CHEMISTRY THE CENTRAL SCIENCE
4 REACTIONS IN AQUEOUS SOLUTION

CAVERNS SUCH AS THIS FORM when underground rivers erode limestone bedrock through to the surface. These caverns, called cenotes, are but one of many ways in which chemical reactions that occur in water shape our planet and impact our lives.
WHAT'S HEAD
4.1 GENERAL PROPERTIES OF AQUEOUS SOLUTIONS
We begin by examining whether substances dissolved in water exist as ions, molecules, or a mixture of the two.
4.2 PRECIPITATION REACTIONS
We identify reactions in which soluble reactants yield an insoluble product.
4.3 ACIDS, BASES, AND NEUTRALIZATION REACTIONS
We explore reactions in which protons, H+ ions, are transferred from one reactant to another.
4.4 OXIDATION-REDUCTION REACTIONS
We examine reactions in which electrons are transferred from one reactant to another.
4.5 CONCENTRATIONS OF SOLUTIONS
We learn how the amount of a compound dissolved in a given volume of a solution can be expressed as a concentration. Concentration can be defined in a number of ways, the most commonly used being moles of compound per liter of solution (molarity).
4.6 SOLUTION STOICHIOMETRY AND CHEMICAL ANALYSIS
We see how the concepts of stoichiometry and concentration can be used to calculate amounts or concentrations of substances in solution through a process called titration.
WATER COVERS NEARLY TWO-THIRDS of our planet, and this simple substance has been the key to much of Earth's evolutionary history. Life almost certainly originated in water, and the need for water by all forms of life has helped determine diverse biological structures.
Given the importance of water for life, it should come as no surprise that development of civilizations has been closely tied to reliable sources of fresh water. During the first millennium, Mayan civilization was one of the most advanced on Earth. Mayan city-states covered much of the Yucatan peninsula in what is now easternmost Mexico. The northern half of the peninsula is a flat shelf of land composed largely of carbonate rocks, such as limestone. Because there are no rivers and very few lakes, the Mayans depended on sinkholes called cenotes, for their fresh water (![]() FIGURE 4.1).
FIGURE 4.1).
Chemical reactions that occur in water are responsible for creation of cenotes. When carbon dioxide, CO2, dissolves in water, the resulting solution is slightly acidic and reacts with CaCO3 in the limestone:
![]()
A solution in which water is the dissolving medium is called an aqueous solution. In this chapter we examine chemical reactions that take place in aqueous solutions. In addition, we extend the concepts of stoichiometry learned in Chapter 3 by considering how solution concentrations are expressed and used.
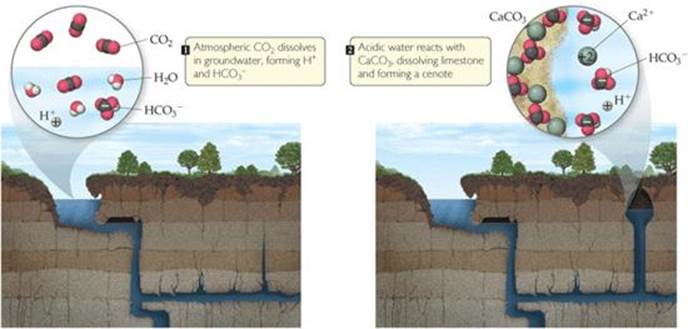
![]() FIGURE 4.1 Cenote formation.
FIGURE 4.1 Cenote formation.
4.1 GENERAL PROPERTIES OF AQUEOUS SOLUTIONS
A solution is a homogeneous mixture of two or more substances. ![]() (Section 1.2) The substance present in the greatest quantity is usually called the solvent, and the other substances are called solutes; they are said to be dissolved in the solvent. When a small amount of sodium chloride (NaCl) is dissolved in a large quantity of water, for example, water is the solvent and sodium chloride is the solute.
(Section 1.2) The substance present in the greatest quantity is usually called the solvent, and the other substances are called solutes; they are said to be dissolved in the solvent. When a small amount of sodium chloride (NaCl) is dissolved in a large quantity of water, for example, water is the solvent and sodium chloride is the solute.
Electrolytic Properties
At a young age we learn not to bring electrical devices into the bathtub so as not to electrocute ourselves. That's a useful lesson because most of the water you encounter in daily life is electrically conducting. Pure water, however, is a very poor conductor of electricity. The conductivity of bathwater originates from the substances dissolved in the water, not from the water itself.
Not all substances that dissolve in water make the resulting solution conducting. Imagine preparing two aqueous solutions—one by dissolving a teaspoon of table salt (sodium chloride) in a cup of water and the other by dissolving a teaspoon of table sugar (sucrose) in a cup of water (![]() FIGURE 4.2). Both solutions are clear and colorless, but they possess very different electrical conductivities: the salt solution is a good conductor of electricity, whereas the sugar solution is not.
