CHEMISTRY THE CENTRAL SCIENCE
4 REACTIONS IN AQUEOUS SOLUTION
4.5 CONCENTRATIONS OF SOLUTIONS
Scientists use the term concentration to designate the amount of solute dissolved in a given quantity of solvent or quantity of solution. The greater the amount of solute dissolved in a certain amount of solvent, the more concentrated the resulting solution. In chemistry we often need to express the concentrations of solutions quantitatively.
Molarity
Molarity (symbol M) expresses the concentration of a solution as the number of moles of solute in a liter of solution (soln):
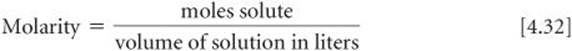
A 1.00 molar solution (written 1.00 M) contains 1.00 mol of solute in every liter of solution. ![]() FIGURE 4.16 shows the preparation of 250.0 mL of a 1.00 M solution of CuSO4. The molarity of the solution is (0.250 mol CuSO4)/(0.250 L soln) = 1.00M.
FIGURE 4.16 shows the preparation of 250.0 mL of a 1.00 M solution of CuSO4. The molarity of the solution is (0.250 mol CuSO4)/(0.250 L soln) = 1.00M.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Which is more concentrated, a solution prepared by dissolving 21.0 g of NaF (0.500 mol) in enough water to make 500 mL of solution or a solution prepared by dissolving 10.5 g (0.250 mol) of NaF in enough water to make 100 mL of solution?

![]() FIGURE 4.16 Preparing 0.250 L of a 1.00 M solution of CuSO4.
FIGURE 4.16 Preparing 0.250 L of a 1.00 M solution of CuSO4.
SAMPLE EXERCISE 4.11 Calculating Molarity
Calculate the molarity of a solution made by dissolving 23.4 g of sodium sulfate (Na2SO4) in enough water to form 125 mL of solution.
SOLUTION
Analyze We are given the number of grams of solute (23.4 g), its chemical formula (Na2SO4), and the volume of the solution (125 mL) and asked to calculate the molarity of the solution.
Plan We can calculate molarity using Equation 4.32. To do so, we must convert the number of grams of solute to moles and the volume of the solution from milliliters to liters.
Solve The number of moles of Na2SO4 is obtained by using its molar mass:
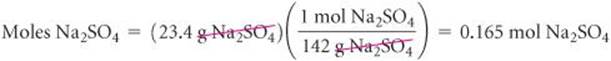
Converting the volume of the solution to liters:
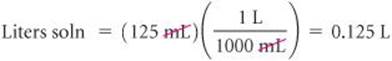
Thus, the molarity is
![]()
Check Because the numerator is only slightly larger than the denominator, it is reasonable for the answer to be a little over 1 M. The units (mol/L) are appropriate for molarity, and three significant figures are appropriate for the answer because each of the initial pieces of data had three significant figures.
PRACTICE EXERCISE
Calculate the molarity of a solution made by dissolving 5.00 g of glucose (C6H12O6) in sufficient water to form exactly 100 mL of solution.
Answer: 0.278 M
Expressing the Concentration of an Electrolyte
When an ionic compound dissolves, the relative concentrations of the ions in the solution depend on the chemical formula of the compound. For example, a 1.0 M solution of NaCl is 1.0 M in Na+ ions and 1.0 M in Cl− ions, and a 1.0 M solution of Na2SO4 is 2.0 M in Na+ ions and 1.0 M in SO42− ions. Thus, the concentration of an electrolyte solution can be specified either in terms of the compound used to make the solution (1.0 M Na2SO4) or in terms of the ions in the solution (2.0 M Na+ and 1.0 M SO42−).
SAMPLE EXERCISE 4.12 Calculating Molar Concentrations of Ions
What is the molar concentration of each ion present in a 0.025 M aqueous solution of calcium nitrate?
SOLUTION:
Analyze We are given the concentration of the ionic compound used to make the solution and asked to determine the concentrations of the ions in the solution.
Plan We can use the subscripts in the chemical formula of the compound to determine the relative ion concentrations.
Solve Calcium nitrate is composed of calcium ions (Ca2+) and nitrate ions (NO3−), so its chemical formula is Ca(NO3)2. Because there are two NO3− ions for each Ca2+ ion, each mole of Ca(NO3)2 that dissolves dissociates into 1 mol of Ca2+ and 2 mol of NO3−. Thus, a solution that is 0.025M in Ca(NO3)2 is 0.025 M in Ca2+ and 2 × 0.025 M = 0.050 M in NO3−:
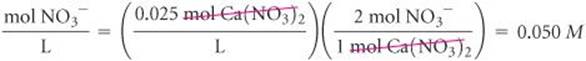
Check The concentration of NO3− ions is twice that of Ca2+ ions, as the subscript 2 after the NO3− in the chemical formula Ca(NO3)2 suggests it should be.
