CHEMISTRY THE CENTRAL SCIENCE
4 REACTIONS IN AQUEOUS SOLUTION
4.3 ACIDS, BASES, AND NEUTRALIZATION REACTIONS
Many acids and bases are industrial and household substances (![]() FIGURE 4.5), and some are important components of biological fluids. Hydrochloric acid, for example, is an important industrial chemical and the main constituent of gastric juice in your stomach. Acids and bases are also common electrolytes.
FIGURE 4.5), and some are important components of biological fluids. Hydrochloric acid, for example, is an important industrial chemical and the main constituent of gastric juice in your stomach. Acids and bases are also common electrolytes.

![]() FIGURE 4.5 Vinegar and lemon juice are common household acids. Ammonia and baking soda (sodium bicarbonate) are common household bases.
FIGURE 4.5 Vinegar and lemon juice are common household acids. Ammonia and baking soda (sodium bicarbonate) are common household bases.
Acids
As noted in Section 2.8, acids are substances that ionize in aqueous solution to form hydrogen ions H+ (aq). Because a hydrogen atom consists of a proton and an electron, H+ is simply a proton. Thus, acids are often called proton donors. Molecular models of three common acids are shown in ![]() FIGURE 4.6.
FIGURE 4.6.
Protons in aqueous solution are solvated by water molecules, just as other cations are [Figure 4.3(a)]. In writing chemical equations involving protons in water, therefore, we write H+(aq).
Molecules of different acids ionize to form different numbers of H+ ions. Both HCl and HNO3 are monoprotic acids, yielding one H+ per molecule of acid. Sulfuric acid, H2SO4, is a diprotic acid, one that yields two H+ per molecule of acid. The ionization of H2SO4 and other diprotic acids occurs in two steps:
![]()
![]()
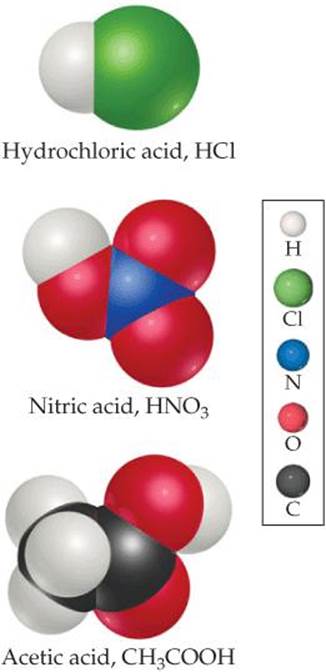
![]() FIGURE 4.6 Molecular models of three common acids.
FIGURE 4.6 Molecular models of three common acids.
Although H2SO4 is a strong electrolyte, only the first ionization (Equation 4.9) is complete. Thus, aqueous solutions of sulfuric acid contain a mixture of H+(aq), HSO4−(aq), and SO42−(aq).
The molecule CH3COOH (acetic acid) that we have mentioned frequently is the primary component in vinegar. Acetic acid has four hydrogens, as Figure 4.6 shows, but only one of them, the H in the COOH group, is ionized in water. The three other hydrogens are bound to carbon and do not break their C—H bonds in water.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
The structural formula of citric acid, a main component of citrus fruits, is
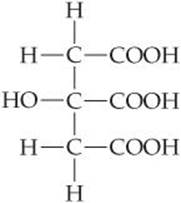
How many H+(aq) can be generated by each citric acid molecule dissolved in water?
Bases
Bases are substances that accept (react with) H+ ions. Bases produce hydroxide ions (OH−) when they dissolve in water. Ionic hydroxide compounds, such as NaOH, KOH, and Ca(OH)2, are among the most common bases. When dissolved in water, they dissociate into ions, introducing OH−ions into the solution.
Compounds that do not contain OH− ions can also be bases. For example, ammonia (NH3) is a common base. When added to water, it accepts an H ion from a water molecule and thereby produces an OH− ion (![]() FIGURE 4.7):
FIGURE 4.7):
![]()
Ammonia is a weak electrolyte because only about 1% of the NH3 forms NH4+ and OH− ions.
