CHEMISTRY THE CENTRAL SCIENCE
3 STOICHIOMETRY: CALCULATIONS WITH CHEMICAL FORMULAS AND EQUATIONS
EXERCISES
VISUALIZING CONCEPTS
3.1 The reaction between reactant A (blue spheres) and reactant B (red spheres) is shown in the following diagram:
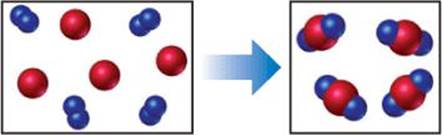
Based on this diagram, which equation best describes the reaction? [Section 3.1]
(a) A2 + B → A2B
(b) A2 + 4 B → 2 AB2
(c) 2 A + B4 → 2 AB2
(d) A + B2 → AB2
3.2 Under appropriate experimental conditions, H2 and CO undergo a combination reaction to form CH3OH. The following drawing represents a sample of H2. Make a corresponding drawing of the CO needed to react completely with the H2. How did you arrive at the number of CO molecules in your drawing? [Section 3.2]

3.3 The following diagram represents the collection of elements formed by a decomposition reaction. (a) If the blue spheres represent N atoms and the red ones represent O atoms, what was the empirical formula of the original compound? (b) Could you draw a diagram representing the molecules of the compound that had been decomposed? Why or why not? [Section 3.2]
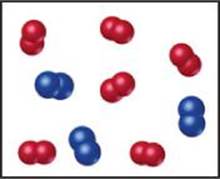
3.4 The following diagram represents the collection of CO2 and H2O molecules formed by complete combustion of a hydrocarbon. What is the empirical formula of the hydrocarbon? [Section 3.2]
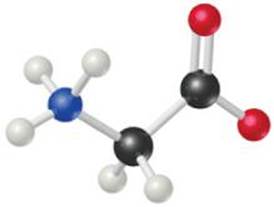
3.5 Glycine, an amino acid used by organisms to make proteins, is represented by the following molecular model.
(a) Write its molecular formula.
(b) Determine its molar mass.
(c) Calculate the mass of 3 moles of glycine.
(d) Calculate the percent nitrogen by mass in glycine. [Sections 3.3 and 3.5]
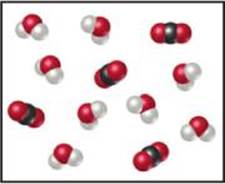
3.6 The following diagram represents a high-temperature reaction between CH4 and H2O. Based on this reaction, how many moles of each product can be obtained starting with 4.0 mol CH4? [Section 3.6]
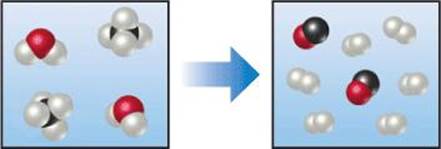
3.7 Nitrogen (N2) and hydrogen (H2) react to form ammonia (NH3). Consider the mixture of N2 and H2 shown in the accompanying diagram. The blue spheres represent N, and the white ones represent H. Draw a representation of the product mixture, assuming that the reaction goes to completion. How did you arrive at your representation? What is the limiting reactant in this case? [Section 3.7]
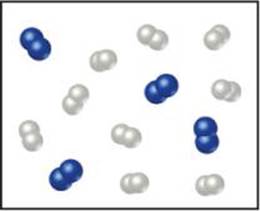
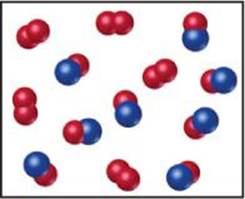
3.8 Nitrogen monoxide and oxygen react to form nitrogen dioxide. Consider the mixture of NO and O2 shown in the accompanying diagram. The blue spheres represent N, and the red ones represent O. (a) Draw a representation of the product mixture, assuming that the reaction goes to completion. What is the limiting reactant in this case? (b) How many NO2 molecules would you draw as products if the reaction had a percent yield of 75%? [Section 3.7]
BALANCING CHEMICAL EQUATIONS (section 3.1)
3.9 (a) What scientific principle or law is used in the process of balancing chemical equations? (b) In balancing equations, why should you not change subscripts in chemical formulas? (c) How would you write out liquid water, water vapor, aqueous sodium chloride, and solid sodium chloride in chemical equations?
3.10 (a) What is the difference between adding a subscript 2 to the end of the formula for CO to give CO2 and adding a coefficient in front of the formula to give 2 CO? (b) Is the following chemical equation, as written, consistent with the law of conservation of mass?
3 Mg(OH)2(s) + 2 H3PO4(aq) → Mg3(PO4)2(s) + 6 H2O(l)
Why or why not?
______
3.11 Balance the following equations:
(a) CO(g) + O2(g) → CO2(g)
(b) N2O5(g) + H2O(l) → HNO3(aq)
(c) CH4(g) + Cl2(g) → CCl4(l) + HCl(g)
(d) Al4C3(s) + H2O(l) → Al(OH)3(s) + CH4(g)
(e) C5H10O2(l) + O2(g) → CO2(g) + H2O(g)
(f) Fe(OH)3(s) + H2SO4(aq) → Fe2(SO4)3(aq) + H2O(l)
(g) Mg3N2(s) + H2SO4(aq) → MgSO4(aq) + (NH4)2SO4(aq)
3.12 Balance the following equations:
(a) Li(s) + N2(g) → Li3N(s)
(b) TiCl4(l) + H2O(l) → TiO2(s) + HCl(aq)
(c) NH4NO3(s) → N2(g) + O2(g) + H2O(g)
(d) Ca3P2(s) + H2O(l) → Ca(OH)2(aq) + PH3(g)
(e) Al(OH)3(s) + H2SO4(aq) → Al2(SO4)3(aq) + H2O(l)
(f) AgNO3(aq) + Na2CO3(aq) → Ag2NO3(s) + Na2CO3(aq)
(g) C2H5NH2(g) + O2(g) → CO2(g) + H2O(g) + N2(g)
______
3.13 Write balanced chemical equations to correspond to each of the following descriptions: (a) Solid calcium carbide, CaC2, reacts with water to form an aqueous solution of calcium hydroxide and acetylene gas, C2H2. (b) When solid potassium chlorate is heated, it decomposes to form solid potassium chloride and oxygen gas. (c) Solid zinc metal reacts with sulfuric acid to form hydrogen gas and an aqueous solution of zinc sulfate. (d) When liquid phosphorus trichloride is added to water, it reacts to form aqueous phosphorous acid, H3PO3(aq), and aqueous hydrochloric acid. (e) When hydrogen sulfide gas is passed over solid hot iron(III) hydroxide, the resultant reaction produces solid iron(III) sulfide and gaseous water.
