CHEMISTRY THE CENTRAL SCIENCE
8 BASIC CONCEPTS OF CHEMICAL BONDING
8.6 RESONANCE STRUCTURES
We sometimes encounter molecules and ions in which the experimentally determined arrangement of atoms is not adequately described by a single dominant Lewis structure. Consider ozone, O3, which is a bent molecule with two equal O—O bond lengths (![]() FIGURE 8.12). Because each oxygen atom contributes 6 valence electrons, the ozone molecule has 18 valence electrons. This means the Lewis structure must have one O—O single bond and one O═O double bond to attain an octet about each atom:
FIGURE 8.12). Because each oxygen atom contributes 6 valence electrons, the ozone molecule has 18 valence electrons. This means the Lewis structure must have one O—O single bond and one O═O double bond to attain an octet about each atom:
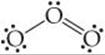
However, this single structure cannot by itself be dominant because it requires that one O—O bond be different from the other, contrary to the observed structure—we would expect the O═O double bond to be shorter than the O—O single bond. ![]() (Section 8.3) In drawing the Lewis structure, however, we could just as easily have put the O═O bond on the left:
(Section 8.3) In drawing the Lewis structure, however, we could just as easily have put the O═O bond on the left:
![]() GO FIGURE
GO FIGURE
What feature of this structure suggests that the two outer O atoms are in some way equivalent to each other?
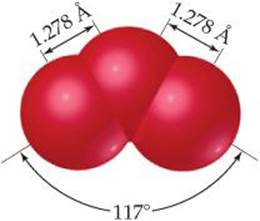
![]() FIGURE 8.12 Molecular structure of ozone.
FIGURE 8.12 Molecular structure of ozone.
![]() GO FIGURE
GO FIGURE
Is the electron density consistent with equal weights for the two resonance structures for O3? Explain.
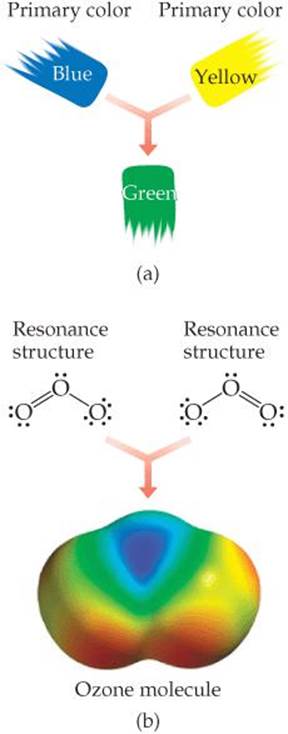
![]() FIGURE 8.13 Resonance. Describing a molecule as a blend of different resonance structures is similar to describing a paint color as a blend of primary colors. (a) Green paint is a blend of blue and yellow. We cannot describe green as a single primary color. (b) The ozone molecule is a blend of two resonance structures. We cannot describe the ozone molecule in terms of a single Lewis structure.
FIGURE 8.13 Resonance. Describing a molecule as a blend of different resonance structures is similar to describing a paint color as a blend of primary colors. (a) Green paint is a blend of blue and yellow. We cannot describe green as a single primary color. (b) The ozone molecule is a blend of two resonance structures. We cannot describe the ozone molecule in terms of a single Lewis structure.
There is no reason for one of these Lewis structures to be dominant because they are equally valid representations of the molecule. The placement of the atoms in these two alternative but completely equivalent Lewis structures is the same, but the placement of the electrons is different. Lewis structures of this sort are called resonance structures. To describe the structure of ozone properly, we write both resonance structures and use a double-headed arrow to indicate that the real molecule is described by an average of the two:

To understand why certain molecules require more than one resonance structure, we can draw an analogy to mixing paint (![]() FIGURE 8.13). Blue and yellow are both primary colors of paint pigment. An equal blend of blue and yellow pigments produces green pigment. We cannot describe green paint in terms of a single primary color, yet it still has its own identity. Green paint does not oscillate between its two primary colors: It is not blue part of the time and yellow the rest of the time. Similarly, molecules such as ozone cannot be described as oscillating between the two individual Lewis structures shown previously—there are two equivalent dominant Lewis structures that contribute equally to the actual structure of the molecule.
FIGURE 8.13). Blue and yellow are both primary colors of paint pigment. An equal blend of blue and yellow pigments produces green pigment. We cannot describe green paint in terms of a single primary color, yet it still has its own identity. Green paint does not oscillate between its two primary colors: It is not blue part of the time and yellow the rest of the time. Similarly, molecules such as ozone cannot be described as oscillating between the two individual Lewis structures shown previously—there are two equivalent dominant Lewis structures that contribute equally to the actual structure of the molecule.
The actual arrangement of the electrons in molecules such as O3 must be considered as a blend of two (or more) Lewis structures. By analogy to the green paint, the molecule has its own identity separate from the individual resonance structures. For example, the ozone molecule always has two equivalent O—O bonds whose lengths are intermediate between the lengths of an oxygen-oxygen single bond and an oxygen-oxygen double bond. Another way of looking at it is to say that the rules for drawing Lewis structures do not allow us to have a single dominant structure for the ozone molecule. For example, there are no rules for drawing half-bonds. We can get around this limitation by drawing two equivalent Lewis structures that, when averaged, amount to something very much like what is observed experimentally.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
The O—O bonds in ozone are often described as “one-and-a-half” bonds. Is this description consistent with the idea of resonance?
