CHEMISTRY THE CENTRAL SCIENCE
8 BASIC CONCEPTS OF CHEMICAL BONDING
8.7 EXCEPTIONS TO THE OCTET RULE
The octet rule is so simple and useful in introducing the basic concepts of bonding that you might assume it is always obeyed. In Section 8.2, however, we noted its limitation in dealing with ionic compounds of the transition metals. The rule also fails in many situations involving covalent bonding. These exceptions to the octet rule are of three main types:
1. Molecules and polyatomic ions containing an odd number of electrons
2. Molecules and polyatomic ions in which an atom has fewer than an octet of valence electrons
3. Molecules and polyatomic ions in which an atom has more than an octet of valence electrons
Odd Number of Electrons
In the vast majority of molecules and polyatomic ions, the total number of valence electrons is even, and complete pairing of electrons occurs. However, in a few molecules and polyatomic ions, such as ClO2, NO, NO2, and O2-, the number of valence electrons is odd. Complete pairing of these electrons is impossible, and an octet around each atom cannot be achieved. For example, NO contains 5 + 6 = 11 valence electrons. The two most important Lewis structures for this molecule are
![]()
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Which of the Lewis structures for NO is dominant based on analysis of the formal charges?
Less than an Octet of Valence Electrons
A second type of exception occurs when there are fewer than eight valence electrons around an atom in a molecule or polyatomic ion. This situation is also relatively rare (with the exception of hydrogen and helium as we have already discussed), most often encountered in compounds of boron and beryllium. As an example, let's consider boron trifluoride, BF3. If we follow the first four steps of our procedure for drawing Lewis structures, we obtain the structure

which has only six electrons around the boron atom. The formal charge is zero on both B and F, and we could complete the octet around boron by forming a double bond (step 5). In so doing, we see that there are three equivalent resonance structures (the formal charges are shown in red):

Each of these structures forces a fluorine atom to share additional electrons with the boron atom, which is inconsistent with the high electronegativity of fluorine. In fact, the formal charges tell us that this is an unfavorable situation. In each structure, the F atom involved in the B═F double bond has a formal charge of +1, while the less electronegative B atom has a formal charge of -1. Thus, the resonance structures containing a B═F double bond are less important than the one in which there are fewer than an octet of valence electrons around boron:
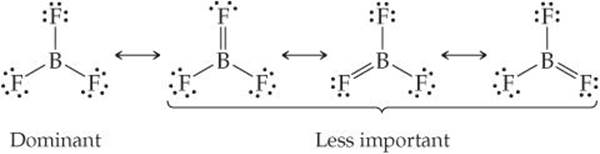
We usually represent BF3 solely by the dominant resonance structure, in which there are only six valence electrons around boron. The chemical behavior of BF3 is consistent with this representation. In particular, BF3 reacts energetically with molecules having an unshared pair of electrons that can be used to form a bond with boron, as, for example, in the reaction

In the stable compound NH3BF3, boron has an octet of valence electrons.
More than an Octet of Valence Electrons
The third and largest class of exceptions consists of molecules or polyatomic ions in which there are more than eight electrons in the valence shell of an atom. When we draw the Lewis structure for PF5, for example, we are forced to place ten electrons around the central phosphorus atom:

Molecules and ions with more than an octet of electrons around the central atom are often called hypervalent. Other examples of hypervalent species are SF4, AsF6-, and ![]() . The corresponding molecules with a second-period atom as the central atom, such as NCl5 and OF4, do notexist.
. The corresponding molecules with a second-period atom as the central atom, such as NCl5 and OF4, do notexist.
Hypervalent molecules are formed only for central atoms from period 3 and below in the periodic table. The principal reason for their formation is the relatively larger size of the central atom. For example, a P atom is large enough that five F (or even five Cl) atoms can be bonded to it without being too crowded. By contrast, an N atom is too small to accommodate five atoms bonded to it. Because size is a factor, hypervalent molecules occur most often when the central atom is bonded to the smallest and most electronegative atoms—F, Cl, and O.
The notion of a valence shell containing more than an octet of electrons is also consistent with the presence of unfilled nd orbitals in atoms from period 3 and below. By comparison, elements of the second period have only the 2s and 2p valence orbitals available for bonding. Detailed analyses of the bonding in molecules such as PF5 and SF6 suggest that the presence of unfilled 3d orbitals in P and S has a relatively minor impact on the formation of hypervalent molecules, and the general current belief is that the increased size of third-period atoms is the more important factor.
SAMPLE EXERCISE 8.11 Lewis Structure for an Ion with More than an Octet of Electrons
Draw the Lewis structure for ![]() .
.
SOLUTION
Iodine (group 7A) has seven valence electrons. Each chlorine atom (group 7A) also has seven. An extra electron is added to account for the 1- charge of the ion. Therefore, the total number of valence electrons is
7 + (4 × 7) + 1 = 36
The I atom is the central atom in the ion. Putting eight electrons around each Cl atom (including a pair of electrons between I and each Cl to represent the single bond between these atoms) requires 8 × 4 = 32 electrons.
We are thus left with 36 - 32 = 4 electrons to be placed on the larger iodine:

Iodine has 12 valence electrons around it, four more than needed for an octet.
PRACTICE EXERCISE
(a) Which of the following atoms is never found with more than an octet of valence electrons around it: S, C, P, Br? (b) Draw the Lewis structure for XeF2.
Answers: (a) C, ![]()
Finally, there are Lewis structures where you might have to choose between satisfying the octet rule and obtaining the most favorable formal charges by using more than an octet of electrons. For example, consider these Lewis structures for the phosphate ion, ![]() :
:

The formal charges on the atoms are shown in red. In the left structure, the P atom obeys the octet rule. In the right structure, however, the P atom has five electron pairs, leading to smaller formal charges on the atoms. (You should be able to see that there are three additional resonance structures for the Lewis structure on the right.)
Chemists are still debating which of these two structures is dominant for ![]() . Recent theoretical calculations based on quantum mechanics suggest to some researchers that the left structure is the dominant one. Other researchers claim that the bond lengths in the ion are more consistent with the right structure being dominant. This disagreement is a convenient reminder that, in general, multiple Lewis structures can contribute to the actual electron distribution in an atom or molecule.
. Recent theoretical calculations based on quantum mechanics suggest to some researchers that the left structure is the dominant one. Other researchers claim that the bond lengths in the ion are more consistent with the right structure being dominant. This disagreement is a convenient reminder that, in general, multiple Lewis structures can contribute to the actual electron distribution in an atom or molecule.