CHEMISTRY THE CENTRAL SCIENCE
9 MOLECULAR GEOMETRY AND BONDING THEORIES
9.3 MOLECULAR SHAPE AND MOLECULAR POLARITY
Recall that bond polarity is a measure of how equally the electrons in a bond are shared between the two atoms of the bond. As the difference in electronegativity between the two atoms increases, so does the bond polarity. ![]() (Section 8.4) We saw that the dipole moment of a diatomic molecule is a measure of the amount of charge separation in the molecule.
(Section 8.4) We saw that the dipole moment of a diatomic molecule is a measure of the amount of charge separation in the molecule.
For a molecule consisting of more than two atoms, the dipole moment depends on both the polarities of the individual bonds and the geometry of the molecule. For each bond in the molecule, we consider the bond dipole, which is the dipole moment due only to the two atoms in that bond. Consider the linear CO2 molecule, for example. As shown in ![]() FIGURE 9.10, each C═O bond is polar, and because the C═O bonds are identical, the bond dipoles are equal in magnitude. A plot of the molecule's electron density clearly shows that the individual bonds are polar, but what can we say about the overall dipole moment of the molecule?
FIGURE 9.10, each C═O bond is polar, and because the C═O bonds are identical, the bond dipoles are equal in magnitude. A plot of the molecule's electron density clearly shows that the individual bonds are polar, but what can we say about the overall dipole moment of the molecule?
Bond dipoles and dipole moments are vector quantities; that is, they have both a magnitude and a direction. The dipole moment of a polyatomic molecule is the vector sum of its bond dipoles. Both the magnitudes and the directions of the bond dipoles must be considered when summing vectors. The two bond dipoles in CO2, although equal in magnitude, are opposite in direction. Adding them is the same as adding two numbers that are equal in magnitude but opposite in sign, such as 100 + (– 100). The bond dipoles, like the numbers, “cancel” each other. Therefore, the dipole moment of CO2 is zero, even though the individual bonds are polar. The geometry of the molecule dictates that the overall dipole moment be zero, making CO2 a nonpolar molecule.
Now consider H2O, a bent molecule with two polar bonds (![]() FIGURE 9.11). Again, the two bonds are identical, and the bond dipoles are equal in magnitude. Because the molecule is bent, however, the bond dipoles do not directly oppose each other and therefore do not cancel. Hence, the H2O molecule has an overall nonzero dipole moment (μ= 1.85 D) and is therefore a polar molecule. The oxygen atom carries a partial negative charge, and the hydrogen atoms each have a partial positive charge, as shown in the electron-density model.
FIGURE 9.11). Again, the two bonds are identical, and the bond dipoles are equal in magnitude. Because the molecule is bent, however, the bond dipoles do not directly oppose each other and therefore do not cancel. Hence, the H2O molecule has an overall nonzero dipole moment (μ= 1.85 D) and is therefore a polar molecule. The oxygen atom carries a partial negative charge, and the hydrogen atoms each have a partial positive charge, as shown in the electron-density model.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
The molecule O ═ C ═ S is linear and has a Lewis structure analogous to that of CO2. Would you expect this molecule to have a dipole moment?
![]() FIGURE 9.12 shows some polar and nonpolar molecules, all with polar bonds. The molecules in which the central atom is symmetrically surrounded by identical atoms (BF3 and CCl4) are nonpolar. For ABn molecules in which all the B atoms are the same, certain symmetrical shapes—linear (AB2), trigonal planar (AB3), tetrahedral and square planar (AB4), trigonal bipyramidal (AB5), and octahedral (AB6)—must lead to nonpolar molecules even though the individual bonds might be polar.
FIGURE 9.12 shows some polar and nonpolar molecules, all with polar bonds. The molecules in which the central atom is symmetrically surrounded by identical atoms (BF3 and CCl4) are nonpolar. For ABn molecules in which all the B atoms are the same, certain symmetrical shapes—linear (AB2), trigonal planar (AB3), tetrahedral and square planar (AB4), trigonal bipyramidal (AB5), and octahedral (AB6)—must lead to nonpolar molecules even though the individual bonds might be polar.
![]() GO FIGURE
GO FIGURE
Explain how the directions of the red bond dipole arrows relate to the electron density picture.