FIGURE 4.2). Both solutions are clear and colorless, but they possess very different electrical conductivities: the salt solution is a good conductor of electricity, whereas the sugar solution is not.
In order for the bulb in the device of Figure 4.2 to light up, there must be a current (that is, a flow of electrically charged particles) between two electrodes immersed in the solution. The conductivity of pure water is not sufficient to complete the electrical circuit and light the bulb. The situation changes when ions are present in solution because the ions carry electrical charge from one electrode to the other, completing the circuit. Thus, the conductivity of NaCl solutions indicates the presence of ions. The lack of conductivity of sucrose solutions indicates the absence of ions. When NaCl dissolves in water, the solution contains Na+ and Cl− ions, each surrounded by water molecules. When sucrose (C12H22O11) dissolves in water, the solution contains only neutral sucrose molecules surrounded by water molecules.

![]() FIGURE 4.2 Electrical conductivities of water and two aqueous solutions.
FIGURE 4.2 Electrical conductivities of water and two aqueous solutions.
A substance (such as NaCl) whose aqueous solutions contain ions is called an electrolyte. A substance (such as C12H22O11) that does not form ions in solution is called a nonelectrolyte. The different classifications of NaCl and C12H22O11 arise largely because NaCl is ionic, whereas C12H22O11 is molecular.
Ionic Compounds in Water
Recall from Figure 2.21 that solid NaCl consists of an orderly arrangement of Na+ and Cl− ions. When NaCl dissolves in water, each ion separates from the solid structure and disperses throughout the solution [![]() FIGURE 4.3 (a)]. The ionic solid dissociates into its component ions as it dissolves.
FIGURE 4.3 (a)]. The ionic solid dissociates into its component ions as it dissolves.
![]() GO FIGURE
GO FIGURE
Which solution, NaCl(aq) or CH3OH(aq), conducts electricity?
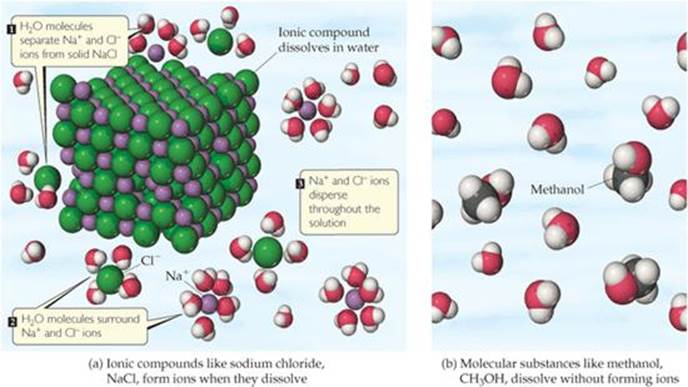
![]() FIGURE 4.3 Dissolution in water. (a) When an ionic compound, such as sodium chloride, NaCl, dissolves in water, H2O molecules separate, surround, and uniformly disperse the ions into the liquid. (b) Molecular substances that dissolve in water, such as methanol, CH3OH, usually do so without forming ions. We can think of this as a simple mixing of two molecular species. In both (a) and (b) the water molecules have been moved apart so that the solute particles can be seen clearly.
FIGURE 4.3 Dissolution in water. (a) When an ionic compound, such as sodium chloride, NaCl, dissolves in water, H2O molecules separate, surround, and uniformly disperse the ions into the liquid. (b) Molecular substances that dissolve in water, such as methanol, CH3OH, usually do so without forming ions. We can think of this as a simple mixing of two molecular species. In both (a) and (b) the water molecules have been moved apart so that the solute particles can be seen clearly.
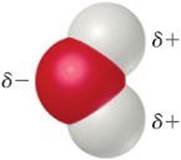
Water is a very effective solvent for ionic compounds. Although H2O is an electrically neutral molecule, the O atom is rich in electrons and has a partial negative charge, denoted by δ− Each H atom has a partial positive charge, denoted by δ+ Cations are attracted by the negative end of H2O, and anions are attracted by the positive end.
As an ionic compound dissolves, the ions become surrounded by H2O molecules, as shown in Figure 4.3(a). The ions are said to be solvated. In chemical equations, we denote solvated ions by writing them as Na+ (aq) and Cl−(aq), where aq is an abbreviation for “aqueous.” ![]() (Section 3.1) Solvation helps stabilize the ions in solution and prevents cations and anions from recombining. Furthermore, because the ions and their shells of surrounding water molecules are free to move about, the ions become dispersed uniformly throughout the solution.
(Section 3.1) Solvation helps stabilize the ions in solution and prevents cations and anions from recombining. Furthermore, because the ions and their shells of surrounding water molecules are free to move about, the ions become dispersed uniformly throughout the solution.
