CHEMISTRY THE CENTRAL SCIENCE
4 REACTIONS IN AQUEOUS SOLUTION
4.6 SOLUTION STOICHIOMETRY AND CHEMICAL ANALYSIS
In Chapter 3 we learned that given the chemical equation for a reaction and the amount of one reactant consumed in the reaction, you can calculate the quantities of other reactants and products. In this section we extend this concept to reactions involving solutions.
Recall that the coefficients in a balanced equation give the relative number of moles of reactants and products. ![]() (Section 3.6) To use this information, we must convert the masses of substances involved in a reaction into moles. When dealing with pure substances, as we were inChapter 3, we use molar mass to convert between grams and moles of the substances. This conversion is not valid when working with a solution because both solute and solvent contribute to its mass. However, if we know the solute concentration, we can use molarity and volume to determine the number of moles (moles solute = M × V).
(Section 3.6) To use this information, we must convert the masses of substances involved in a reaction into moles. When dealing with pure substances, as we were inChapter 3, we use molar mass to convert between grams and moles of the substances. This conversion is not valid when working with a solution because both solute and solvent contribute to its mass. However, if we know the solute concentration, we can use molarity and volume to determine the number of moles (moles solute = M × V). ![]() FIGURE 4.18 summarizes this approach to using stoichiometry for the reaction between a pure substance and a solution.
FIGURE 4.18 summarizes this approach to using stoichiometry for the reaction between a pure substance and a solution.
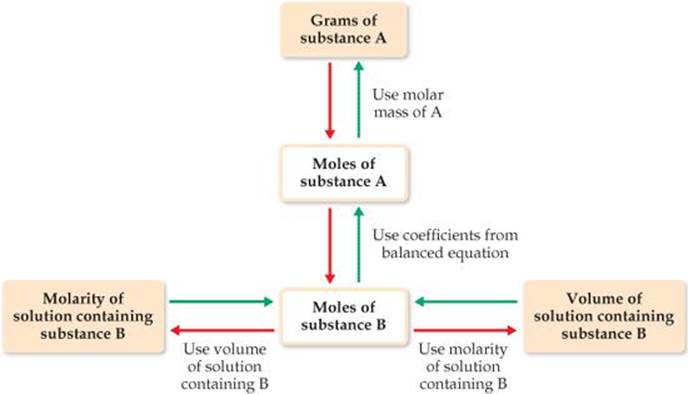
![]() FIGURE 4.18 Procedure for solving stoichiometry problems involving reactions between a pure substance A and a solution containing a known concentration of substance B. Starting from a known mass of substance A, we follow the red arrows to determine either the volume of the solution containing B (if the molarity of B is known) or the molarity of the solution containing B (if the volume of B is known). Starting from either a known volume or known molarity of the solution containing B, we follow the green arrows to determine the mass of substance A.
FIGURE 4.18 Procedure for solving stoichiometry problems involving reactions between a pure substance A and a solution containing a known concentration of substance B. Starting from a known mass of substance A, we follow the red arrows to determine either the volume of the solution containing B (if the molarity of B is known) or the molarity of the solution containing B (if the volume of B is known). Starting from either a known volume or known molarity of the solution containing B, we follow the green arrows to determine the mass of substance A.
SAMPLE EXERCISE 4.15 Using Mass Relations in a Neutralization Reaction
How many grams of Ca(OH)2 are needed to neutralize 25.0 mL of 0.100 M HNO3?
SOLUTION
Analyze The reactants are an acid, HNO3, and a base, Ca(OH)2. The volume and molarity of HNO3 are given, and we are asked how many grams of Ca(OH)2 are needed to neutralize this quantity of HNO3.
Plan Following the steps outlined by the green arrows in Figure 4.18, we use the molarity and volume of the HNO3 solution (substance B in Figure 4.18) to calculate the number of moles of HNO3. We then use the balanced equation to relate moles of HNO3 to moles of Ca(OH)2 (substance A). Finally, we use the molar mass to convert moles to grams of Ca(OH)2:
![]()
Solve The product of the molar concentration of a solution and its volume in liters gives the number of moles of solute:
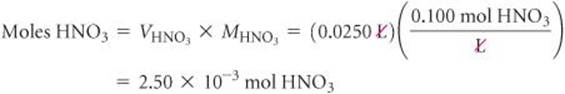
Because this is a neutralization reaction, HNO3 and Ca(OH)2 react to form H2O and the salt containing Ca2+ and NO3−:
2 HNO3(aq) + Ca(OH)2(s) → 2 H2O(l) + Ca(NO3)2(aq)
Thus, 2 mol HNO3![]() mol Ca(OH)2, Therefore,
mol Ca(OH)2, Therefore,
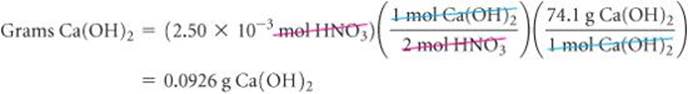
Check The answer is reasonable because a small volume of dilute acid requires only a small amount of base to neutralize it.