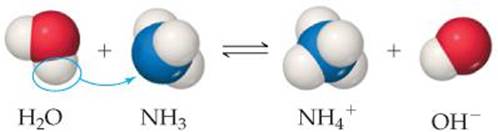
![]() FIGURE 4.7 Hydrogen ion transfer. An H2O molecule acts as a proton donor (acid), and NH3 acts as a proton acceptor (base). Only a fraction of the NH3 molecules react with H2O. Consequently, NH3 is a weak electrolyte.
FIGURE 4.7 Hydrogen ion transfer. An H2O molecule acts as a proton donor (acid), and NH3 acts as a proton acceptor (base). Only a fraction of the NH3 molecules react with H2O. Consequently, NH3 is a weak electrolyte.
Strong and Weak Acids and Bases
Acids and bases that are strong electrolytes (completely ionized in solution) are strong acids and strong bases. Those that are weak electrolytes (partly ionized) are weak acids and weak bases. When reactivity depends only on H+(aq) concentration, strong acids are more reactive than weak acids. The reactivity of an acid, however, can depend on the anion as well as on H+(aq) concentration. For example, hydrofluoric acid (HF) is a weak acid (only partly ionized in aqueous solution), but it is very reactive and vigorously attacks many substances, including glass. This reactivity is due to the combined action of H+(aq) and F−(aq).
![]() TABLE 4.2 lists the strong acids and bases we are most likely to encounter. You need to commit this information to memory in order to correctly identify strong electrolytes and write net ionic equations. The brevity of this list tells us that most acids are weak. (For H2SO4, as we noted earlier, only the first proton completely ionizes.) The only common strong bases are the common soluble metal hydroxides. Most other metal hydroxides are insoluble in water. The most common weak base is NH3, which reacts with water to form OH− ions (Equation 4.11).
TABLE 4.2 lists the strong acids and bases we are most likely to encounter. You need to commit this information to memory in order to correctly identify strong electrolytes and write net ionic equations. The brevity of this list tells us that most acids are weak. (For H2SO4, as we noted earlier, only the first proton completely ionizes.) The only common strong bases are the common soluble metal hydroxides. Most other metal hydroxides are insoluble in water. The most common weak base is NH3, which reacts with water to form OH− ions (Equation 4.11).
TABLE 4.2 • Common Strong Acids and Bases

![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Why isn't Al(OH)3 classified as a strong base?
SAMPLE EXERCISE 4.5 Comparing Acid Strengths
The following diagrams represent aqueous solutions of acids HX, HY, and HZ, with water molecules omitted for clarity. Rank the acids from strongest to weakest.
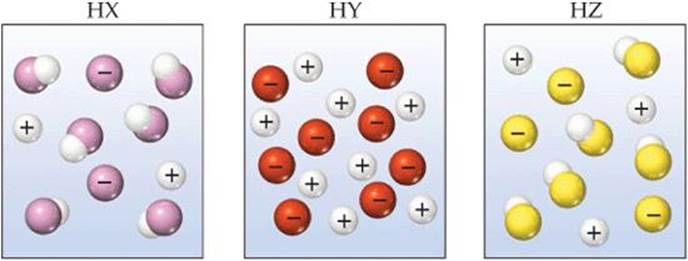
SOLUTION
Analyze We are asked to rank three acids from strongest to weakest, based on schematic drawings of their solutions.
Plan We can determine the relative numbers of uncharged molecular species in the diagrams. The strongest acid is the one with the most H+ ions and fewest undissociated molecules in solution. The weakest acid is the one with the largest number of undissociated molecules.
Solve The order is HY > HZ > HX. HY is a strong acid because it is totally ionized (no HY molecules in solution), whereas both HX and HZ are weak acids, whose solutions consist of a mixture of molecules and ions. Because HZ contains more H+ ions and fewer molecules than HX, it is a stronger acid.
PRACTICE EXERCISE
Imagine a diagram showing 10 Na+ ions and 10 OH− ions. If this solution were mixed with the one pictured above for HY, what species would be present in a diagram that represents the combined solutions after any possible reaction?