We can usually predict the nature of the ions in a solution of an ionic compound from the chemical name of the substance. Sodium sulfate (Na2SO4), for example, dissociates into sodium ions (Na+) and sulfate ions (SO42−). You must remember the formulas and charges of common ions (Tables 2.4 and 2.5) to understand the forms in which ionic compounds exist in aqueous solution.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What dissolved species are present in a solution of
a. KCN,
b. NaClO4?
Molecular Compounds in Water
When a molecular compound dissolves in water, the solution usually consists of intact molecules dispersed throughout the solution. Consequently, most molecular compounds are nonelectrolytes. As we have seen, table sugar (sucrose) is a nonelectrolyte. As another example, a solution of methanol (CH3OH) in water consists entirely of CH3OH molecules dispersed in the water [Figure 4.3(b)].
A few molecular substances have aqueous solutions that contain ions. Acids are the most important of these solutions. For example, when HCl(g) dissolves in water to form hydrochloric acid, HCl(aq), it ionizes; that is, it dissociates into H+ (aq) and Cl−(aq) ions.
Strong and Weak Electrolytes
Electrolytes differ in the extent to which they conduct electricity. Strong electrolytes are those solutes that exist in solution completely or nearly completely as ions. Essentially all water-soluble ionic compounds (such as NaCl) and a few molecular compounds (such as HCl) are strong electrolytes. Weak electrolytes are those solutes that exist in solution mostly in the form of neutral molecules with only a small fraction in the form of ions. For example, in a solution of acetic acid (CH3COOH) most of the solute is present as CH3COOH(aq) molecules. Only a small fraction (about 1%) of the CH3COOH has dissociated into H+(aq) and CH3COO−(aq) ions.*
We must be careful not to confuse the extent to which an electrolyte dissolves (its solubility) with whether it is strong or weak. For example, CH3COOH is extremely soluble in water but is a weak electrolyte. Ca(OH)2, on the other hand, is not very soluble in water, but the amount that does dissolve dissociates almost completely. Thus, Ca(OH)2 is a strong electrolyte.
When a weak electrolyte, such as acetic acid, ionizes in solution, we write the reaction in the form
![]()
The half-arrows pointing in opposite directions mean that the reaction is significant in both directions. At any given moment some CH3COOH molecules are ionizing to form H+ and CH3COO− ions but H+ and CH3COO− ions are recombining to form CH3COOH. The balance between these opposing processes determines the relative numbers of ions and neutral molecules. This balance produces a state of chemical equilibrium in which the relative numbers of each type of ion or molecule in the reaction are constant over time. Chemists use half-arrows pointing in opposite directions to represent the ionization of weak electrolytes and a single arrow to represent the ionization of strong electrolytes. Because HCl is a strong electrolyte, we write the equation for the ionization of HCl as
![]()
The absence of a left-pointing arrow indicates that the H+ and Cl− ions have no tendency to recombine to form HCl molecules.
In the following sections we will look at how a compound's composition lets us predict whether it is a strong electrolyte, weak electrolyte, or nonelectrolyte. For the moment, you need only to remember that water-soluble ionic compounds are strong electrolytes. Ionic compounds can usually be identified by the presence of both metals and nonmetals [for example, NaCl, FeSO4, and Al(NO3)3]. Ionic compounds containing the ammonium ion, NH4 [for example, NH4Br and (NH4)2CO3], are exceptions to this rule of thumb.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Which solute will cause the lightbulb in Figure 4.2 to glow most brightly, CH3OH, NaOH, or CH3COOH?
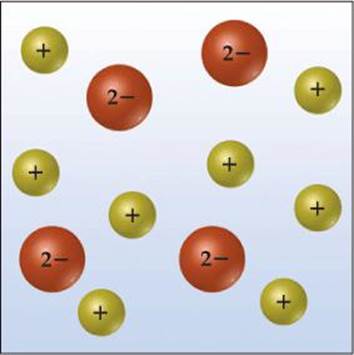
SAMPLE EXERCISE 4.1 Relating Relative Numbers of Anions and Cations to Chemical Formulas
The accompanying diagram represents an aqueous solution of either MgCl2, KCl, or K2SO4. Which solution does the drawing best represent?
SOLUTION
Analyze We are asked to associate the charged spheres in the diagram with ions present in a solution of an ionic substance.
Plan We examine each ionic substance given to determine the relative numbers and charges of its ions. We then correlate these ionic species with the ones shown in the diagram.
Solve The diagram shows twice as many cations as anions, consistent with the formulation K2SO4.
Check Notice that the net charge in the diagram is zero, as it must be if it is to represent an ionic substance.
PRACTICE EXERCISE
If you were to draw diagrams representing aqueous solutions of (a) NiSO4, (b) Ca(NO3)2, (c) Na3PO4, (d) Al2(SO4)3, how many anions would you show if each diagram contained six cations?
Answers: (a) 6, (b) 12, (c) 2, (d) 9