PRACTICE EXERCISE
What is the molar concentration of K+ ions in a 0.015 M solution of potassium carbonate?
Answer: 0.030 M
Interconverting Molarity, Moles, and Volume
If we know any two of the three quantities in Equation 4.32, we can calculate the third. For example, if we know the molarity of an HNO3 solution to be 0.200 M, which means 0.200 mol of HNO3 per liter of solution, we can calculate the number of moles of solute in a given volume, say 2.0 L. Molarity therefore is a conversion factor between volume of solution and moles of solute:
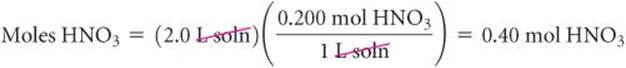
To illustrate the conversion of moles to volume, let's calculate the volume of 0.30 M HNO3 solution required to supply 2.0 mol of HNO3:
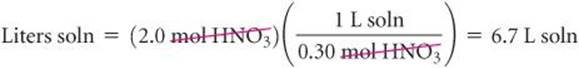
In this case we must use the reciprocal of molarity in the conversion: ![]()
SAMPLE EXERCISE 4.13 Using Molarity to Calculate Grams of Solute
How many grams of Na2SO4 are required to make 0.350 L of 0.500 M Na2SO4?
SOLUTION
Analyze We are given the volume of the solution (0.350 L), its concentration (0.500 M), and the identity of the solute Na2SO4 and asked to calculate the number of grams of the solute in the solution.
Plan We can use the definition of molarity (Equation 4.32) to determine the number of moles of solute, and then convert moles to grams using the molar mass of the solute.
![]()
Solve Calculating the moles of Na2SO4 using the molarity and volume of solution gives

Because each mole of Na2SO4 has a mass of 142 g, the required number of grams of Na2SO4 is
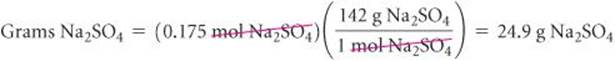
Check The magnitude of the answer, the units, and the number of significant figures are all appropriate.
PRACTICE EXERCISE
(a) How many grams of Na2SO4 are there in 15 mL of 0.50 M Na2SO4? (b) How many milliliters of 0.50 M Na2SO4 solution are needed to provide 0.038 mol of this salt?
Answers: (a) 1.1 g, (b) 76 mL
Dilution
Solutions used routinely in the laboratory are often purchased or prepared in concentrated form (called stock solutions). Solutions of lower concentrations can then be obtained by adding water, a process called dilution.*
Let's see how we can prepare a dilute solution from a concentrated one. Suppose we want to prepare 250.0 mL (that is, 0.2500 L) of 0.100 M CuSO4 solution by diluting a 1.00 M CuSO4 stock solution. The main point to remember is that when solvent is added to a solution, the number of moles of solute remains unchanged:
![]()
Because we know both the volume (250 mL) and the concentration (0.100 mol/L) of the dilute solution, we can calculate the number of moles of CuSO4 it contains:
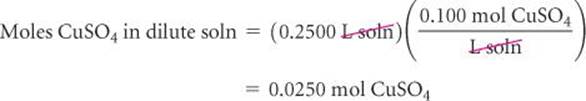
The volume of stock solution needed to provide 0.0250 mol CuSO4 is therefore:

![]() FIGURE 4.17 shows the dilution carried out in the laboratory. Notice that the diluted solution is less intensely colored than the concentrated one.
FIGURE 4.17 shows the dilution carried out in the laboratory. Notice that the diluted solution is less intensely colored than the concentrated one.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
How is the molarity of a 0.50 M KBr solution changed when water is added to double its volume?
In laboratory situations, calculations of this sort are often made with an equation derived by remembering that the number of moles of solute is the same in both the concentrated and dilute solutions and that moles = molarity × liters:
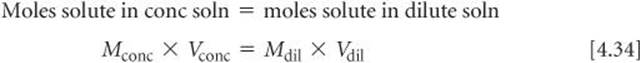
Although we derived Equation 4.34 in terms of liters, any volume unit can be used as long as it is used on both sides of the equation. For example, in the calculation we did for the CuSO4 solution, we have
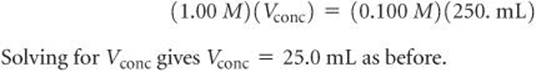
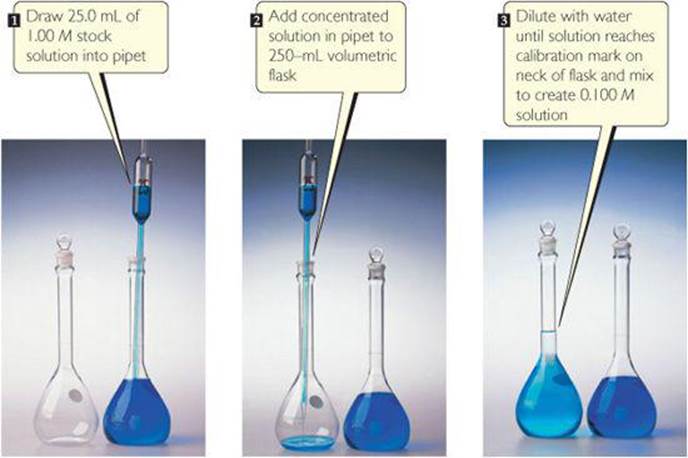
![]() FIGURE 4.17 Preparing 250 mL of 0.100 M CuSO4 by dilution of 1.00 M CuSO4.
