product Formed from a Limiting Reactant
The most important commercial process for converting N2 from the air into nitrogen-containing compounds is based on the reaction of N2 and H2 to form ammonia (NH3):
N2(g) + 3 H2(g) → 2 NH3(g)
How many moles of NH3 can be formed from 3.0 mol of N2 and 6.0 mol of H2?
SOLUTION
Analyze We are asked to calculate the number of moles of product, NH3, given the quantities of each reactant, N2 and H2, available in a reaction. This is a limiting reactant problem.
Plan If we assume one reactant is completely consumed, we can calculate how much of the second reactant is needed. By comparing the calculated quantity of the second reactant with the amount available, we can determine which reactant is limiting. We then proceed with the calculation, using the quantity of the limiting reactant.
Solve
The number of moles of H2 needed for complete consumption of 3.0 mol of N2 is:
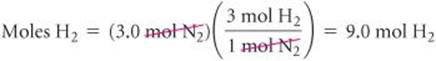
Because only 6.0 mol H2 is available, we will run out of H2 before the N2 is gone, which tells us that H2 is the limiting reactant. Therefore, we use the quantity of H2 to calculate the quantity of NH3 produced:
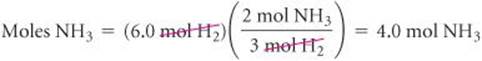
Comment The table on the right summarizes this example:

Notice that we can calculate not only the number of moles of NH3 formed but also the number of moles of each reactant remaining after the reaction. Notice also that although the initial number of moles of H2 is greater than the final number of moles of N2, the H2 is nevertheless the limiting reactant because of its larger coefficient in the balanced equation.
Check The Change row of the summary table shows that the mole ratio of reactants consumed and product formed, 2:6:4, conforms to the coefficients in the balanced equation, 1:3:2. Because H2 is the limiting reactant, it is completely consumed in the reaction, leaving 0 mol at the end. Because 6.0 mol H2 has two significant figures, our answer has two significant figures.
PRACTICE EXERCISE
(a) When 1.50 mol of Al and 3.00 mol of Cl2 combine in the reaction 2 Al(s) + 3 Cl2(g) → 2 AlCl3(s), which is the limiting reactant? (b) How many moles of AlCl3 are formed? (c) How many moles of the excess reactant remain at the end of the reaction?
Answers: (a) Al, (b) 1.50 mol, (c) 0.75 mol Cl2
SAMPLE EXERCISE 3.19 Calculating the Amount of Product Formed from a Limiting Reactant
The reaction
2 H2(g) + O2(g) → 2 H2O(g)
is used to produce electricity in a hydrogen fuel cell. Suppose a fuel cell contains 150 g of H2(g) and 1500 g of O2(g) (each measured to two significant figures). How many grams of water can form?
SOLUTION
Analyze We are asked to calculate the amount of a product, given the amounts of two reactants, so this is a limiting reactant problem.
Plan To identify the limiting reactant, we can calculate the number of moles of each reactant and compare their ratio with the ratio of coefficients in the balanced equation. We then use the quantity of the limiting reactant to calculate the mass of water that forms.
Solve From the balanced equation, we have the stoichiometric relations
2 mol H2![]() 1 mol O2
1 mol O2![]() 2 mol H2O
2 mol H2O
Using the molar mass of each substance, we calculate the number of moles of each reactant:
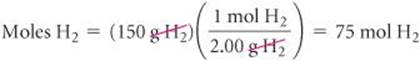
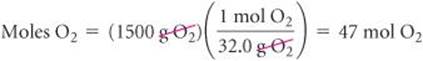
The coefficients in the balanced equation indicate that the reaction requires 2 mol of H2 for every 1 mol of O2. Therefore, for all the O2 to completely react, we would need 2 × 47 = 94 mol of H2. Since there are only 75 mol of H2, all of the O2 cannot react, so it is the excess reactant, and H2must be the limiting reactant. (Notice that the limiting reactant is not merely the one present in the lowest amount.)
We use the given quantity of H2 (the limiting reactant) to calculate the quantity of water formed. We could begin this calculation with the given H2 mass, 150 g, but we can save a step by starting with the moles of H2, 75 mol, we just calculated:
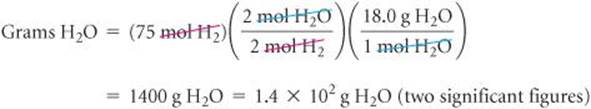
Check The magnitude of the answer seems reasonable based on the amounts of the reactants. The units are correct, and the number of significant figures (two) corresponds to those in the values given in the problem statement.
Comment The quantity of the limiting reactant, H2, can also be used to determine the quantity of O2 used:
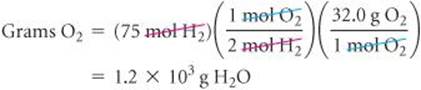
The mass of O2 remaining at the end of the reaction equals the starting amount minus the amount consumed:
1500 g – 1200 g = 300 g.
PRACTICE EXERCISE
When a 2.00-g strip of zinc metal is placed in an aqueous solution containing 2.50 g of silver nitrate, the reaction is
Zn(s) + 2 AgNO3(aq) → 2 Ag(s) + Zn(NO3)2(aq)
(a) Which reactant is limiting? (b) How many grams of Ag form? (c) How many grams of Zn(NO3)2 form? (d) How many grams of the excess reactant are left at the end of the reaction?
