CHEMISTRY THE CENTRAL SCIENCE
4 REACTIONS IN AQUEOUS SOLUTION
4.2 PRECIPITATION REACTIONS
![]() FIGURE 4.4 shows two clear solutions being mixed. One solution contains potassium iodide, KI, dissolved in water and the other contains lead nitrate, Pb(NO3)2, dissolved in water. The reaction between these two solutes produces a water-insoluble yellow solid. Reactions that result in the formation of an insoluble product are called precipitation reactions. A precipitate is an insoluble solid formed by a reaction in solution. In Figure 4.4 the precipitate is lead iodide (PbI2), a compound that has a very low solubility in water:
FIGURE 4.4 shows two clear solutions being mixed. One solution contains potassium iodide, KI, dissolved in water and the other contains lead nitrate, Pb(NO3)2, dissolved in water. The reaction between these two solutes produces a water-insoluble yellow solid. Reactions that result in the formation of an insoluble product are called precipitation reactions. A precipitate is an insoluble solid formed by a reaction in solution. In Figure 4.4 the precipitate is lead iodide (PbI2), a compound that has a very low solubility in water:
![]()
The other product of this reaction, potassium nitrate (KNO3), remains in solution.
![]() GO FIGURE
GO FIGURE
Which ions remain in solution after PbI2 precipitation is complete?
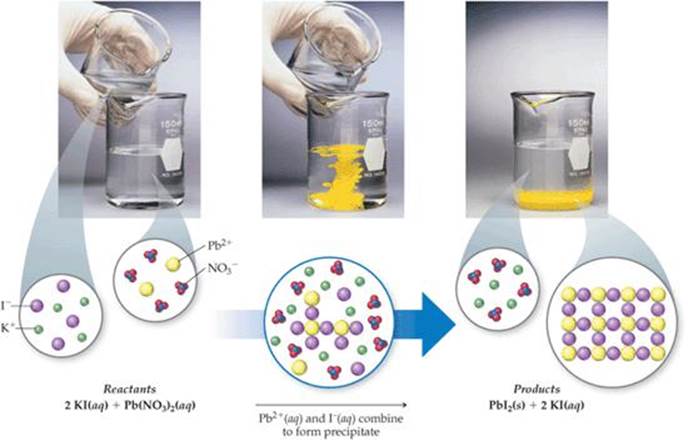
![]() FIGURE 4.4 A precipitation reaction.
FIGURE 4.4 A precipitation reaction.
Precipitation reactions occur when pairs of oppositely charged ions attract each other so strongly that they form an insoluble ionic solid. To predict whether certain combinations of ions form insoluble compounds, we must consider some guidelines concerning the solubilities of common ionic compounds.
Solubility Guidelines for Ionic Compounds
The solubility of a substance at a given temperature is the amount of the substance that can be dissolved in a given quantity of solvent at the given temperature. In our discussions, any substance with a solubility less than 0.01 mol/L will be referred to as insoluble. In those cases the attraction between the oppositely charged ions in the solid is too great for the water molecules to separate the ions to any significant extent; the substance remains largely undissolved.
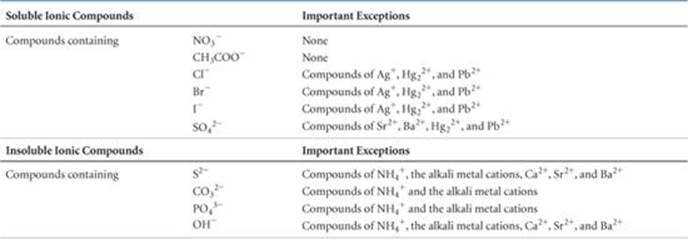
Unfortunately, there are no rules based on simple physical properties such as ionic charge to guide us in predicting whether a particular ionic compound will be soluble. Experimental observations, however, have led to guidelines for predicting solubility for ionic compounds. For example, experiments show that all common ionic compounds that contain the nitrate anion, NO3−, are soluble in water. ![]() TABLE 4.1 summarizes the solubility guidelines for common ionic compounds. The table is organized according to the anion in the compound, but it also reveals many important facts about cations. Note that all common ionic compounds of the alkali metal ions (group 1A of the periodic table) and of the ammonium ion (NH4+) are soluble in water.
TABLE 4.1 summarizes the solubility guidelines for common ionic compounds. The table is organized according to the anion in the compound, but it also reveals many important facts about cations. Note that all common ionic compounds of the alkali metal ions (group 1A of the periodic table) and of the ammonium ion (NH4+) are soluble in water.
TABLE 4.1 • Solubility Guidelines for Common Ionic Compounds in Water
SAMPLE EXERCISE 4.2 Using Solubility Rules
Classify these ionic compounds as soluble or insoluble in water: (a) sodium carbonate, Na2CO3, (b) lead sulfate, PbSO4.
SOLUTION
Analyze We are given the names and formulas of two ionic compounds and asked to predict whether they are soluble or insoluble in water.