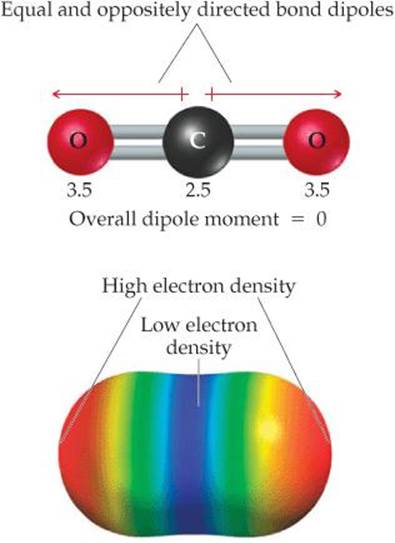
![]() FIGURE 9.10 CO2, a nonpolar molecule. The numbers are electronegativity values for these two atoms.
FIGURE 9.10 CO2, a nonpolar molecule. The numbers are electronegativity values for these two atoms.
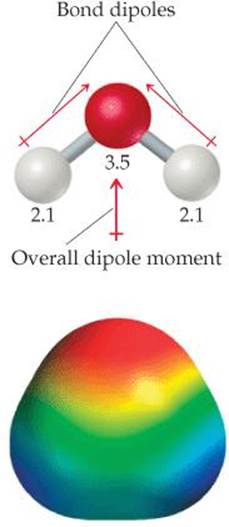
![]() FIGURE 9.11 H2O, a polar molecule. The numbers are electronegativity values.
FIGURE 9.11 H2O, a polar molecule. The numbers are electronegativity values.
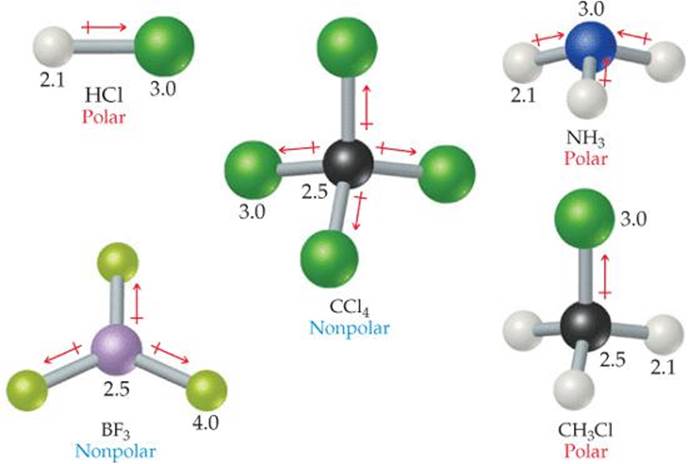
![]() FIGURE 9.12 Polar and nonpolar molecules containing polar bonds. The numbers are electronegativity values.
FIGURE 9.12 Polar and nonpolar molecules containing polar bonds. The numbers are electronegativity values.
SAMPLE EXERCISE 9.4 Polarity of Molecules
Predict whether these molecules are polar or nonpolar: (a) BrCl, (b) SO2, (c) SF6.
SOLUTION
Analyze We are given three molecular formulas and asked to predict whether the molecules are polar.
Plan A molecule containing only two atoms is polar if the atoms differ in electronegativity. The polarity of a molecule containing three or more atoms depends on both the molecular geometry and the individual bond polarities. Thus, we must draw a Lewis structure for each molecule containing three or more atoms and determine its molecular geometry. We then use electronegativity values to determine the direction of the bond dipoles. Finally, we see whether the bond dipoles cancel to give a nonpolar molecule or reinforce each other to give a polar one.
Solve
(a) Chlorine is more electronegative than bromine. All diatomic molecules with polar bonds are polar molecules. Consequently, BrCl is polar, with chlorine carrying the partial negative charge:
![]()
The measured dipole moment of BrCl is μ = 0.57 D.
(b) Because oxygen is more electronegative than sulfur, SO2 has polar bonds. Three resonance forms can be written:
![]()
For each of these, the VSEPR model predicts a bent molecular geometry. Because the molecule is bent, the bond dipoles do not cancel, and the molecule is polar:
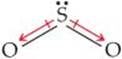
Experimentally, the dipole moment of SO2μ = 1.63 D.
(c) Fluorine is more electronegative than sulfur, so the bond dipoles point toward fluorine. For clarity, only one S—F dipole is shown. The six S—F bonds are arranged octahedrally around the central sulfur:
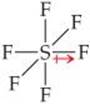
Because the octahedral molecular geometry is symmetrical, the bond dipoles cancel, and the molecule is nonpolar, meaning that μ = 0.
PRACTICE EXERCISE
Determine whether the following molecules are polar or nonpolar: (a) NF3, (b) BCl3.
Answers: (a) polar because polar bonds are arranged in a trigonal-pyramidal geometry, (b) nonpolar because polar bonds are arranged in a trigonal-planar geometry