CHEMISTRY THE CENTRAL SCIENCE
8 BASIC CONCEPTS OF CHEMICAL BONDING
8.3 COVALENT BONDING
The vast majority of chemical substances do not have the characteristics of ionic materials. Most of the substances with which we come into daily contact—such as water—tend to be gases, liquids, or solids with low melting points. Many, such as gasoline, vaporize readily. Many are pliable in their solid forms—for example, plastic bags and paraffin.
For the very large class of substances that do not behave like ionic substances, we need a different model for the bonding between atoms. G. N. Lewis reasoned that atoms might acquire a noble-gas electron configuration by sharing electrons with other atoms. A chemical bond formed by sharing a pair of electrons is a covalent bond.
The hydrogen molecule, H2, provides the simplest example of a covalent bond. When two hydrogen atoms are close to each other, the two positively charged nuclei repel each other, the two negatively charged electrons repel each other, and the nuclei and electrons attract each other, as shown in ![]() FIGURE 8.6 (a). Because the molecule is stable, we know that the attractive forces must overcome the repulsive ones. Let's take a closer look at the attractive forces that hold this molecule together.
FIGURE 8.6 (a). Because the molecule is stable, we know that the attractive forces must overcome the repulsive ones. Let's take a closer look at the attractive forces that hold this molecule together.
By using quantum mechanical methods analogous to those used for atoms in Section 6.5, we can calculate the distribution of electron density in molecules. Such a calculation for H2 shows that the attractions between the nuclei and the electrons cause electron density to concentrate between the nuclei, as shown in Figure 8.6(b). As a result, the overall electrostatic interactions are attractive. Thus, the atoms in H2 are held together principally because the two positive nuclei are attracted to the concentration of negative charge between them. In essence, the shared pair of electrons in any covalent bond acts as a kind of “glue” to bind atoms together.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Ionizing an H2 molecule to H2+ changes the strength of the bond. Based on the description of covalent bonding given previously, do you expect the H—H bond in H2+ to be weaker or stronger than the H—H bond in H2?
Lewis Structures
The formation of covalent bonds can be represented with Lewis symbols. The formation of the H2 molecule from two H atoms, for example, can be represented as
![]()
In forming the covalent bond, each hydrogen atom acquires a second electron, achieving the stable, two-electron, noble-gas electron configuration of helium.
Formation of a covalent bond between two Cl atoms to give a Cl2 molecule can be represented in a similar way:

By sharing the bonding electron pair, each chlorine atom has eight electrons (an octet) in its valence shell, thus achieving the noble-gas electron configuration of argon.
The structures shown here for H2 and Cl2 are called either Lewis structures or Lewis electron-dot structures. In writing Lewis structures, we usually show each shared electron pair as a line and any unshared electron pairs as dots. Written this way, the Lewis structures for H2 and Cl2are
![]()
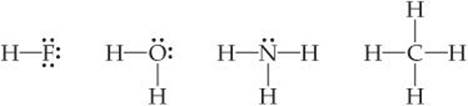
For nonmetals, the number of valence electrons in a neutral atom is the same as the group number. Therefore, one might predict that 7A elements, such as F, would form one covalent bond to achieve an octet; 6A elements, such as O, would form two covalent bonds; 5A elements, such as N, would form three; and 4A elements, such as C, would form four. These predictions are borne out in many compounds, as in, for example, the compounds with hydrogen of the nonmetals of the second row of the periodic table:
SAMPLE EXERCISE 8.3 Lewis Structure of a Compound
Given the Lewis symbols for nitrogen and fluorine in Table 8.1, predict the formula of the stable binary compound (a compound composed of two elements) formed when nitrogen reacts with fluorine and draw its Lewis structure.
SOLUTION
Analyze The Lewis symbols for nitrogen and fluorine reveal that nitrogen has five valence electrons and fluorine has seven.
Plan We need to find a combination of the two elements that results in an octet of electrons around each atom. Nitrogen requires three additional electrons to complete its octet, and fluorine requires one. Sharing a pair of electrons between one N atom and one F atom will result in an octet of electrons for fluorine but not for nitrogen. We therefore need to figure out a way to get two more electrons for the N atom.
Solve Nitrogen must share a pair of electrons with three fluorine atoms to complete its octet. Thus, the binary compound these two elements form must be NF3:

Check The Lewis structure in the center shows that each atom is surrounded by an octet of electrons. Once you are accustomed to thinking of each line in a Lewis structure as representing two electrons, you can just as easily use the structure on the right to check for octets.
PRACTICE EXERCISE
Compare the Lewis symbol for neon with the Lewis structure for methane, CH4. In what important way are the electron arrangements about neon and carbon alike? In what important way are they different?
Answer: Both atoms have an octet of electrons. However, the electrons about neon are unshared electron pairs, whereas those about carbon are shared with four hydrogen atoms.
Multiple Bonds
A shared electron pair constitutes a single covalent bond, generally referred to simply as a single bond. In many molecules, atoms attain complete octets by sharing more than one pair of electrons. When two electron pairs are shared, two lines are drawn in the Lewis structure, representing adouble bond. In carbon dioxide, for example, bonding occurs between carbon, with four valence electrons, and oxygen, with six:
![]()
As the diagram shows, each oxygen atom acquires an octet by sharing two electron pairs with carbon. Carbon acquires an octet by sharing two electron pairs with each of the two oxygen atoms; each double bond involves four electrons.
A triple bond corresponds to the sharing of three pairs of electrons, such as in the N2 molecule:
![]()
Because each nitrogen atom has five valence electrons, three electron pairs must be shared to achieve the octet configuration.
The properties of N2 are in complete accord with its Lewis structure. Nitrogen is a diatomic gas with exceptionally low reactivity that results from the very stable nitrogen–nitrogen bond. The nitrogen atoms are separated by only 1.10 Å. The short separation distance between the two N atoms is a result of the triple bond between the atoms. From studies of the structures of many different substances in which nitrogen atoms share one or two electron pairs, we have learned that the average distance between bonded nitrogen atoms varies with the number of shared electron pairs:
![]()
As a general rule, the length of the bond between two atoms decreases as the number of shared electron pairs increases.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
The C—O bond length in carbon monoxide, CO, is 1.13 Å, whereas the C—O bond length in CO2 is 1.24 Å. Without drawing a Lewis structure, do you think that CO contains a single, double, or triple bond?