Plan We can use Table 4.1 to answer the question. Thus, we need to focus on the anion in each compound because the table is organized by anions.
Solve
(a) According to Table 4.1, most carbonates are insoluble. But carbonates of the alkali metal cations (such as sodium ion) are an exception to this rule and are soluble. Thus, Na2CO3 is soluble in water.
(b) Table 4.1 indicates that although most sulfates are water soluble, the sulfate of Pb2+ is an exception. Thus, PbSO4 is insoluble in water.
PRACTICE EXERCISE
Classify the following compounds as soluble or insoluble in water: (a) cobalt(II) hydroxide, (b) barium nitrate, (c) ammonium phosphate.
Answers: (a) insoluble, (b) soluble, (c) soluble
To predict whether a precipitate forms when we mix aqueous solutions of two strong electrolytes, we must (1) note the ions present in the reactants, (2) consider the possible cation-anion combinations, and (3) use Table 4.1 to determine if any of these combinations is insoluble. For example, will a precipitate form when solutions of Mg(NO3)2 and NaOH are mixed? Both substances are soluble ionic compounds and strong electrolytes. Mixing the solutions first produces a solution containing Mg2+, NO3−, Na+, and OH− ions. Will either cation interact with either anion to form an insoluble compound? Knowing from Table 4.1 that Mg(NO3)2 and NaOH are both soluble in water, our only possibilities are Mg2+ with OH− and Na+ with NO3−. From Table 4.1 we see that hydroxides are generally insoluble. Because Mg2+ is not an exception, Mg(OH)2 is insoluble and thus forms a precipitate. NaNO3, however, is soluble, so Na+ and NO3− remain in solution. The balanced equation for the precipitation reaction is
![]()
Exchange (Metathesis) Reactions
Notice in Equation 4.5 that the reactant cations exchange anions—Mg2+ ends up with OH−, and Na+ ends up with NO3−. The chemical formulas of the products are based on the charges of the ions—two OH− ions are needed to give a neutral compound with Mg2+, and one NO3− ion is needed to give a neutral compound with Na+. ![]() (Section 2.7) The equation can be balanced only after the chemical formulas of the products have been determined.
(Section 2.7) The equation can be balanced only after the chemical formulas of the products have been determined.
Reactions in which cations and anions appear to exchange partners conform to the general equation
Example: 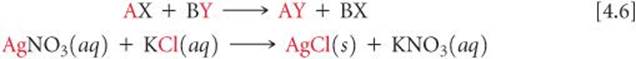
Such reactions are called either exchange reactions or metathesis reactions (meh-TATH-eh-sis, Greek for “to transpose”). Precipitation reactions conform to this pattern, as do many neutralization reactions between acids and bases, as we will see in Section 4.3.
To complete and balance the equation for a metathesis reaction, we follow these steps:
1. Use the chemical formulas of the reactants to determine which ions are present.
2. Write the chemical formulas of the products by combining the cation from one reactant with the anion of the other, using the ionic charges to determine the subscripts in the chemical formulas.
3. Check the water solubilities of the products. For a precipitation reaction to occur, at least one product must be insoluble in water.
4. Balance the equation.
SAMPLE EXERCISE 4.3 Predicting a Metathesis Reaction
(a) Predict the identity of the precipitate that forms when aqueous solutions of BaCl2 and K2SO4 are mixed. (b) Write the balanced chemical equation for the reaction.
SOLUTION
Analyze We are given two ionic reactants and asked to predict the insoluble product that they form.
Plan We need to write the ions present in the reactants and exchange the anions between the two cations. Once we have written the chemical formulas for these products, we can use Table 4.1 to determine which is insoluble in water. Knowing the products also allows us to write the equation for the reaction.
Solve
(a) The reactants contain Ba2+, Cl−, K+, and SO42− ions. Exchanging the anions gives us BaSO4 and KCl. According to Table 4.1, most compounds of SO42− are soluble but those of Ba2+ are not. Thus, BaSO4 is insoluble and will precipitate from solution. KCl is soluble.
(b) From part (a) we know the chemical formulas of the products, BaSO4 and KCl. The balanced equation is
BaCl2(aq) + K2SO4(aq) → BaSO4(s) + 2 KCl(aq)
PRACTICE EXERCISE
(a) What compound precipitates when aqueous solutions of Fe2(SO4)3 and LiOH are mixed?
(b) Write a balanced equation for the reaction. (c) Will a precipitate form when solutions of Ba(NO3)2 and KOH are mixed?