3.14 Write balanced chemical equations to correspond to each of the following descriptions: (a) When sulfur trioxide gas reacts with water, a solution of sulfuric acid forms. (b) Boron sulfide, B2S3(s), reacts violently with water to form dissolved boric acid, H3BO3, and hydrogen sulfide gas. (c) Phosphine, PH3(g), combusts in oxygen gas to form water vapor and solid tetraphosphorus decaoxide. (d) When solid mercury(II) nitrate is heated, it decomposes to form solid mercury(II) oxide, gaseous nitrogen dioxide, and oxygen. (e) Copper metal reacts with hot concentrated sulfuric acid solution to form aqueous copper(II) sulfate, sulfur dioxide gas, and water.
PATTERNS OF CHEMICAL REACTIVITY (section 3.2)
3.15 (a) When the metallic element sodium combines with the nonmetallic element bromine, Br2(l), how can you determine the chemical formula of the product? How do you know whether the product is a solid, liquid, or gas at room temperature? Write the balanced chemical equation for the reaction. (b) When a hydrocarbon burns in air, what reactant besides the hydrocarbon is involved in the reaction? What products are formed? Write a balanced chemical equation for the combustion of benzene, C6H6(l), in air.
3.16 (a) Determine the chemical formula of the product formed when the metallic element aluminum combines with the non-metallic element bromine, Br2. Write the balanced chemical equation for the reaction. (b) What products form when a compound containing C, H, and O is completely combusted in air? Write a balanced chemical equation for the combustion of acetone, C3H6O(l), in air.
______
3.17 Write a balanced chemical equation for the reaction that occurs when (a) Mg(s) reacts with Cl2(g); (b) barium carbonate decomposes into barium oxide and carbon dioxide gas when heated; (c) the hydrocarbon styrene, C8H8(l), is combusted in air; (d) dimethylether, CH3OCH3(g), is combusted in air.
3.18 Write a balanced chemical equation for the reaction that occurs when (a) calcium metal undergoes a combination reaction with O2(g); (b) copper(II) hydroxide decomposes into copper(II) oxide and water when heated; (c) heptane, C7H16(l), burns in air; (d) methyl tert-butyl ether, C5H12O(l), burns in air.
______
3.19 Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions:
(a) C3H6(g) + O2(g) → CO2(g) + H2O(g)
(b) NH4NO3(s) → N2O(g) + H2O(g)
(c) C5H6O(l) + O2(g) → CO2(g) + H2O(g)
(d) N2(g) + H2(g) → NH3(g)
(e) K2O(s) + H2O(l) → KOH(aq)
3.20 Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions:
(a) PbCO3(s) → PbO(s) + CO2(g)
(b) C2H4(g) + O2(g) → CO2(g) + H2O(g)
(c) Mg(s) + N2(g) → Mg3N2(s)
(d) C7H8O2(l) + O2(g) → CO2(g) + H2O(g)
(e) Al(s) + Cl2(g) → AlCl3(s)
FORMULA WEIGHTS (section 3.3)
3.21 Determine the formula weights of each of the following compounds: (a) nitric acid, HNO3; (b) KMnO4; (c) Ca3(PO4)2; (d) quartz, SiO2; (e) gallium sulfide, (f) chromium(III) sulfate, (g) phosphorus trichloride.
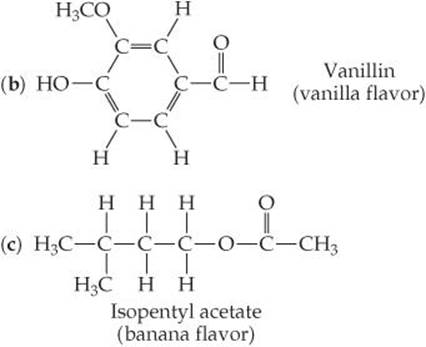
3.22 Determine the formula weights of each of the following compounds: (a) nitrous oxide, N2O, known as laughing gas and used as an anesthetic in dentistry; (b) benzoic acid, HC7H5O2, a substance used as a food preservative; (c) Mg(OH)2, the active ingredient in milk of magnesia;(d) urea, (NH2)2CO, a compound used as a nitrogen fertilizer; (e) isopentyl acetate, CH3CO2C5H11, responsible for the odor of bananas.
______
3.23 Calculate the percentage by mass of oxygen in the following compounds: (a) morphine, C17H19NO3; (b) codeine, C18H21NO3(c) cocaine, C17H21NO4; (d) tetracycline, C22H24N2O8; (e) digitoxin, C41H64O13; (f) vancomycin, C66H75Cl2NO24.