Answer: The diagram would show 10 Na+ ions, 2 OH− ions, 8 Y− ions, and 8 H2O molecules.
Identifying Strong and Weak Electrolytes
If we remember the common strong acids and bases (Table 4.2) and also remember that NH3 is a weak base, we can make reasonable predictions about the electrolytic strength of a great number of water-soluble substances. ![]() TABLE 4.3 summarizes our observations about electrolytes. To classify a soluble substance as strong electrolyte, weak electrolyte, or nonelectrolyte, we work our way down and across this table. We first ask whether the substance is ionic or molecular. If it is ionic, it is a strong electrolyte. The second column of Table 4.3 tells us that all ionic compounds are strong electrolytes. If the substance is molecular, we ask whether it is an acid or a base. (It is an acid if it either has H first in the chemical formula or contains a COOH group.) If it is an acid, we use Table 4.2 to determine whether it is a strong or weak electrolyte: All strong acids are strong electrolytes, and all weak acids are weak electrolytes. If an acid is not listed in Table 4.2, it is probably a weak acid and therefore a weak electrolyte.
TABLE 4.3 summarizes our observations about electrolytes. To classify a soluble substance as strong electrolyte, weak electrolyte, or nonelectrolyte, we work our way down and across this table. We first ask whether the substance is ionic or molecular. If it is ionic, it is a strong electrolyte. The second column of Table 4.3 tells us that all ionic compounds are strong electrolytes. If the substance is molecular, we ask whether it is an acid or a base. (It is an acid if it either has H first in the chemical formula or contains a COOH group.) If it is an acid, we use Table 4.2 to determine whether it is a strong or weak electrolyte: All strong acids are strong electrolytes, and all weak acids are weak electrolytes. If an acid is not listed in Table 4.2, it is probably a weak acid and therefore a weak electrolyte.
TABLE 4.3 • Summary of the Electrolytic Behavior of Common
Soluble Ionic and Molecular Compounds
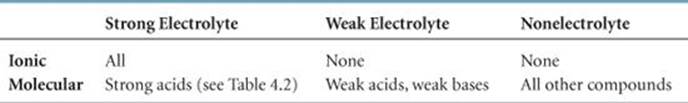
If our substance is a base, we use Table 4.2 to determine whether it is a strong base. NH3 is the only molecular base that we consider in this chapter, and Table 4.3 tells us it is a weak electrolyte. Finally, any molecular substance that we encounter in this chapter that is not an acid or NH3is probably a nonelectrolyte.
SAMPLE EXERCISE 4.6 Identifying Strong, Weak, and Nonelectrolytes
Classify these dissolved substances as strong, weak, or nonelectrolyte: CaCl2, HNO3, C2H5OH (ethanol), HCOOH (formic acid), KOH.
SOLUTION
Analyze We are given several chemical formulas and asked to classify each substance as a strong electrolyte, weak electrolyte, or nonelectrolyte.
Plan The approach we take is outlined in Table 4.3. We can predict whether a substance is ionic or molecular based on its composition. As we saw in Section 2.7, most ionic compounds we encounter in this text are composed of a metal and a nonmetal, whereas most molecular compounds are composed only of nonmetals.
Solve Two compounds fit the criteria for ionic compounds: CaCl2 and KOH. Because Table 4.3 tells us that all ionic compounds are strong electrolytes, that is how we classify these two substances. The three remaining compounds are molecular. Two, HNO3 and HCOOH, are acids. Nitric acid, HNO3, is a common strong acid, as shown in Table 4.2, and therefore is a strong electrolyte. Because most acids are weak acids, our best guess would be that HCOOH is a weak acid (weak electrolyte). This is correct. The remaining molecular compound, C2H5OH, is neither an acid nor a base, so it is a nonelectrolyte.
Comment Although C2H5OH has an OH group, it is not a metal hydroxide and so not a base. Rather, it is a member of a class of organic compounds that have C—OH bonds, which are known as alcohols. ![]() (Section 2.9) Organic compounds containing the COOH group are called carboxylic acids (Chapter 16). Molecules that have this group are weak acids.
