CHEMISTRY THE CENTRAL SCIENCE
9 MOLECULAR GEOMETRY AND BONDING THEORIES
9.7 MOLECULAR ORBITALS
Valence-bond theory and hybrid orbitals allow us to move in a straightforward way from Lewis structures to rationalizing the observed geometries of molecules in terms of atomic orbitals. The valence-bond model, however, does not explain all aspects of bonding. It is not successful, for example, in describing the excited states of molecules, which we must understand to explain how molecules absorb light, giving them color.
Some aspects of bonding are better explained by a more sophisticated model called molecular orbital theory. In Chapter 6 we saw that electrons in atoms can be described by wave functions, which we call atomic orbitals. In a similar way, molecular orbital theory describes the electrons in molecules by using specific wave functions called molecular orbitals (MO).
Molecular orbitals have many of the same characteristics as atomic orbitals. For example, an MO can hold a maximum of two electrons (with opposite spins), it has a definite energy, and we can visualize its electron-density distribution by using a contour representation, as we did with atomic orbitals. Unlike atomic orbitals, however, MOs are associated with an entire molecule, not with a single atom.
The Hydrogen Molecule
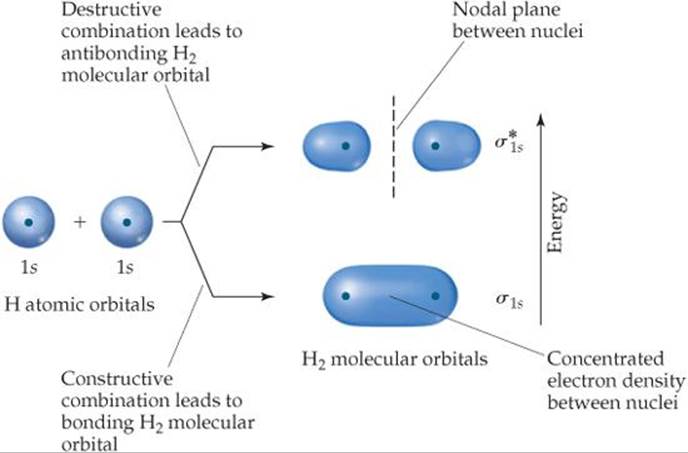
We begin our study of MO theory with the hydrogen molecule, H2. Whenever two atomic orbitals overlap, two molecular orbitals form. Thus, the overlap of the 1s orbitals of two hydrogen atoms to form H2 produces two MOs (![]() FIGURE 9.32). One MO is formed by adding the wave functions for the two 1s orbitals. We refer to this as constructive combination. The energy of the resulting MO is lower than the energy of the two atomic orbitals from which it was made. It is called the bonding molecular orbital.
FIGURE 9.32). One MO is formed by adding the wave functions for the two 1s orbitals. We refer to this as constructive combination. The energy of the resulting MO is lower than the energy of the two atomic orbitals from which it was made. It is called the bonding molecular orbital.
The other MO is formed by combining the two atomic orbitals in a way that causes the electron density to be more or less canceled in the central region where the two overlap. We refer to this as destructive combination. The process is discussed more fully in the “Closer Look” box later in the chapter; we don't need to concern ourselves with it to understand molecular orbital bond formation. The energy of the resulting MO, referred to as the antibonding molecular orbital, is higher than the energy of the atomic orbitals.
As illustrated in Figure 9.32, in the bonding MO electron density is concentrated in the region between the two nuclei. This sausage-shaped MO results from summing the two atomic orbitals so that the atomic orbital wave functions combine in the region between the two nuclei. Because an electron in this MO is attracted to both nuclei, the electron is more stable (it has lower energy) than it is in the 1s atomic orbital of an isolated hydrogen atom. Further, because this bonding MO concentrates electron density between the nuclei, it holds the atoms together in a covalent bond.
![]() FIGURE 9.32 The two molecular orbitals of H2, one a bonding MO and one an antibonding MO.
FIGURE 9.32 The two molecular orbitals of H2, one a bonding MO and one an antibonding MO.
By contrast, the antibonding MO has very little electron density between the nuclei. Instead of combining in the region between the nuclei, the atomic orbital wave functions cancel each other in this region, leaving the greatest electron density on opposite sides of the two nuclei. Thus, this MO excludes electrons from the very region in which a bond must be formed. Antibonding orbitals invariably have a nodal plane in the region between the nuclei, where the electron density is zero. (The nodal plane is shown as a dashed line in Figure 9.32 and subsequent figures.) An electron in an antibonding MO is repelled from the bonding region and is therefore less stable (it has higher energy) than it is in the 1s atomic orbital of a hydrogen atom.
Notice from Figure 9.32 that the electron density in both the bonding MO and the antibonding MO of H2 is centered about the internuclear axis. MOs of this type are called sigma (σ) molecular orbitals (by analogy to σ bonds). The bonding sigma MO of H2 is labeled σs1s; the subscript indicates that the MO is formed from two 1s orbitals. The antibonding sigma MO of H2 is labeled ![]() (read “sigma-star-one-s”); the asterisk denotes that the MO is antibonding.
(read “sigma-star-one-s”); the asterisk denotes that the MO is antibonding.
The relative energies of two 1s atomic orbitals and the molecular orbitals formed from them are represented by an energy-level diagram (also called a molecular orbital diagram). Such diagrams show the interacting atomic orbitals on the left and right and the MOs in the middle, as shown in ![]() FIGURE 9.33. Like atomic orbitals, each MO can accommodate two electrons with their spins paired (Pauli exclusion principle).