3.24 Calculate the percentage by mass of the indicated element in the following compounds: (a) carbon in acetylene, C2H2, a gas used in welding; (b) hydrogen in ascorbic acid, HC6H7O6, also known as vitamin C; (c) hydrogen in ammonium sulfate, (NH4)2SO4, a substance used as a nitrogen fertilizer; (d) platinum in PtCl2(NH3)2, a chemotherapy agent called cisplatin; (e) oxygen in the female sex hormone estradiol, C18H24O2; (f) carbon in capsaicin, C18H27NO3, the compound that gives the hot taste to chili peppers.
______
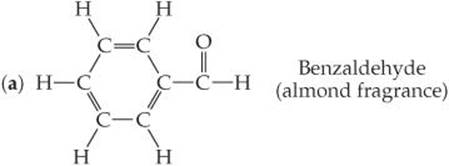
3.25 Based on the following structural formulas, calculate the percentage of carbon by mass present in each compound:
3.26 Calculate the percentage of carbon by mass in each of the compounds represented by the following models:
AVOGADRO'S NUMBER AND THE MOLE (section 3.4)
3.27 (a) What is Avogadro's number, and how is it related to the mole? (b) What is the relationship between the formula weight of a substance and its molar mass?
3.28 (a) What is the mass, in grams, of a mole of 12C? (b) How many carbon atoms are present in a mole of 12C?
______
3.29 Without doing any detailed calculations (but using a periodic table to give atomic weights), rank the following samples in order of increasing number of atoms: 0.50 mol H2O, 23 g Na, 6.0 × 1023 N2 molecules.
3.30 Without doing any detailed calculations (but using a periodic table to give atomic weights), rank the following samples in order of increasing number of atoms: 9.0 × 1023 molecules of H2O2, 2.0 mol CH4, 16 g O2.
______
3.31 What is the mass, in kilograms, of an Avogadro's number of people, if the average mass of a person is 160 lb? How does this compare with the mass of Earth, 5.98 × 1024 kg?
3.32 If Avogadro's number of pennies is divided equally among the 300 million men, women, and children in the United States, how many dollars would each receive? How does this compare with the gross domestic product (GDP) of the United States, which was $14.4 trillion in 2008? (The GDP is the total market value of the nation's goods and services.)
______
3.33 Calculate the following quantities:
(a) mass, in grams, of 0.105 mole of sucrose (C12H22O11)
(b) moles of Zn(NO3)2 in 143.50 g of this substance
(c) number of molecules in 1.0 × 10−6 mol CH3CH2OH
(d) number of N atoms in 0.410 mol NH3
3.34 Calculate the following quantities:
(a) mass, in grams, of 1.50 × 10−2 mol of CdS
(b) number of moles of NH4Cl in 86.6 g of this substance
(c) number of molecules in 8.447 × 10−2 mol C6H6
(d) number of O atoms in 6.25 × 10−3 mol Al(NO3)3
______
3.35 (a) What is the mass, in grams, of 2.50 × 10−3 mol of ammonium phosphate?
(b) How many moles of chloride ions are in 0.2550 g of aluminum chloride?
(c) What is the mass, in grams, of 7.70 × 1020 molecules of caffeine, C8H10N4O2?
(d) What is the molar mass of cholesterol if 0.00105 mol has a mass of 0.406 g?
3.36 (a) What is the mass, in grams, of 1.223 mol of iron(III) sulfate?
(b) How many moles of ammonium ions are in 6.955 g of ammonium carbonate?
(c) What is the mass, in grams, of 1.50 × 1021 molecules of aspirin, C9H8O4?
(d) What is the molar mass of diazepam (Valium®) if 0.05570 mol has a mass of 15.86 g?
______
3.37 The molecular formula of allicin, the compound responsible for the characteristic smell of garlic, is C6H10OS2. (a) What is the molar mass of allicin? (b) How many moles of allicin are present in 5.00 mg of this substance? (c) How many molecules of allicin are in 5.00 mg of this substance? (d) How many S atoms are present in 5.00 mg of allicin?
3.38 The molecular formula of aspartame, the artificial sweetener marketed as NutraSweet®, is C14H18N2O5. (a) What is the molar mass of aspartame? (b) How many moles of aspartame are present in 1.00 mg of aspartame? (c) How many molecules of aspartame are present in 1.00 mg of aspartame? (d) How many hydrogen atoms are present in 1.00 mg of aspartame?
______
3.39 A sample of glucose, C6H12O6, contains 1.250 × 1021 carbon atoms. (a) How many atoms of hydrogen does it contain? (b) How many molecules of glucose does it contain? (c) How many moles of glucose does it contain? (d) What is the mass of this sample in grams?
3.40 A sample of the male sex hormone testosterone, C19H28O2, contains 3.88 × 1021 hydrogen atoms. (a) How many atoms of carbon does it contain? (b) How many molecules of testosterone does it contain? (c) How many moles of testosterone does it contain? (d) What is the mass of this sample in grams?
______
3.41 The allowable concentration level of vinyl chloride, C2H3Cl, in the atmosphere in a chemical plant is 2.0 × 10−6 g/L. How many moles of vinyl chloride in each liter does this represent? How many molecules per liter?
3.42 At least 25 μg of tetrahydrocannabinol (THC), the active ingredient in marijuana, is required to produce intoxication. The molecular formula of THC is C21H30O2. How many moles of THC does this 25 μg represent? How many molecules?
EMPIRICAL FORMULAS (section 3.5)
3.43 Give the empirical formula of each of the following compounds if a sample contains (a) 0.0130 mol C, 0.0390 mol H, and 0.0065 mol O; (b) 11.66 g iron and 5.01 g oxygen; (c) 40.0% C, 6.7% H, and 53.3% O by mass.