(Section 2.9) Organic compounds containing the COOH group are called carboxylic acids (Chapter 16). Molecules that have this group are weak acids.
PRACTICE EXERCISE
Consider solutions in which 0.1 mol of each of the following compounds is dissolved in 1 L of water: Ca(NO3)2 (calcium nitrate), C6H12O6 (glucose), NaCH3COO (sodium acetate), and CH3COOH (acetic acid). Rank the solutions in order of increasing electrical conductivity, based on the fact that the greater the number of ions in solution, the greater the conductivity.
Answers: C6H12O6 (nonelectrolyte) < CH3COOH (weak electrolyte, existing mainly in the form of molecules with few ions) < NaCH3COO (strong electrolyte that provides two ions, Na+ and CH3COO−) < Ca(NO3)2 (strong electrolyte that provides three ions, Ca2+ and 2 NO3−)
Neutralization Reactions and Salts
The properties of acidic solutions are quite different from those of basic solutions. Acids have a sour taste, whereas bases have a bitter taste.* Acids change the colors of certain dyes in a way that differs from the way bases affect the same dyes. This is the principle behind the indicator known as litmus paper (![]() FIGURE 4.8). In addition, acidic and basic solutions differ in chemical properties in several other important ways that we explore in this chapter and in later chapters.
FIGURE 4.8). In addition, acidic and basic solutions differ in chemical properties in several other important ways that we explore in this chapter and in later chapters.
When a solution of an acid and a solution of a base are mixed, a neutralization reaction occurs. The products of the reaction have none of the characteristic properties of either the acidic solution or the basic solution. For example, when hydrochloric acid is mixed with a solution of sodium hydroxide, the reaction is
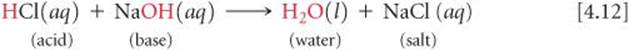

![]() FIGURE 4.8 Litmus paper. Litmus paper is coated with dyes that change color in response to exposure to either acids or bases.
FIGURE 4.8 Litmus paper. Litmus paper is coated with dyes that change color in response to exposure to either acids or bases.
Water and table salt, NaCl, are the products of the reaction. By analogy to this reaction, the term salt has come to mean any ionic compound whose cation comes from a base (for example, Na+ from NaOH) and whose anion comes from an acid (for example, Cl−from HCl). In general, a neutralization reaction between an acid and a metal hydroxide produces water and a salt.
Because HCl, NaOH, and NaCl are all water-soluble strong electrolytes, the complete ionic equation associated with Equation 4.12 is
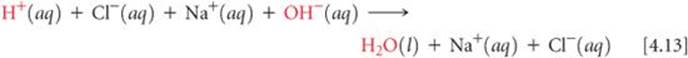
Therefore, the net ionic equation is
![]()
Equation 4.14 summarizes the main feature of the neutralization reaction between any strong acid and any strong base: H+(aq) and OH−(aq) ions combine to form H2O.
![]() FIGURE 4.9 shows the neutralization reaction between hydrochloric acid and the water-insoluble base Mg(OH)2:
FIGURE 4.9 shows the neutralization reaction between hydrochloric acid and the water-insoluble base Mg(OH)2:
Molecular equation:
![]()
Net ionic equation:
![]()
Notice that the OH− ions (this time in a solid reactant) and H+ ions combine to form H2O. Because the ions exchange partners, neutralization reactions between acids and metal hydroxides are metathesis reactions.
![]() GO FIGURE
GO FIGURE
Adding just a few drops of hydrochloric acid would not be sufficient to dissolve all the Mg(OH)2(s). Why not?