Answers: (a) AgNO3, (b) 1.59 g, (c) 1.39 g, (d) 1.52 g Zn
Theoretical Yields
The quantity of product calculated to form when all of a limiting reactant is consumed is called the theoretical yield. The amount of product actually obtained, called the actual yield, is almost always less than (and can never be greater than) the theoretical yield. There are many reasons for this difference. Part of the reactants may not react, for example, or they may react in a way different from that desired (side reactions). In addition, it is not always possible to recover all of the product from the reaction mixture. The percent yield of a reaction relates actual and theoretical yields:
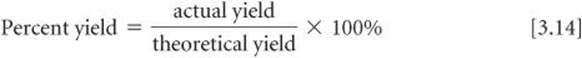
SAMPLE EXERCISE 3.20 Calculating Theoretical Yield and Percent Yield
Adipic acid, H2C6H8O4, used to produce nylon, is made commercially by a reaction between cyclohexane (C6H12) and O2:
2 C6H12(l) + 5 O2(g) → 2 H2C6H8O4(l) + 2 H2O(g)
(a) Assume that you carry out this reaction with 25.0 g of cyclohexane and that cyclohexane is the limiting reactant. What is the theoretical yield of adipic acid? (b) If you obtain 33.5 g of adipic acid, what is the percent yield for the reaction?
SOLUTION
Analyze We are given a chemical equation and the quantity of the limiting reactant (25.0 g of C6H12). We are asked to calculate the theoretical yield of a product H2C6H8O4 and the percent yield if only 33.5 g of product is obtained.
Plan
(a) The theoretical yield, which is the calculated quantity of adipic acid formed, can be calculated using the sequence of conversions shown in Figure 3.16.
(b) The percent yield is calculated by using Equation 3.14 to compare the given actual yield (33.5 g) with the theoretical yield.
Solve
(a) The theoretical yield is
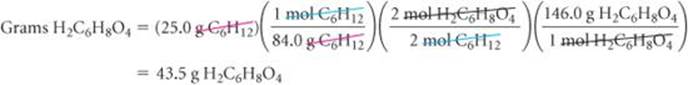
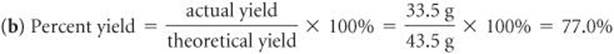
Check We can check our answer in (a) by doing a ballpark calculation. From the balanced equation we know that each mole of cyclohexane gives 1 mol adipic acid. We have 25/84 ≈ 25.75 = 0.3 mol hexane, so we expect 0.3 mol adipic acid, which equals about 0.3 × 150 = 45 g, about the same magnitude as the 43.5 g obtained in the more detailed calculation given previously. In addition, our answer has the appropriate units and significant figures. In (b) the answer is less than 100%, as it must be from the definition of percent yield.
PRACTICE EXERCISE
Imagine you are working on ways to improve the process by which iron ore containing Fe2O3 is converted into iron:
Fe2O3(s) + 3 CO(g) → 2 Fe(s) + 3 CO2(g)
(a) If you start with 150 g of Fe2O3 as the limiting reactant, what is the theoretical yield of Fe?
(b) If your actual yield is 87.9 g, what is the percent yield?
Answers: (a) 105 g Fe, (b) 83.7%
 STRATEGIES IN CHEMISTRY
STRATEGIES IN CHEMISTRY
HOW TO TAKE A TEST
At about this time in your study of chemistry, you are likely to face your first hour-long examination. The best way to prepare is to study, do homework diligently, and get help from the instructor on any material that is unclear or confusing. (See the advice for learning and studying chemistry presented in the preface of the book.) We present here some general guidelines for taking tests.
Depending on the nature of your course, the exam could consist of a variety of different types of questions.
1. Multiple-choice questions In large-enrollment courses, the most common kind of test question is the multiple-choice question. You are given the problem and presented with four or five answers from which you must select the correct one. The first thing to realize is that the instructor has written the question so that at first glance all the answers appear to be correct. (There would be little point in offering choices you could tell were wrong even without knowing much about the concept being tested.) Thus, you should not jump to the conclusion that because one of the choices looks correct, it must be correct.
If a multiple-choice question involves a calculation, do the calculation, check your work, and only then compare your answer with the choices. If you find a match, you have probably found the correct answer. Keep in mind, though, that your instructor has anticipated the most common errors you might make in solving a given problem and has probably listed the incorrect answers resulting from those errors. Always double-check your reasoning and use dimensional analysis to arrive at the correct numeric answer and the correct units.
In multiple-choice questions that do not involve calculations, if you are not sure of the correct choice, eliminate all the choices you know for sure to be incorrect. The reasoning you use in eliminating incorrect choices will help you in reasoning about which of the remaining choices is correct.
2. Calculations in which you must show your work In questions of this kind, you may receive partial credit even if you do not arrive at the correct answer, depending on whether the instructor can follow your line of reasoning. It is important, therefore, to be neat and organized in your calculations. Pay particular attention to what information is given and to what your unknown is. Think about how you can get from the given information to your unknown.
You may want to write a few words or a diagram on the test paper to indicate your approach. Then write out your calculations as neatly as you can. Show the units for every number you write down, and use dimensional analysis as much as you can, showing how units cancel.
3. Questions requiring drawings Questions of this kind will come later in the course, but it is useful to talk about them here. (You should review this box before each exam to remind yourself of good exam-taking practices.) Be sure to label your drawing as completely as possible.
4. Other types of questions Other exam questions you might encounter include true-false questions and ones in which you are given a list and asked to indicate which members of the list match some criterion given in the question. Often students answer such questions incorrectly because, in their haste, they misunderstand the nature of the question. Whatever the form of the question, ask yourself this: What is the instructor testing here? What material am I supposed to know that this question covers?
Finally, if you find that you simply do not understand how to arrive at a reasoned response to a question, do not linger over the question. Put a check next to it and go on to the next one. If time permits, you can come back to the unanswered questions, but lingering over a question when nothing is coming to mind is wasting time you may need to finish the exam.