3.44 Determine the empirical formula of each of the following compounds if a sample contains (a) 0.104 mol K, 0.052 mol C, and 0.156 mol O; (b) 5.28 g Sn and 3.37 g F; (c) 87.5% N and 12.5% H by mass.
______
3.45 Determine the empirical formulas of the compounds with the following compositions by mass:
(a) 10.4% C, 27.8% S, and 61.7% Cl
(b) 21.7% C, 9.6% O, and 68.7% F
(c) 32.79% Na, 13.02% Al, and the remainder F
3.46 Determine the empirical formulas of the compounds with the following compositions by mass:
(a) 55.3% K, 14.6% P, and 30.1% O
(b) 24.5% Na, 14.9% Si, and 60.6% F
(c) 62.1% C, 5.21% H, 12.1% N, and the remainder O
______
3.47 A compound whose empirical formula is XF3 consists of 65% F by mass. What is the atomic mass of X?
3.48 The compound XCl4 contains 75.0% Cl by mass. What is the element X?
______
3.49 What is the molecular formula of each of the following compounds?
(a) empirical formula CH2, molar mass = 84 g/mol
(b) empirical formula NH2Cl, molar mass = 51.5 g/mol
3.50 What is the molecular formula of each of the following compounds?
(a) empirical formula HCO2, molar mass = 90.0 g/mol
(b) empirical formula C2H4O, molar mass = 88 g/mol
______
3.51 Determine the empirical and molecular formulas of each of the following substances:
(a) Styrene, a compound substance used to make Styrofoam® cups and insulation, contains 92.3% C and 7.7% H by mass and has a molar mass of 104 g/mol.
(b) Caffeine, a stimulant found in coffee, contains 49.5% C, 5.15% H, 28.9% N, and 16.5% O by mass and has a molar mass of 195 g/mol.
(c) Monosodium glutamate (MSG), a flavor enhancer in certain foods, contains 35.51% C, 4.77% H, 37.85% O, 8.29% N, and 13.60% Na, and has a molar mass of 169 g/mol.
3.52 Determine the empirical and molecular formulas of each of the following substances:
(a) Ibuprofen, a headache remedy, contains 75.69% C, 8.80% H, and 15.51% O by mass, and has a molar mass of 206 g/mol.
(b) Cadaverine, a foul-smelling substance produced by the action of bacteria on meat, contains 58.55% C, 13.81% H, and 27.40% N by mass; its molar mass is 102.2 g/mol.
(c) Epinephrine (adrenaline), a hormone secreted into the bloodstream in times of danger or stress, contains 59.0% C, 7.1% H, 26.2% O, and 7.7% N by mass; its MW is about 180 amu.
______
3.53 (a) Combustion analysis of toluene, a common organic solvent, gives 5.86 mg of CO2 and 1.37 mg of H2O. If the compound contains only carbon and hydrogen, what is its empirical formula? (b) Menthol, the substance we can smell in mentholated cough drops, is composed of C, H, and O. A 0.1005-g sample of menthol is combusted, producing 0.2829 g of CO2 and 0.1159 g of H2O. What is the empirical formula for menthol? If menthol has a molar mass of 156 g/mol, what is its molecular formula?
3.54 (a) The characteristic odor of pineapple is due to ethyl butyrate, a compound containing carbon, hydrogen, and oxygen. Combustion of 2.78 mg of ethyl butyrate produces 6.32 mg of CO2 and 2.58 mg of H2O. What is the empirical formula of the compound? (b) Nicotine, a component of tobacco, is composed of C, H, and N. A 5.250-mg sample of nicotine was combusted, producing 14.242 mg of CO2 and 4.083 mg of H2O. What is the empirical formula for nicotine? If nicotine has a molar mass of 160 ± 5 g/mol, what is its molecular formula?
______
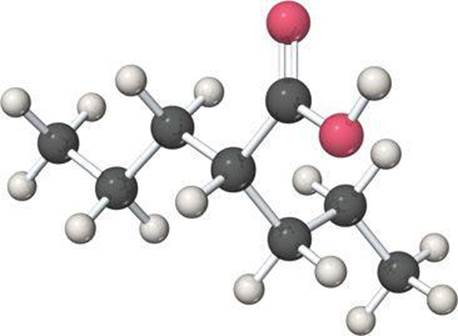
3.55 Valproic acid, used to treat seizures and bipolar disorder, is composed of C, H, and O. A 0.165-g sample is combusted in an apparatus such as that shown in Figure 3.14. The gain in mass of the H2O absorber is 0.166 g, whereas that of the CO2 absorber is 0.403 g. What empirical formula for valproic acid do these results indicate? Is this empirical formula consistent with the molecular model shown here?
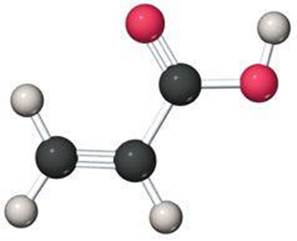
3.56 Propenoic acid, as shown here, is a reactive organic liquid used in the manufacture of plastics, coatings, and adhesives. An unlabeled container is thought to contain this acid. A 0.2033-g sample is combusted in an apparatus such as that shown in Figure 3.14. The gain in mass of the H2O absorber is 0.102 g, whereas that of the CO2 absorber is 0.374 g. Is this analysis consistent with the contents of the container being propenoic acid?
______
3.57 Washing soda, a compound used to prepare hard water for washing laundry, is a hydrate, which means that a certain number of water molecules are included in the solid structure. Its formula can be written as Na2CO3 • xH2O, where x is the number of moles of H2O per mole of Na2CO3. When a 2.558-g sample of washing soda is heated at 25 °C, all the water of hydration is lost, leaving 0.948 g of Na2CO3. What is the value of x?