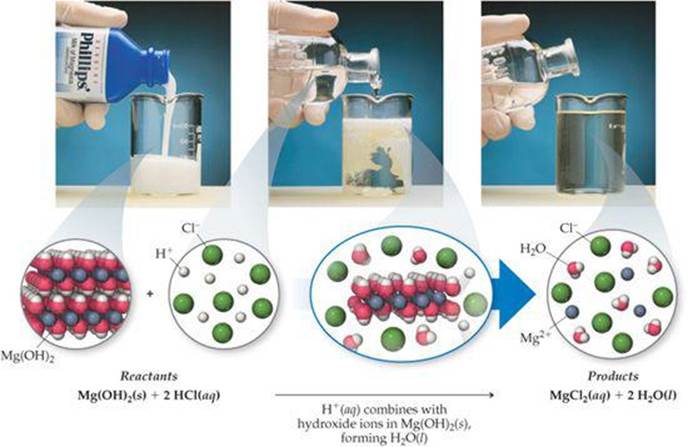
![]() FIGURE 4.9 Neutralization reaction between Mg(OH)2(s) and hydrochloric acid. Milk of magnesia is a suspension of water-insoluble magnesium hydroxide, Mg(OH)2(s), in water. When sufficient hydrochloric acid, HCl(aq), is added a reaction ensues that leads to an aqueous solution containing Mg2+(aq) and Cl−(aq) ions.
FIGURE 4.9 Neutralization reaction between Mg(OH)2(s) and hydrochloric acid. Milk of magnesia is a suspension of water-insoluble magnesium hydroxide, Mg(OH)2(s), in water. When sufficient hydrochloric acid, HCl(aq), is added a reaction ensues that leads to an aqueous solution containing Mg2+(aq) and Cl−(aq) ions.
SAMPLE EXERCISE 4.7 Writing Chemical Equations for a Neutralization Reaction
For the reaction between aqueous solutions of acetic acid (CH3COOH) and barium hydroxide, Ba(OH)2, write (a) the balanced molecular equation, (b) the complete ionic equation, (c) the net ionic equation.
SOLUTION
Analyze We are given the chemical formulas for an acid and a base and asked to write a balanced molecular equation, a complete ionic equation, and a net ionic equation for their neutralization reaction.
Plan As Equation 4.12 and the italicized statement that follows it indicate, neutralization reactions form two products, H2O and a salt. We examine the cation of the base and the anion of the acid to determine the composition of the salt.
Solve
(a) The salt contains the cation of the base (Ba2+) and the anion of the acid (CH3COO−). Thus, the salt formula is Ba(CH3COO)2. According to Table 4.1, this compound is soluble in water. The unbalanced molecular equation for the neutralization reaction is
CH3COOH(aq) + Ba(OH)2(aq) → H2O(l) + Ba(CH3COO)2(aq)
To balance this equation, we must provide two molecules of CH3COOH to furnish the two CH3COO− ions and to supply the two H+ ions needed to combine with the two OH− ions of the base. The balanced molecular equation is
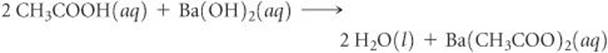
(b) To write the complete ionic equation, we identify the strong electrolytes and break them into ions. In this case Ba(OH)2 and Ba(CH3COO)2 are both water-soluble ionic compounds and hence strong electrolytes. Thus, the complete ionic equation is
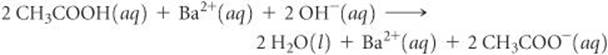
(c) Eliminating the spectator ion, Ba2+, and simplifying coefficients gives the net ionic equation:
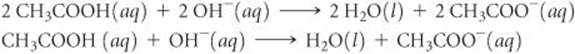
Check We can determine whether the molecular equation is balanced by counting the number of atoms of each kind on both sides of the arrow (10 H, 6 O, 4 C, and 1 Ba on each side). However, it is often easier to check equations by counting groups: There are 2 CH3COO groups, as well as 1 Ba, and 4 additional H atoms and 2 additional O atoms on each side of the equation. The net ionic equation checks out because the numbers of each kind of element and the net charge are the same on both sides of the equation.
PRACTICE EXERCISE
For the reaction of phosphorous acid (H3PO3) and potassium hydroxide (KOH), write (a) the balanced molecular equation and (b) the net ionic equation.
Answers: (a) H3PO3(aq) + 3 KOH(aq) → 3 H2O(l) + K3PO3(aq), (b) H3PO3(aq) + 3 OH (aq) → 3 H2O(l) + PO33−(aq). (H3PO3 is a weak acid and therefore a weak electrolyte, whereas KOH, a strong base, and K3PO3, an ionic compound, are strong electrolytes.)