As an additional example of resonance structures, consider the nitrate ion, ![]() , for which three equivalent Lewis structures can be drawn:
, for which three equivalent Lewis structures can be drawn:
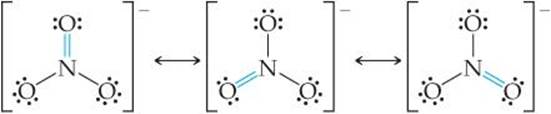
Notice that the arrangement of atoms is the same in each structure—only the placement of electrons differs. In writing resonance structures, the same atoms must be bonded to each other in all structures, so that the only differences are in the arrangements of electrons. All three ![]() Lewis structures are equally dominant and taken together adequately describe the ion, in which all three N—O bond lengths are the same.
Lewis structures are equally dominant and taken together adequately describe the ion, in which all three N—O bond lengths are the same.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
In the same sense that we describe the O—O bonds in O3 as “one-and-a-half” bonds, how would you describe the N—O bonds in ![]() ?
?
In some instances, all the possible Lewis structures for a species may not be equivalent to one another. Instead, one or more may be dominant. We will encounter examples of this as we proceed.
SAMPLE EXERCISE 8.10 Resonance Structures
Which is predicted to have the shorter sulfur–oxygen bonds, SO3 or ![]() ?
?
SOLUTION
The sulfur atom has six valence electrons, as does oxygen. Thus, SO3 contains 24 valence electrons. In writing the Lewis structure, we see that three equivalent resonance structures can be drawn:
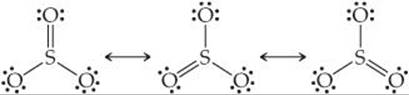
As with ![]() , the actual structure of SO3 is an equal blend of all three. Thus, each S—O bond length should be about one-third of the way between the length of a single bond and the length of a double bond. That is, they should be shorter than single bonds but not as short as double bonds.
, the actual structure of SO3 is an equal blend of all three. Thus, each S—O bond length should be about one-third of the way between the length of a single bond and the length of a double bond. That is, they should be shorter than single bonds but not as short as double bonds.
The ![]() ion has 26 electrons, which leads to a dominant Lewis structure in which all the S—O bonds are single:
ion has 26 electrons, which leads to a dominant Lewis structure in which all the S—O bonds are single:
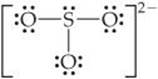
Our analysis of the Lewis structures leads us to conclude that SO3 should have the shorter S—O bonds and ![]() the longer ones. This conclusion is correct: The experimentally measured SO bond lengths are 1.42 Å in SO3 and 1.51 Å in
the longer ones. This conclusion is correct: The experimentally measured SO bond lengths are 1.42 Å in SO3 and 1.51 Å in ![]() .
.
PRACTICE EXERCISE
Draw two equivalent resonance structures for the formate ion, ![]() .
.
Answer: 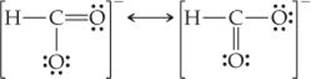
Resonance in Benzene
Resonance is an important concept in describing the bonding in organic molecules, particularly aromatic organic molecules, a category that includes the hydrocarbon benzene, C6H6. The six C atoms are bonded in a hexagonal ring, and one H atom is bonded to each C atom. We can write two equivalent dominant Lewis structures for benzene, each of which satisfies the octet rule. These two structures are in resonance:
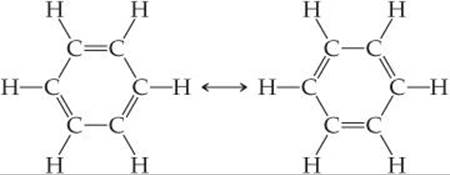
Note that the double bonds are in different places in the two structures. Each of these resonance structures shows three carbon-carbon single bonds and three carbon-carbon double bonds. However, experimental data show that all six C—C bonds are of equal length, 1.40 Å, intermediate between the typical bond lengths for a C—C single bond (1.54 Å) and a C═C double bond (1.34 Å). Each of the C—C bonds in benzene can be thought of as a blend of a single bond and a double bond (![]() FIGURE 8.14).
FIGURE 8.14).
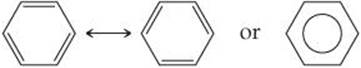
Benzene is commonly represented by omitting the hydrogen atoms and showing only the carbon-carbon framework with the vertices unlabeled. In this convention, the resonance in the molecule is represented either by two structures separated by a double-headed arrow or by a shorthand notation in which we draw a hexagon with a circle inside:
![]() GO FIGURE
GO FIGURE
What is the significance of the dashed bonds in this ball-and-stick model?
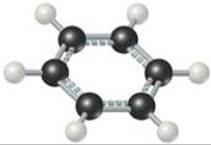
![]() FIGURE 8.14 Benzene, an “aromatic” organic compound. The benzene molecule is a regular hexagon of carbon atoms with a hydrogen atom bonded to each one. The dashed lines represent the blending of two equivalent resonance structures, leading to C—C bonds that are intermediate between single and double bonds.
FIGURE 8.14 Benzene, an “aromatic” organic compound. The benzene molecule is a regular hexagon of carbon atoms with a hydrogen atom bonded to each one. The dashed lines represent the blending of two equivalent resonance structures, leading to C—C bonds that are intermediate between single and double bonds.
The shorthand notation reminds us that benzene is a blend of two resonance structures—it emphasizes that the C═C double bonds cannot be assigned to specific edges of the hexagon. Chemists use both representations of benzene interchangeably.
The bonding arrangement in benzene confers special stability to the molecule. As a result, millions of organic compounds contain the six-membered ring characteristic of benzene. Many of these compounds are important in biochemistry, in pharmaceuticals, and in the production of modern materials.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Each Lewis structure of benzene has three C═C double bonds. Another hydrocarbon containing three C═C double bonds is hexatriene, C6H8. A Lewis structure of hexatriene is
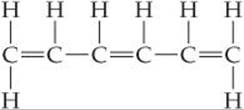
Do you expect hexatriene to have multiple resonance structures? If not, why is this molecule different from benzene with respect to resonance?