3.58 Epsom salts, a strong laxative used in veterinary medicine, is a hydrate, which means that a certain number of water molecules are included in the solid structure. The formula for Epsom salts can be written as MgSO4 • xH2O, where x indicates the number of moles of H2O per mole of MgSO4. When 5.061 g of this hydrate is heated to 250 °C, all the water of hydration is lost, leaving 2.472 g of MgSO4. What is the value of x?
CALCULATIONS BASED ON CHEMICAL EQUATIONS (section 3.6)
3.59 Why is it essential to use balanced chemical equations when determining the quantity of a product formed from a given quantity of a reactant?
3.60 What parts of balanced chemical equations give information about the relative numbers of moles of reactants and products involved in a reaction?
______
3.61 Hydrofluoric acid, HF(aq), cannot be stored in glass bottles because compounds called silicates in the glass are attacked by the HF(aq). Sodium silicate (Na2SiO3), for example, reacts as follows:
Na2SiO3(s) + 8 HF(aq) → H2SiF6(aq) + 2 NaF(aq) + 3 H2O(l)
(a) How many moles of HF are needed to react with 0.300 mol of Na2SiO3?
(b) How many grams of NaF form when 0.500 mol of HF reacts with excess Na2SiO3?
(c) How many grams of Na2SiO3 can react with 0.800 g of HF?
3.62 The reaction between potassium superoxide, KO2, and CO2,
4 KO2 + 2 CO2 → 2K2CO3 + 3 O2
is used as a source of O2 and absorber of CO2 in self-contained breathing equipment used by rescue workers.

(a) How many moles of O2 are produced when 0.400 mol of KO2 reacts in this fashion?
(b) How many grams of KO2 are needed to form 7.50 g of O2?
(c) How many grams of CO2 are used when 7.50 g of O2 are produced?
______
3.63 Several brands of antacids use Al(OH)3 to react with stomach acid, which contains primarily HCl:
Al(OH)3(s) + HCl(aq) → AlCl3(aq) + H2O(l)
(a) Balance this equation.
(b) Calculate the number of grams of HCl that can react with 0.500 g of Al(OH)3.
(c) Calculate the number of grams of AlCl3 and the number of grams of H2O formed when 0.500 g of Al(OH)3 reacts.
(d) Show that your calculations in parts (b) and (c) are consistent with the law of conservation of mass.
3.64 An iron ore sample contains Fe2O3 together with other substances. Reaction of the ore with CO produces iron metal:
Fe2O3(s) + CO(g) → Fe(s) + CO2(g)
(a) Balance this equation.
(b) Calculate the number of grams of CO that can react with 0.350 kg of Fe2O3.
(c) Calculate the number of grams of Fe and the number of grams of CO2 formed when 0.350 kg of Fe2O3 reacts.
(d) Show that your calculations in parts (b) and (c) are consistent with the law of conservation of mass.
______
3.65 Aluminum sulfide reacts with water to form aluminum hydroxide and hydrogen sulfide. (a) Write the balanced chemical equation for this reaction. (b) How many grams of aluminum hydroxide are obtained from 14.2 g of aluminum sulfide?
3.66 Calcium hydride reacts with water to form calcium hydroxide and hydrogen gas. (a) Write a balanced chemical equation for the reaction. (b) How many grams of calcium hydride are needed to form 4.500 g of hydrogen?
______
3.67 Automotive air bags inflate when sodium azide, NaN3, rapidly decomposes to its component elements:
2 NaN3(s) → 2 Na(s) + 3 N2(g)
(a) How many moles of N2 are produced by the decomposition of 1.50 mol of NaN3?
(b) How many grams of NaN3 are required to form 10.0 g of nitrogen gas?
(c) How many grams of NaN3 are required to produce 10.0 ft3 of nitrogen gas, about the size of an automotive air bag, if the gas has a density of 1.25 g/L?
3.68 The complete combustion of octane, C8H18, the main component of gasoline, proceeds as follows:
2 C8H18(l) + 25 O2(g) → 16 CO2(g) + 18 H2O(g)
(a) How many moles of O2 are needed to burn 1.50 mol of C8H18?
(b) How many grams of O2 are needed to burn 10.0 g of C8H18?
(c) Octane has a density of 0.692 g/mL at 20 °C. How many grams of O2 are required to burn 15.0 gal of C8H18 (the capacity of an average fuel tank)?
(d) How many grams of CO2 are produced when 15.0 gal of C8H18 are combusted?
______
3.69 A piece of aluminum foil 1.00 cm square and 0.550 mm thick is allowed to react with bromine to form aluminum bromide.

(a) How many moles of aluminum were used? (The density of aluminum is 2.699 g/cm3.) (b) How many grams of aluminum bromide form, assuming the aluminum reacts completely?
3.70 Detonation of nitroglycerin proceeds as follows:
4 C3H5N3O9(l) → 12 CO2(g) + 6 N2(g) + O2(g) + 10 H2O(g)
(a) If a sample containing 2.00 mL of nitroglycerin (density = 1.592 g/mL) is detonated, how many total moles of gas are produced? (b) If each mole of gas occupies 55 L under the conditions of the explosion, how many liters of gas are produced? (c) How many grams of N2 are produced in the detonation?
LIMITING REACTANTS (section 3.7)
3.71 (a) Define the terms limiting reactant and excess reactant. (b) Why are the amounts of products formed in a reaction determined only by the amount of the limiting reactant? (c) Why should you base your choice of which compound is the limiting reactant on its number of initial moles, not on its initial mass in grams?
3.72 (a) Define the terms theoretical yield, actual yield, and percent yield. (b) Why is the actual yield in a reaction almost always less than the theoretical yield? (c) Can a reaction ever have 110% actual yield?