Neutralization Reactions with Gas Formation
Many bases besides OH− react with H+ to form molecular compounds. Two of these that you might encounter in the laboratory are the sulfide ion and the carbonate ion. Both of these anions react with acids to form gases that have low solubilities in water. Hydrogen sulfide (H2S), the substance that gives rotten eggs their foul odor, forms when an acid such as HCl(aq) reacts with a metal sulfide such as Na2S:
Molecular equation:
![]()
Net ionic equation:
![]()
Carbonates and bicarbonates react with acids to form CO2(g). Reaction of CO32− or HCO3− with an acid first gives carbonic acid (H2CO3). For example, when hydrochloric acid is added to sodium bicarbonate, the reaction is
![]()
Carbonic acid is unstable. If present in solution in sufficient concentrations, it decomposes to H2O and CO2, which escapes from the solution as a gas:
![]()
The overall reaction is summarized by the equations
Molecular equation:
![]()
Net ionic equation:
![]()
Both NaHCO3(s) and Na2CO3(s) are used as neutralizers in acid spills, either salt is added until the fizzing caused by CO2(g) formation stops. Sometimes sodium bicarbonate is used as an antacid to soothe an upset stomach. In that case the HCO3− reacts with stomach acid to form CO2(g).
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
By analogy to examples given in the text, predict what gas forms when Na2SO3(s) reacts with HCl(aq).
 CHEMISTRY PUT TO WORK
CHEMISTRY PUT TO WORK
Antacids
Your stomach secretes acids to help digest foods. These acids, which include hydrochloric acid, contain about 0.1 mol of H+ per liter of solution. The stomach and digestive tract are normally protected from the corrosive effects of stomach acid by a mucosal lining. Holes can develop in this lining, however, allowing the acid to attack the underlying tissue, causing painful damage. These holes, known as ulcers, can be caused by the secretion of excess acids or by a weakness in the digestive lining. Studies indicate, however, that many ulcers are caused by bacterial infection. Between 10 and 20% of Americans suffer from ulcers at some point in their lives. Many others experience occasional indigestion or heartburn due to digestive acids entering the esophagus.

![]() FIGURE 4.10 Antacids. These products all serve as acid-neutralizing agents in the stomach.
FIGURE 4.10 Antacids. These products all serve as acid-neutralizing agents in the stomach.
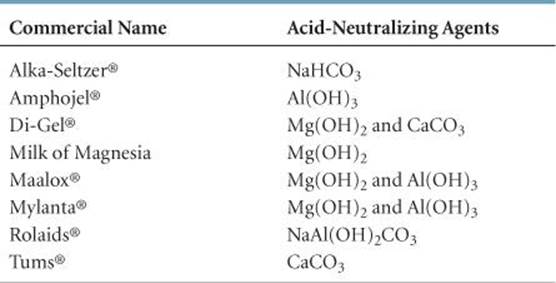
We can address the problem of excess stomach acid in two ways: (1) removing the excess acid or (2) decreasing the production of acid. Substances that remove excess acid are called antacids, whereas those that decrease acid production are called acid inhibitors. ![]() FIGURE 4.10 shows several common over-the-counter antacids, which usually contain hydroxide, carbonate, or bicarbonate ions (
FIGURE 4.10 shows several common over-the-counter antacids, which usually contain hydroxide, carbonate, or bicarbonate ions (![]() TABLE 4.4). Antiulcer drugs, such as Tagamet® and Zantac®, are acid inhibitors. They act on acid-producing cells in the lining of the stomach. Formulations that control acid in this way are now available as over-the-counter drugs.
TABLE 4.4). Antiulcer drugs, such as Tagamet® and Zantac®, are acid inhibitors. They act on acid-producing cells in the lining of the stomach. Formulations that control acid in this way are now available as over-the-counter drugs.
RELATED EXERCISE: 4.95
TABLE 4.4 • Some Common Antacids