PRACTICE EXERCISE
(a) How many grams of NaOH are needed to neutralize 20.0 mL of 0.150 M H2SO4 solution?
(b) How many liters of 0.500 M HCl(aq) are needed to react completely with 0.100 mol of Pb(NO3)2(aq), forming a precipitate of PbCl2(s)?
Answers: (a) 0.240 g, (b) 0.400 L
Titrations
To determine the concentration of a particular solute in a solution, chemists often carry out a titration, which involves combining a solution where the solute concentration is not known with a reagent solution of known concentration, called a standard solution. Just enough standard solution is added to completely react with the solute in the solution of unknown concentration. The point at which stoichiometrically equivalent quantities are brought together is known as the equivalence point.
Titrations can be conducted using neutralization, precipitation, or oxidation-reduction reactions. ![]() FIGURE 4.19 illustrates a typical neutralization titration, one between a HCl solution of unknown concentration and a NaOH solution we know to have a concentration of 0.100 M (our standard solution). To determine the HCl concentration, we first add a specific volume of the HCl solution, 20.0 mL in this example, to a flask. Next a few drops of an acid-base indicator are added. The acid-base indicator is a dye that changes color on passing the equivalence point.* For example, the dye phenolphthalein is colorless in acidic solution but pink in basic solution. The standard solution is then slowly added until the solution turns pink, telling us that the neutralization reaction between HCl and NaOH is complete. The standard solution is added from a buret so that we can accurately determine the added volume of NaOH solution. Knowing the volumes of both solutions and the concentration of the standard solution we can calculate the concentration of the unknown solution as diagrammed in
FIGURE 4.19 illustrates a typical neutralization titration, one between a HCl solution of unknown concentration and a NaOH solution we know to have a concentration of 0.100 M (our standard solution). To determine the HCl concentration, we first add a specific volume of the HCl solution, 20.0 mL in this example, to a flask. Next a few drops of an acid-base indicator are added. The acid-base indicator is a dye that changes color on passing the equivalence point.* For example, the dye phenolphthalein is colorless in acidic solution but pink in basic solution. The standard solution is then slowly added until the solution turns pink, telling us that the neutralization reaction between HCl and NaOH is complete. The standard solution is added from a buret so that we can accurately determine the added volume of NaOH solution. Knowing the volumes of both solutions and the concentration of the standard solution we can calculate the concentration of the unknown solution as diagrammed in ![]() FIGURE 4.20.
FIGURE 4.20.
![]() GO FIGURE
GO FIGURE
How would the volume of standard solution added change if that solution were Ba(OH)2(aq) instead of NaOH(aq)?

![]() FIGURE 4.19 Procedure for titrating an acid against a standard solution of NaOH. The acid-base indicator, phenolphthalein, is colorless in acidic solution but takes on a pink color in basic solution.
FIGURE 4.19 Procedure for titrating an acid against a standard solution of NaOH. The acid-base indicator, phenolphthalein, is colorless in acidic solution but takes on a pink color in basic solution.
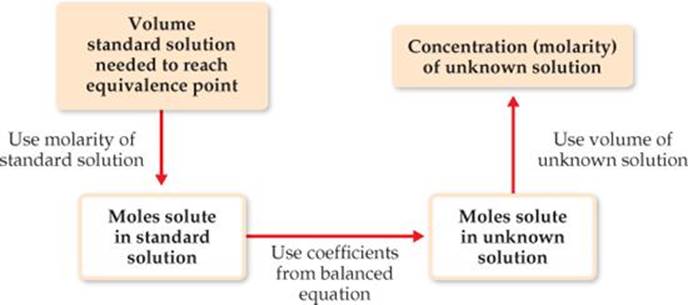
![]() FIGURE 4.20 Procedure for determining the concentration of a solution from titration with a standard solution.
FIGURE 4.20 Procedure for determining the concentration of a solution from titration with a standard solution.