______
3.73 A manufacturer of bicycles has 4815 wheels, 2305 frames, and 2255 handlebars. (a) How many bicycles can be manufactured using these parts? (b) How many parts of each kind are left over? (c) Which part limits the production of bicycles?
3.74 A bottling plant has 126,515 bottles with a capacity of 355 mL, 108,500 caps, and 48,775 L of beverage. (a) How many bottles can be filled and capped? (b) How much of each item is left over? (c) Which component limits the production?
______
3.75 Sodium hydroxide reacts with carbon dioxide as follows:
2 NaOH(s) + CO2(g) → Na2CO3(s) + H2O(l)
Which is the limiting reactant when 1.85 mol NaOH and 1.00 mol CO2 are allowed to react? How many moles of Na2CO3 can be produced? How many moles of the excess reactant remain after the completion of the reaction?
3.76 Aluminum hydroxide reacts with sulfuric acid as follows:
2 Al(OH)3(s) + 3 H2SO4(aq) → Al2(SO4)3(aq) + 6 H2O(l)
Which is the limiting reactant when 0.500 mol Al(OH)3 and 0.500 mol H2SO4 are allowed to react? How many moles of Al2(SO4)3 can form under these conditions? How many moles of the excess reactant remain after the completion of the reaction?
______
3.77 The fizz produced when an Alka-Seltzer® tablet is dissolved in water is due to the reaction between sodium bicarbonate (NaHCO3) and citric acid (H3C6H5O7):
3 NaHCO3(aq) + H3C6H5O7(aq) → 3 CO2(g) + 3 H2O(l) + Na3C6H5O7(aq)
In a certain experiment 1.00 g of sodium bicarbonate and 1.00 g of citric acid are allowed to react. (a) Which is the limiting reactant? (b) How many grams of carbon dioxide form? (c) How many grams of the excess reactant remain after the limiting reactant is completely consumed?

3.78 One of the steps in the commercial process for converting ammonia to nitric acid is the conversion of NH3 to NO:
4 NH3(g) + 5 O2(g) → 4 NO(g) + 6 H2O(g)
In a certain experiment, 2.00 g of NH3 reacts with 2.50 g of O2(a) Which is the limiting reactant? (b) How many grams of NO and of H2O form? (c) How many grams of the excess reactant remain after the limiting reactant is completely consumed? (d) Show that your calculations in parts (b) and (c) are consistent with the law of conservation of mass.
______
3.79 Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. A solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. How many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
3.80 Solutions of sulfuric acid and lead(II) acetate react to form solid lead(II) sulfate and a solution of acetic acid. If 5.00 g of sulfuric acid and 5.00 g of lead(II) acetate are mixed, calculate the number of grams of sulfuric acid, lead(II) acetate, lead(II) sulfate, and acetic acid present in the mixture after the reaction is complete.
______
3.81 When benzene (C6H6) reacts with bromine (Br2), bromoben-zene (C6H5Br) is obtained:
C6H6 + Br2 → C6H5Br + HBr
(a) When 30.0 g of benzene reacts with 65.0 g of bromine, what is the theoretical yield of bromobenzene? (b) If the actual yield of bromobenzene is 42.3 g, what is the percentage yield?
3.82 When ethane (C2H6) reacts with chlorine (Cl2), the main product is C2H5Cl, but other products containing Cl, such as C2H4Cl2, are also obtained in small quantities. The formation of these other products reduces the yield of C2H5Cl. (a) Calculate the theoretical yield of C2H5Cl when 125 g of C2H6 reacts with 255 g of Cl2, assuming that C2H6 and Cl2 react only to form C2H2Cl and HCl. (b) Calculate the percent yield of C2H5Cl if the reaction produces 206 g of C2H5Cl.
______
3.83 Hydrogen sulfide is an impurity in natural gas that must be removed. One common removal method is called the Claus process, which relies on the reaction:
8 H2S(g) + 4 O2(g) → S8(l) + 8 H2O(g)
Under optimal conditions the Claus process gives 98% yield of S8 from H2S. If you started with 30.0 grams of H2S and 50.0 grams of O2, how many grams of S8 would be produced, assuming 98% yield?
3.84 When hydrogen sulfide gas is bubbled into a solution of sodium hydroxide, the reaction forms sodium sulfide and water. How many grams of sodium sulfide are formed if 1.25 g of hydrogen sulfide is bubbled into a solution containing 2.00 g of sodium hydroxide, assuming that the sodium sulfide is made in 92.0% yield?
ADDITIONAL EXERCISES
3.85 Write the balanced chemical equations for (a) the complete combustion of acetic acid (CH3COOH), the main active ingredient in vinegar; (b) the decomposition of solid calcium hydroxide into solid calcium(II) oxide (lime) and water vapor; (c) the combination reaction between nickel metal and chlorine gas.
3.86 If 1.5 mol C2H5OH, 1.5 mol C3H8, and 1.5 mol CH3CH2 COCH3 are completely combusted in oxygen, which produces the largest number of moles of H2O? Which produces the least? Explain.
3.87 The effectiveness of nitrogen fertilizers depends on both their ability to deliver nitrogen to plants and the amount of nitrogen they can deliver. Four common nitrogen-containing fertilizers are ammonia, ammonium nitrate, ammonium sulfate, and urea [(NH2)2CO]. Rank these fertilizers in terms of the mass percentage nitrogen they contain.
3.88 (a) The molecular formula of acetylsalicylic acid (aspirin), one of the most common pain relievers, is C9H8O4. How many moles of C9H8O4 are in a 0.500-g tablet of aspirin? (b) How many molecules of C9H8O4 are in this tablet? (c) How many carbon atoms are in the tablet?
