CHEMISTRY THE CENTRAL SCIENCE
10 GASES

EARTH'S ATMOSPHERE, which is mostly made of nitrogen and oxygen gases, extends about 350 miles up from Earth's surface. Over 95% of the mass of the atmosphere, however, is in the 30 miles closest to the surface. Most of what we perceive as “weather” occurs in the first 10 miles above the surface.
WHAT'S AHEAD
10.1 CHARACTERISTICS OF GASES
We begin by comparing the distinguishing characteristics of gases with those of liquids and solids.
10.2 PRESSURE
We then study gas pressure, how it is measured, and the units used to express it, as well as consider Earth's atmosphere and the pressure it exerts.
10.3 THE GAS LAWS
We next see that the state of a gas can be expressed in terms of its volume, pressure, temperature, and quantity and examine several gas laws, which are empirical relationships among these four variables.
10.4 THE IDEAL-GAS EQUATION
We find that the gas laws yield the ideal-gas equation, PV = nRT. Although this equation is not obeyed exactly by any real gas, most gases come very close to obeying it at ordinary temperatures and pressures.
10.5 FURTHER APPLICATIONS OF THE IDEAL-GAS EQUATION
We use the ideal-gas equation in many calculations, such as the calculation of the density or molar mass of a gas.
10.6 GAS MIXTURES AND PARTIAL PRESSURES
We recognize that in a mixture of gases, each gas exerts a pressure that is part of the total pressure. This partial pressure is the pressure the gas would exert if it were by itself.
10.7 THE KINETIC-MOLECULAR THEORY OF GASES
We see that this theory helps us understand gas behavior on the molecular level. According to the theory, the atoms or molecules that make up a gas move with an average kinetic energy that is proportional to the gas temperature.
10.8 MOLECULAR EFFUSION AND DIFFUSION
We observe that the kinetic-molecular theory helps us account for such gas properties as effusion, movement through tiny openings, and diffusion, movement through another substance.
10.9 REAL GASES: DEVIATIONS FROM IDEAL BEHAVIOR
We learn that real gases deviate from ideal behavior because the gas molecules have finite volume and because attractive forces exist between molecules. The van der Waals equation gives an accurate account of real gas behavior at high pressures and low temperatures.
IN THE PAST SEVERAL CHAPTERS we learned about the electronic structure of atoms and about how atoms combine to form molecules and ionic substances. In everyday life, however, we do not have any direct experience with atoms. Instead, we encounter matter as gases, liquids, or solids made up of enormous numbers of atoms or molecules. Large collections of gas atoms and gas molecules in the atmosphere, for example, are responsible for weather—the gentle breezes and the gales, the humidity and the rain. Hurricanes, such as the one shown in the chapter-opening photo, are large storms (up to 500 miles in diameter) that form over the ocean and are characterized by a low-pressure center (the “eye”), heavy rains, and violent winds, with wind speeds up to 200 miles per hour. Hurricane formation is still not completely understood, but in general the atoms and molecules of the atmosphere are driven by a combination of low pressures, warm temperatures at the sea surface that rapidly cool with height, and high humidity to produce the storms.
We now know that the properties of gases (as well as those of liquids and solids) are readily understood in terms of the behavior of their component atoms, ions, and molecules; in this chapter we examine the physical properties of gases and explain these properties in terms of the behavior of gas molecules.
10.1 CHARACTERISTICS OF GASES
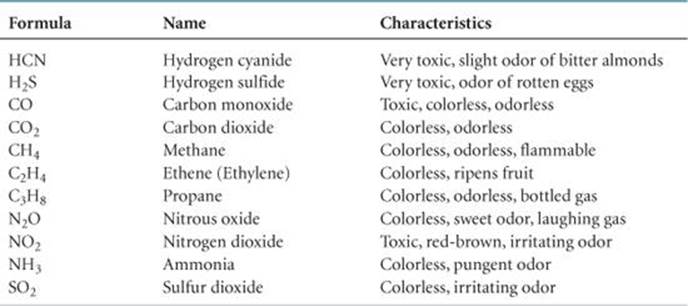
In many ways gases are the most easily understood form of matter. Even though different gaseous substances may have very different chemical properties, they behave quite similarly as far as their physical properties are concerned. For example, we live in an atmosphere composed of the mixture of gases we refer to as air, a mixture of primarily N2 (78%) and O2 (21%), with small amounts of several other gases, including Ar (0.9%). Although N2 and O2 have very different chemical properties—O2 supports human life but N2 does not, to name just one difference—these two components of air behave physically as one gaseous material because their physical properties are essentially identical. Of the few elements that exist as gases at ordinary temperatures and pressures, He, Ne, Ar, Kr, and Xe are monatomic and H2, N2, O2, F2, and Cl2 are diatomic. Many molecular compounds are gases, and ![]() TABLE 10.1 lists a few of them. Notice that all of these gases are composed entirely of nonmetallic elements. Furthermore, all have simple molecular formulas and, therefore, low molar masses.
TABLE 10.1 lists a few of them. Notice that all of these gases are composed entirely of nonmetallic elements. Furthermore, all have simple molecular formulas and, therefore, low molar masses.
Substances that are liquids or solids under ordinary conditions can also exist in the gaseous state, where they are often referred to as vapors. The substance H2O, for example, can exist as liquid water, solid ice, or water vapor.
Gases differ significantly from solids and liquids in several respects. For example, a gas expands spontaneously to fill its container. Consequently, the volume of a gas equals the volume of its container. Gases also are highly compressible: When pressure is applied to a gas, its volume readily decreases. Solids and liquids, on the other hand, do not expand to fill their containers and are not readily compressible.
Two or more gases form a homogeneous mixture regardless of the identities or relative proportions of the gases; the atmosphere serves as an excellent example. Two or more liquids or two or more solids may or may not form homogeneous mixtures, depending on their chemical nature. For example, when water and gasoline are mixed, the two liquids remain as separate layers. In contrast, the water vapor and gasoline vapors above the liquids form a homogeneous gas mixture.
The characteristic properties of gases—expanding to fill a container, being highly compressible, forming homogeneous mixtures—arise because the molecules are relatively far apart. In any given volume of air, for example, the molecules take up only about 0.1% of the total volume with the rest being empty space. Thus, each molecule behaves largely as though the others were not present. As a result, different gases behave similarly even though they are made up of different molecules.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Do the compounds in Table 10.1 have small (less than 100 g/mol) or large molecular weights?
TABLE 10.1 • Some Common Compounds That Are Gases at Room Temperature