SAMPLE EXERCISE 4.16 Determining Solution Concentration by an Acid–Base Titration
One commercial method used to peel potatoes is to soak them in a NaOH solution for a short time, then remove them and spray off the peel. The NaOH concentration is normally 3 to 6 M, and the solution must be analyzed periodically. In one such analysis, 45.7 mL of 0.500 M H2SO4 is required to neutralize 20.0 mL of NaOH solution. What is the concentration of the NaOH solution?
SOLUTION
Analyze We are given the volume (45.7 mL) and molarity (0.500 M) of an H2SO4 solution (the standard solution) that reacts completely with 20.0 mL of NaOH solution. We are asked to calculate the molarity of the NaOH solution.
Plan Following the steps of Figure 4.20, we use the H2SO4 volume and molarity to calculate the number of moles of H2SO4. Then we can use this quantity and the balanced equation for the reaction to calculate moles of NaOH. Finally, we can use moles of NaOH and the NaOH volume to calculate NaOH molarity.
Solve The number of moles of H2SO4 is the product of the volume and molarity of this solution:
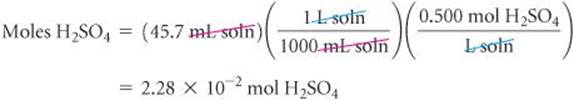
Acids react with metal hydroxides to form water and a salt. Thus, the balanced equation for the neutralization reaction is
H2SO4(aq) + 2 NaOH(aq) → 2 H2O(l) + Na2SO4(aq)
According to the balanced equation, 1 mol H2SO4![]() 2 mol NaOH. Therefore,
2 mol NaOH. Therefore,
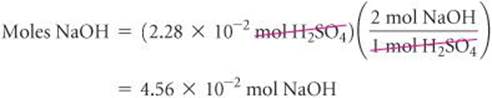
Knowing the number of moles of NaOH in 20.0 mL of solution allows us to calculate the molarity of this solution:
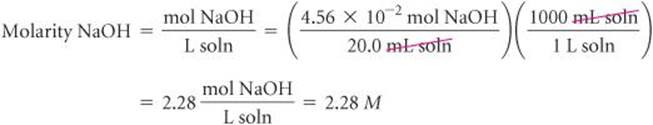
PRACTICE EXERCISE
What is the molarity of an NaOH solution if 48.0 mL neutralizes 35.0 mL of 0.144 M H2SO4?
Answer: 0.210 M
SAMPLE EXERCISE 4.17 Determining the Quantity of Solute by Titration
The quantity of Cl− in a municipal water supply is determined by titrating the sample with Ag+. The precipitation reaction taking place during the titration is
Ag+(aq) + Cl−(aq) → AgCl(s)
The end point in this type of titration is marked by a change in color of a special type of indicator. (a) How many grams of chloride ion are in a sample of the water if 20.2 mL of 0.100 M Ag+ is needed to react with all the chloride in the sample? (b) If the sample has a mass of 10.0 g, what percent Cl− does it contain?
SOLUTION
Analyze We are given the volume (20.2 mL) and molarity (0.100 M) of a solution of Ag+ and the chemical equation for reaction of this ion with Cl−. We are asked to calculate the number of grams of Cl− in the sample and the mass percent of Cl− in the sample.
(a) Plan We can use the procedure outlined by the green arrows in Figure 4.18. We begin by using the volume and molarity of Ag+ to calculate the number of moles of Ag+ used in the titration. We then use the balanced equation to determine the moles of Cl− in the sample and from that the grams of Cl−.
Solve
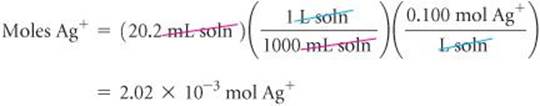
From the balanced equation we see that 1 mol Ag+![]() 1 mol Cl−. Using this information and the molar mass of Cl, we have
1 mol Cl−. Using this information and the molar mass of Cl, we have
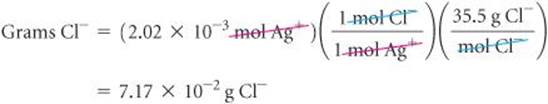
(b) Plan To calculate the percentage of Cl− in the sample, we compare the number of grams of Cl− in the sample, 7.17 × 10−2g, with the original mass of the sample, 10.0 g.
Solve
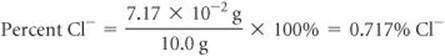
Comment Chloride ion is one of the most common ions in water and sewage. Ocean water contains 1.92% Cl−. Whether water containing Cl− tastes salty depends on the other ions present. If the only accompanying ions are Na+, a salty taste may be detected with as little as 0.03% Cl−.