3.89 Very small crystals composed of 1000 to 100,000 atoms, called quantum dots, are being investigated for use in electronic devices.
(a) A quantum dot was made of solid silicon in the shape of a sphere, with a diameter of 4 nm. Calculate the mass of the quantum dot, using the density of silicon (2.3 g/cm3).
(b) How many silicon atoms are in the quantum dot?
(c) The density of germanium is 5.325 g/cm3. If you made a 4-nm quantum dot of germanium, how many Ge atoms would it contain? Assume the dot is spherical.
3.90 (a) One molecule of the antibiotic penicillin G has a mass of 5.342 × 10−21 g. What is the molar mass of penicillin G? (b) Hemoglobin, the oxygen-carrying protein in red blood cells, has four iron atoms per molecule and contains 0.340% iron by mass. Calculate the molar mass of hemoglobin.
3.91 Serotonin is a compound that conducts nerve impulses in the brain. It contains 68.2 mass percent C, 6.86 mass percent H, 15.9 mass percent N, and 9.08 mass percent O. Its molar mass is 176 g/mol. Determine its molecular formula.
3.92 The koala dines exclusively on eucalyptus leaves. Its digestive system detoxifies the eucalyptus oil, a poison to other animals. The chief constituent in eucalyptus oil is a substance called eucalyptol, which contains 77.87% C, 11.76% H, and the remainder O. (a) What is the empirical formula for this substance? (b) A mass spectrum of eucalyptol shows a peak at about 154 amu. What is the molecular formula of the substance?
3.93 Vanillin, the dominant flavoring in vanilla, contains C, H, and O. When 1.05 g of this substance is completely combusted, 2.43 g of CO2 and 0.50 g of H2O are produced. What is the empirical formula of vanillin?
[3.94] An organic compound was found to contain only C, H, and Cl. When a 1.50-g sample of the compound was completely combusted in air, 3.52 g of CO2 was formed. In a separate experiment the chlorine in a 1.00-g sample of the compound was converted to 1.27 g of AgCl. Determine the empirical formula of the compound.
[3.95] A compound, KBrOx, where x is unknown, is analyzed and found to contain 52.92% Br. What is the value of x?
[3.96] An element X forms an iodide (Xl3) and a chloride (XCl3). The iodide is quantitatively converted to the chloride when it is heated in a stream of chlorine:
2 XI3 + 3 Cl2 → 2 XCl3 + 3 I2
If 0.5000 g of Xl3 is treated, 0.2360 g of XCl3 is obtained. (a) Calculate the atomic weight of the element X. (b) Identify the element X.
3.97 A method used by the U. S. Environmental Protection Agency (EPA) for determining the concentration of ozone in air is to pass the air sample through a “bubbler” containing sodium iodide, which removes the ozone according to the following equation:
O3(g) + 2 NaI(aq) + H2O(l) → O2(g) + I2(s) + 2 NaOH(aq)
(a) How many moles of sodium iodide are needed to remove 5.95 × 10−6 mol of O3? (b) How many grams of sodium iodide are needed to remove 1.3 mg of O3?
3.98 A chemical plant uses electrical energy to decompose aqueous solutions of NaCl to give Cl2, H2, and NaOH:
2 NaCl(aq) + 2 H2O(l) → 2 NaOH(aq) + H2(g) + Cl2(g)
If the plant produces 1.5 × 106 kg (1500 metric tons) of Cl2 daily, estimate the quantities of H2 and NaOH produced.
3.99 The fat stored in a camel's hump is a source of both energy and water. Calculate the mass of H2O produced by metabolism of 1.0 kg of fat, assuming the fat consists entirely of tristearin (C57H110O6), a typical animal fat, and assuming that during metabolism, tristearin reacts with O2 to form only CO2 and H2O.
[3.100] When hydrocarbons are burned in a limited amount of air, both CO and CO2 form. When 0.450 g of a particular hydrocarbon was burned in air, 0.467 g of CO, 0.733 g of CO2, and 0.450 g of H2O were formed. (a) What is the empirical formula of the compound? (b) How many grams of O2 were used in the reaction? (c) How many grams would have been required for complete combustion?
3.101 A mixture of N2(g) and H2(g) reacts in a closed container to form ammonia, NH3(g). The reaction ceases before either reactant has been totally consumed. At this stage 3.0 mol N2, 3.0 mol H2, and 3.0 mol NH3 are present. How many moles of N2 and H2 were present originally?
[3.102] A mixture containing KClO3, K2CO3, KHCO3, and KCl was heated, producing CO2, O2, and H2O gases according to the following equations:
2 KClO3(s) → 2 KCl(s) + 3 O2(g)
2 KHCO3(s) → K2O(s) + H2O(g) + 2 CO2(g)
K2CO3(s) → K2O(s) + CO2(g)
The KCl does not react under the conditions of the reaction. If 100.0 g of the mixture produces 1.80 g of H2O, 13.20 g of CO2, and 4.00 g of O2, what was the composition of the original mixture? (Assume complete decomposition of the mixture.)
3.103 When a mixture of 10.0 g of acetylene (C2H2); and 10.0 g of oxygen (O2) is ignited, the resultant combustion reaction produces CO2 and H2O. (a) Write the balanced chemical equation for this reaction. (b) Which is the limiting reactant? (c) How many grams of C2H2, O2, CO2, and H2O are present after the reaction is complete?
3.104 Aspirin (C9H8O4) is produced from salicylic acid (C7H6O3) and acetic anhydride (C4H6O3):
C7H6O3 + C4H6O3 → C9H8O4 + HC2H3O2
(a) How much salicylic acid is required to produce 1.5 × 102 kg of aspirin, assuming that all of the salicylic acid is converted to aspirin? (b) How much salicylic acid would be required if only 80% of the salicylic acid is converted to aspirin? (c) What is the theoretical yield of aspirin if 185 kg of salicylic acid is allowed to react with 125 kg of acetic anhydride? (d) If the situation described in part (c) produces 182 kg of aspirin, what is the percentage yield?