Answers: (a) Fe(OH)3, (b) Fe2(SO4)3(aq) + 6 LiOH(aq) → 2 Fe(OH)3(s) + 3 Li2SO4(aq), (c) no (both possible products, Ba(OH)2 and KNO3, are water soluble)
Ionic Equations
In writing equations for reactions in aqueous solution, it is often useful to indicate whether the dissolved substances are present predominantly as ions or as molecules. Let's reconsider the precipitation reaction between Pb(NO3)2 and 2 KI:
Pb(NO3)2(aq) + 2 KI(aq) → PbI2(s) + 2 KNO3(aq)
An equation written in this fashion, showing the complete chemical formulas of reactants and products, is called a molecular equation because it shows chemical formulas without indicating ionic character. Because Pb(NO3)2, KI, and KNO3 are all water-soluble ionic compounds and therefore strong electrolytes, we can write the equation in a form that indicates which species exist as ions in the solution:
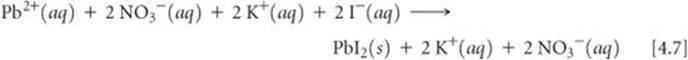
An equation written in this form, with all soluble strong electrolytes shown as ions, is called a complete ionic equation.
Notice that K+(aq) and NO3−(aq) appear on both sides of Equation 4.7. Ions that appear in identical forms on both sides of a complete ionic equation, called spectator ions, play no direct role in the reaction. When spectator ions are omitted from the equation (they cancel out like algebraic quantities), we are left with the net ionic equation, which is one that includes only the ions and molecules directly involved in the reaction:
![]()
Because charge is conserved in reactions, the sum of the ionic charges must be the same on both sides of a balanced net ionic equation. In this case the 2+ charge of the cation and the two 1− charges of the anions add to zero, the charge of the electrically neutral product. If every ion in a complete ionic equation is a spectator, no reaction occurs.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Which ions, if any, are spectator ions in the reaction AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq)?
Net ionic equations illustrate the similarities between various reactions involving electrolytes. For example, Equation 4.8 expresses the essential feature of the precipitation reaction between any strong electrolyte containing Pb2+(aq) and any strong electrolyte containing I−(aq): The ions combine to form a precipitate of PbI2. Thus, a net ionic equation demonstrates that more than one set of reactants can lead to the same net reaction. For example, aqueous solutions of KI and MgI2 share many chemical similarities because both contain I− ions. Either solution when mixed with a Pb(NO3)2 solution produces PbI2(s). The complete ionic equation, on the other hand, identifies the actual reactants that participate in a reaction.
The following steps summarize the procedure for writing net ionic equations:
1. Write a balanced molecular equation for the reaction.
2. Rewrite the equation to show the ions that form in solution when each soluble strong electrolyte dissociates into its ions. Only strong electrolytes dissolved in aqueous solution are written in ionic form.
3. Identify and cancel spectator ions.
SAMPLE EXERCISE 4.4 Writing a Net Ionic Equation
Write the net ionic equation for the precipitation reaction that occurs when aqueous solutions of calcium chloride and sodium carbonate are mixed.
SOLUTION
Analyze Our task is to write a net ionic equation for a precipitation reaction, given the names of the reactants present in solution.
Plan We write the chemical formulas of the reactants and products and then determine which product is insoluble. We then write and balance the molecular equation. Next, we write each soluble strong electrolyte as separated ions to obtain the complete ionic equation. Finally, we eliminate the spectator ions to obtain the net ionic equation.
Solve Calcium chloride is composed of calcium ions, Ca2+, and chloride ions, Cl−; hence, an aqueous solution of the substance is CaCl2(aq). Sodium carbonate is composed of Na+ ions and CO32− ions; hence, an aqueous solution of the compound is Na2CO3(aq). In the molecular equations for precipitation reactions, the anions and cations appear to exchange partners. Thus, we put Ca2+ and CO32− together to give CaCO3 and Na+ and Cl− together to give NaCl. According to the solubility guidelines in Table 4.1, CaCO3 is insoluble and NaCl is soluble. The balanced molecular equation is
CaCl2(aq) + Na2CO3(aq) → CaCO3(s) + 2 NaCl(aq)
In a complete ionic equation, only dissolved strong electrolytes (such as soluble ionic compounds) are written as separate ions. As the (aq) designations remind us, CaCl2, Na2CO3, and NaCl are all dissolved in the solution. Furthermore, they are all strong electrolytes. CaCO3 is an ionic compound, but it is not soluble. We do not write the formula of any insoluble compound as its component ions. Thus, the complete ionic equation is
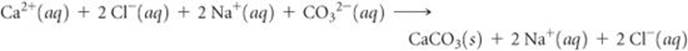
Cl− and Na+ are spectator ions. Canceling them gives the following net ionic equation:
Ca2+(aq) + CO32−(aq) → CaCO3(s)
Check We can check our result by confirming that both the elements and the electric charge are balanced. Each side has one Ca, one C, and three O, and the net charge on each side equals 0.
Comment If none of the ions in an ionic equation is removed from solution or changed in some way, all ions are spectator ions and a reaction does not occur.
PRACTICE EXERCISE
Write the net ionic equation for the precipitation reaction that occurs when aqueous solutions of silver nitrate and potassium phosphate are mixed.
Answer: ![]()