PRACTICE EXERCISE
A sample of an iron ore is dissolved in acid, and the iron is converted to Fe2+. The sample is then titrated with 47.20 mL of 0.02240 M MnO4− solution. The oxidation-reduction reaction that occurs during titration is
MnO4−(aq) + 5 Fe2+(aq) + 8 H+(aq) → Mn2+(aq) + 5 Fe3+(aq) + 4 H2O(l)
(a) How many moles of MnO4− were added to the solution? (b) How many moles of Fe2+ were in the sample? (c) How many grams of iron were in the sample? (d) If the sample had a mass of 0.8890 g, what is the percentage of iron in the sample?
Answers: (a) 1.057 × 10−3 mol MnO4−, (b) 5.286 × 10−3 mol Fe2+, (c) 0.2952 g, (d) 33.21%
SAMPLE INTEGRATIVE EXERCISE Putting Concepts Together
Note: Integrative exercises require skills from earlier chapters as well as ones from the present chapter.
A sample of 70.5 mg of potassium phosphate is added to 15.0 mL of 0.050 M silver nitrate, resulting in the formation of a precipitate. (a) Write the molecular equation for the reaction. (b) What is the limiting reactant in the reaction? (c) Calculate the theoretical yield, in grams, of the precipitate that forms.
SOLUTION
(a) Potassium phosphate and silver nitrate are both ionic compounds. Potassium phosphate contains K+ and PO43− ions, so its chemical formula is K3PO4. Silver nitrate contains Ag+ and NO3− ions, so its chemical formula is AgNO3. Because both reactants are strong electrolytes, the solution contains K+, PO43−, Ag+, and NO3− ions before the reaction occurs. According to the solubility guidelines in Table 4.1, Ag+ and PO43− form an insoluble compound, so Ag3PO4 will precipitate from the solution. In contrast, K+ and NO3− will remain in solution because KNO3 is water soluble. Thus, the balanced molecular equation for the reaction is
K3PO4(aq) + 3 AgNO3(aq) → Ag3PO4(s) + 3 KNO3(aq)
(b) To determine the limiting reactant, we must examine the number of moles of each reactant. ![]() (Section 3.7) The number of moles of K3PO4 is calculated from the mass of the sample using the molar mass as a conversion factor.
(Section 3.7) The number of moles of K3PO4 is calculated from the mass of the sample using the molar mass as a conversion factor. ![]() (Section 3.4) The molar mass of K3PO4 is 3(39.1) + 31.0 + 4(16.0) = 212.3 g/mol. Converting milligrams to grams and then to moles, we have
(Section 3.4) The molar mass of K3PO4 is 3(39.1) + 31.0 + 4(16.0) = 212.3 g/mol. Converting milligrams to grams and then to moles, we have
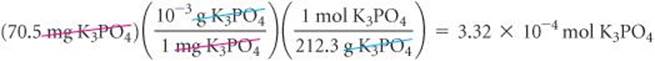
We determine the number of moles of AgNO3 from the volume and molarity of the solution. (Section 4.5) Converting milliliters to liters and then to moles, we have

Comparing the amounts of the two reactants, we find that there are (7.5 × 10−4)/(3.32 × 10−4) = 2.3 times as many moles of AgNO3 as there are moles of K3PO4. According to the balanced equation, however, 1 mol K3PO4 requires 3 mol AgNO3. Thus, there is insufficient AgNO3 to consume the K3PO4, and AgNO3 is the limiting reactant.
(c) The precipitate is Ag3PO4, whose molar mass is 3(107.9) + 31.0 + 4(16.0) = 418.7 g/mol. To calculate the number of grams of Ag3PO4 that could be produced in this reaction (the theoretical yield), we use the number of moles of the limiting reactant, converting mol AgNO3![]() mol Ag3PO4
mol Ag3PO4![]() g Ag3PO4. We use the coefficients in the balanced equation to convert moles of AgNO3 to moles Ag3PO4, and we use the molar mass of Ag3PO4 to convert the number of moles of this substance to grams.
g Ag3PO4. We use the coefficients in the balanced equation to convert moles of AgNO3 to moles Ag3PO4, and we use the molar mass of Ag3PO4 to convert the number of moles of this substance to grams.
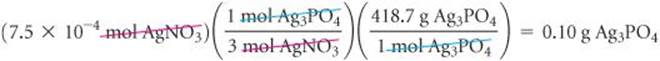
The answer has only two significant figures because the quantity of AgNO3 is given to only two significant figures.