INTEGRATIVE EXERCISES
These exercises require skills from earlier chapters as well as skills from the present chapter.
3.105 Consider a sample of calcium carbonate in the form of a cube measuring 2.005 in. on each edge. If the sample has a density of 2.71 g/cm3, how many oxygen atoms does it contain?
3.106 (a) You are given a cube of silver metal that measures 1.000 cm on each edge. The density of silver is 10.5 g/cm3. How many atoms are in this cube? (b) Because atoms are spherical, they cannot occupy all of the space of the cube. The silver atoms pack in the solid in such a way that 74% of the volume of the solid is actually filled with the silver atoms. Calculate the volume of a single silver atom. (c) Using the volume of a silver atom and the formula for the volume of a sphere, calculate the radius in angstroms of a silver atom.
3.107 (a) If an automobile travels 225 mi with a gas mileage of 20.5 mi/gal, how many kilograms of CO2 are produced? Assume that the gasoline is composed of octane, C8H18(l), whose density is 0.69 g/mL. (b) Repeat the calculation for a truck that has a gas mileage of 5 mi/gal.
3.108 ![]() Section 2.9 introduced the idea of structural isomerism, with 1-propanol and 2-propanol as examples. Determine which of these properties would distinguish these two substances: (a) boiling point; (b) combustion analysis results; (c) molecular weight; (d) density at a given temperature and pressure. You can check on the properties of these two compounds in Wolfram Alpha (http://www.wolframalpha.com/) or the CRC Handbook of Chemistry and Physics.
Section 2.9 introduced the idea of structural isomerism, with 1-propanol and 2-propanol as examples. Determine which of these properties would distinguish these two substances: (a) boiling point; (b) combustion analysis results; (c) molecular weight; (d) density at a given temperature and pressure. You can check on the properties of these two compounds in Wolfram Alpha (http://www.wolframalpha.com/) or the CRC Handbook of Chemistry and Physics.
3.109 A particular coal contains 2.5% sulfur by mass. When this coal is burned at a power plant, the sulfur is converted into sulfur dioxide gas, which is a pollutant. To reduce sulfur dioxide emissions, calcium oxide (lime) is used. The sulfur dioxide reacts with calcium oxide to form solid calcium sulfite. (a) Wr i te the balanced chemical equation for the reaction. (b) If the coal is burned in a power plant that uses 2000 tons of coal per day, what mass of calcium oxide is required daily to eliminate the sulfur dioxide? (c) How many grams of calcium sulfite are produced daily by this power plant?
3.110 Copper is an excellent electrical conductor widely used in making electric circuits. In producing a printed circuit board for the electronics industry, a layer of copper is laminated on a plastic board. A circuit pattern is then printed on the board using a chemically resistant polymer. The board is then exposed to a chemical bath that reacts with the exposed copper, leaving the desired copper circuit, which has been protected by the overlaying polymer. Finally, a solvent removes the polymer. One reaction used to remove the exposed copper from the circuit board is
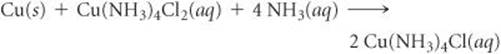
A plant needs to produce 5000 circuit boards, each with a surface area measuring 2.0 in. × 3.0 in. The boards are covered with a 0.65-mm layer of copper. In subsequent processing, 85% of the copper is removed. Copper has a density of 8.96 g/cm3. Calculate the masses of Cu(NH3)4Cl2 and NH3 needed to produce the circuit boards, assuming that the reaction used gives a 97% yield.
3.111 Hydrogen cyanide, HCN, is a poisonous gas. The lethal dose is approximately 300 mg HCN per kilogram of air when inhaled. (a) Calculate the amount of HCN that gives the lethal dose in a small laboratory room measuring 12 × 15 × 8.0 ft. The density of air at 26 °C is 0.00118 g/cm3. (b) If the HCN is formed by reaction of NaCN with an acid such as H2SO4, what mass of NaCN gives the lethal dose in the room?
2 NaCN(s) + H2SO4(aq) → Na2SO4(aq) + 2 HCN(g)
(c) HCN forms when synthetic fibers containing Orlon® or Acrilan® burn. Acrilan® has an empirical formula of CH2CHCN, so HCN is 50.9% of the formula by mass. A rug measures 12 × 15 ft and contains 30 oz of Acrilan® fibers per square yard of carpet. If the rug burns, will a lethal dose of HCN be generated in the room? Assume that the yield of HCN from the fibers is 20% and that the carpet is 50% consumed.
3.112 The source of oxygen that drives the internal combustion engine in an automobile is air. Air is a mixture of gases, principally N2 (˜79%) and O2 (˜20%). In the cylinder of an automobile engine, nitrogen can react with oxygen to produce nitric oxide gas, NO. As NO is emitted from the tailpipe of the car, it can react with more oxygen to produce nitrogen dioxide gas. (a) Write balanced chemical equations for both reactions. (b) Both nitric oxide and nitrogen dioxide are pollutants that can lead to acid rain and global warming; collectively, they are called “NOx” gases. In 2007, the United States emitted an estimated 22 million tons of nitrogen dioxide into the atmosphere. How many grams of nitrogen dioxide is this? (c) The production of NOx gases is an unwanted side reaction of the main engine combustion process that turns octane, C8H18, into CO2 and water. If 85% of the oxygen in an engine is used to combust octane and the remainder used to produce nitrogen dioxide, calculate how many grams of nitrogen dioxide would be produced during the combustion of 500 grams of octane.