FIGURE 9.33. Like atomic orbitals, each MO can accommodate two electrons with their spins paired (Pauli exclusion principle). ![]() (Section 6.7)
(Section 6.7)
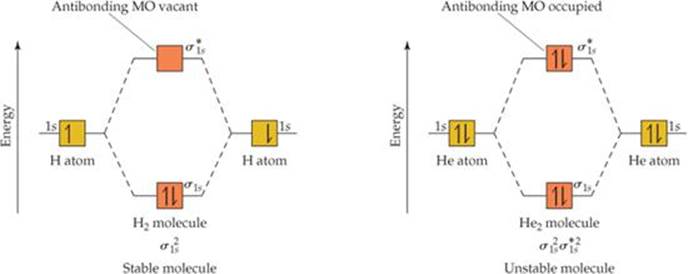
As the MO diagram for H2 in Figure 9.33 shows, each H atom brings one electron to the molecule, so there are two electrons in H2. These two electrons occupy the lower-energy bonding (σ1s) MO, and their spins are paired. Electrons occupying a bonding molecular orbital are calledbonding electrons. Because the σ1s MO is lower in energy than the 1s atomic orbitals, the H2 molecule is more stable than the two separate H atoms.
By analogy with atomic electron configurations, the electron configurations for molecules can be written with superscripts to indicate electron occupancy. The electron configuration for H2, then, is ![]() .
.
![]() GO FIGURE
GO FIGURE
By referring to Figure 9.32, determine which molecular orbital in He2 has a node between the nuclei.
![]() FIGURE 9.33 Energy-level diagrams and electron configurations for H2 and He2.
FIGURE 9.33 Energy-level diagrams and electron configurations for H2 and He2.
Figure 9.33 also shows the energy-level diagram for the hypothetical He2 molecule, which requires four electrons to fill its molecular orbitals. Because only two electrons can go in the σ1s MO, the other two electrons must go in the ![]() MO. The electron configuration of He2 is thus
MO. The electron configuration of He2 is thus ![]() . The energy decrease realized in going from He atomic orbitals to the He bonding MO is offset by the energy increase realized in going from the atomic orbitals to the He antibonding MO.† Hence, He2 is an unstable molecule. Molecular orbital theory correctly predicts that hydrogen forms diatomic molecules but helium does not.
. The energy decrease realized in going from He atomic orbitals to the He bonding MO is offset by the energy increase realized in going from the atomic orbitals to the He antibonding MO.† Hence, He2 is an unstable molecule. Molecular orbital theory correctly predicts that hydrogen forms diatomic molecules but helium does not.
Bond Order
In molecular orbital theory, the stability of a covalent bond is related to its bond order, defined as half the difference between the number of bonding electrons and the number of antibonding electrons:
![]()
We take half the difference because we are used to thinking of bonds as pairs of electrons. A bond order of 1 represents a single bond, a bond order of 2 represents a double bond, and a bond order of 3 represents a triple bond. Because MO theory also treats molecules containing an odd number of electrons, bond orders of 1/2, 3/2, or 5/2 are possible.
Because, as Figure 9.33 shows, H2 has two bonding electrons and zero antibonding electrons, it has a bond order of 1. Because He2 has two bonding electrons and two anti-bonding electrons, it has a bond order of 0. A bond order of 0 means that no bond exists.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Suppose one electron in H2 is excited from the σ1s MO to the ![]() MO. Would you expect the H atoms to remain bonded to each other, or would the molecule fall apart?
MO. Would you expect the H atoms to remain bonded to each other, or would the molecule fall apart?
![]() GO FIGURE
GO FIGURE
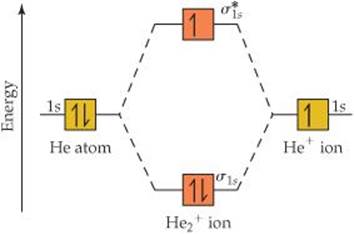
Which electrons in this diagram contribute to the stability of the He2+ ion?
![]() FIGURE 9.34 Energy-level diagram for the He2+ion.
FIGURE 9.34 Energy-level diagram for the He2+ion.
SAMPLE EXERCISE 9.8 Bond Order
What is the bond order of the He2+ ion? Would you expect this ion to be stable relative to the separated He atom and He+ ion?
SOLUTION
Analyze We will determine the bond order for the He2+ ion and use it to predict whether the ion is stable.
Plan To determine the bond order, we must determine the number of electrons in the molecule and how these electrons populate the available MOs. The valence electrons of He are in the 1s orbital, and the 1s orbitals combine to give an MO diagram like that for H2 or He2 (Figure 9.33). If the bond order is greater than 0, we expect a bond to exist, and the ion is stable.
Solve The energy-level diagram for the He2+ ion is shown in ![]() FIGURE 9.34. This ion has three electrons. Two are placed in the bonding orbital and the third in the antibonding orbital. Thus, the bond order is
FIGURE 9.34. This ion has three electrons. Two are placed in the bonding orbital and the third in the antibonding orbital. Thus, the bond order is
![]()
Because the bond order is greater than 0, we predict the He2+ ion to be stable relative to the separated He and He+. Formation of He2+ in the gas phase has been demonstrated in laboratory experiments.
PRACTICE EXERCISE
Determine the bond order of the H2– ion.
Answer: ![]()