FIGURE 4.17 Preparing 250 mL of 0.100 M CuSO4 by dilution of 1.00 M CuSO4.
 CHEMISTRY AND LIFE
CHEMISTRY AND LIFE
DRINKING TOO MUCH WATER CAN KILL YOU
For a long time dehydration was considered a potential danger for people engaged in extended vigorous activity. Thus, athletes were encouraged to drink lots of water while engaged in active sport. The trend toward extensive hydration has spread throughout society, so that today many people carry water bottles everywhere and dutifully keep well hydrated.
In some circumstances, however, drinking too much water is a greater danger than not drinking enough. Excess water consumption can lead to hyponatremia, a condition in which the concentration of sodium ion in the blood is too low. In the past decade at least four marathon runners have died from hyponatremia-related trauma, and dozens more have become seriously ill. For example, a first-time marathoner named Hillary Bellamy, running in the Marine Corps marathon in 2003, collapsed near mile 22 and died the next day. One physician who treated her said that she died from hyponatremia-induced brain swelling, the result of drinking too much water before and during the race.
The normal blood sodium level is 135 to 145 mM (millimolar). When that level drops to 125 mM, dizziness and confusion set in. A concentration below 120 mM can be critical. Dangerously low levels can occur in any active athlete who is sweating out salt (NaCl) at the same time that excessive amounts of NaCl-free water are being drunk to compensate for water loss. The condition affects women more than men because of differences in body composition and patterns of metabolism. Drinking a sport drink that contains some electrolytes helps to prevent hyponatremia.
RELATED EXERCISES: 4.63, 4.64
SAMPLE EXERCISE 4.14 Preparing a Solution by Dilution
How many milliliters of 3.0 M H2SO4 are needed to make 450 mL of 0.10 M H2SO4?
SOLUTION
Analyze We need to dilute a concentrated solution. We are given the molarity of a more concentrated solution (3.0 M) and the volume and molarity of a more dilute one containing the same solute (450 mL of 0.10 M solution). We must calculate the volume of the concentrated solution needed to prepare the dilute solution.
Plan We can calculate the number of moles of solute, H2SO4, in the dilute solution and then calculate the volume of the concentrated solution needed to supply this amount of solute. Alternatively, we can directly apply Equation 4.34. Let's compare the two methods.
Solve Calculating the moles of H2SO4 in the dilute solution:
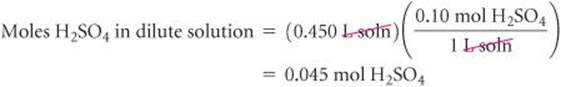
Calculating the volume of the concentrated solution that contains 0.045 mol H2SO4:
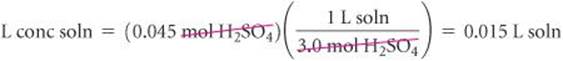
Converting liters to milliliters gives 15 mL.
If we apply Equation 4.34, we get the same result:
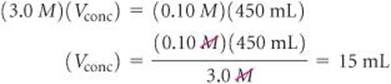
Either way, we see that if we start with 15 mL of 3.0 M H2SO4 and dilute it to a total volume o 450 mL, the desired 0.10 M solution will be obtained.
Check The calculated volume seems reasonable because a small volume of concentrated solution is used to prepare a large volume of dilute solution.
Comment The first approach can also be used to find the final concentration when two solutions of different concentrations are mixed, whereas the second approach, using Equation 4.34, can be used only for diluting a concentrated solution with pure solvent.
PRACTICE EXERCISE
(a) What volume of 2.50 M lead(II) nitrate solution contains 0.0500 mol of Pb2+? (b) How many milliliters of 5.0 M K2Cr2O7 solution must be diluted to prepare 250 mL of 0.10 M solution? (c) If 10.0 mL of a 10.0 M stock solution of NaOH is diluted to 250 mL, what is the con centration of the resulting stock solution?
Answers: (a) 0.0200 L = 20.0 mL, (b) 5.0 mL, (c) 0.40 M