CHEMISTRY THE CENTRAL SCIENCE
10 GASES
10.6 GAS MIXTURES AND PARTIAL PRESSURES
Thus far we have considered mainly pure gases—those that consist of only one substance in the gaseous state. How do we deal with mixtures of two or more different gases? While studying the properties of air, John Dalton ![]() (Section 2.1) made an important observation: The total pressure of a mixture of gases equals the sum of the pressures that each would exert if it were present alone. The pressure exerted by a particular component of a mixture of gases is called the partial pressure of that component. Dalton's observation is known as Dalton's law of partial pressures.
(Section 2.1) made an important observation: The total pressure of a mixture of gases equals the sum of the pressures that each would exert if it were present alone. The pressure exerted by a particular component of a mixture of gases is called the partial pressure of that component. Dalton's observation is known as Dalton's law of partial pressures.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
How is the pressure exerted by N2 gas affected when some O2 is introduced into a container if the temperature and volume remain constant?
If we let Pt be the total pressure of a mixture of gases and P1, P2, P3, and so forth be the partial pressures of the individual gases, we can write Dalton's law of partial pressures as
![]()
This equation implies that each gas behaves independently of the others, as we can see by the following analysis. Let n1, n2, n3, and so forth be the number of moles of each of the gases in the mixture and nt be the total number of moles of gas. If each gas obeys the ideal-gas equation, we can write
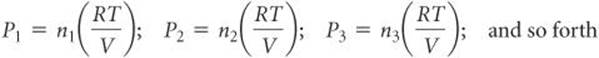
All the gases are at the same temperature and occupy the same volume. Therefore, by substituting into Equation 10.12, we obtain
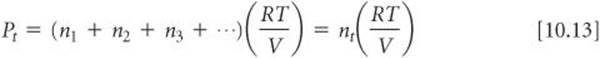
That is, at constant temperature and constant volume the total pressure of a gas sample is determined by the total number of moles of gas present, whether that total represents just one gas or a mixture of gases.
SAMPLE EXERCISE 10.10 Applying Dalton's Law of Partial Pressures
A mixture of 6.00 g O2(g) and 9.00 g CH4(g) is placed in a 15.0-L vessel at 0 °C. What is the partial pressure of each gas, and what is the total pressure in the vessel?
SOLUTION
Analyze We need to calculate the pressure for two gases in the same volume and at the same temperature.
Plan Because each gas behaves independently, we can use the ideal-gas equation to calculate the pressure each would exert if the other were not present. The total pressure is the sum of these two partial pressures.
Solve We first convert the mass of each gas to moles:
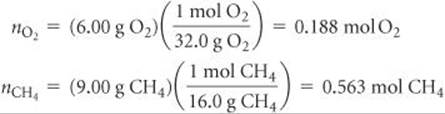
We use the ideal-gas equation to calculate the partial pressure of each gas:

According to Dalton's law of partial pressures (Equation 10.12), the total pressure in the vessel is the sum of the partial pressures:
Pt = PO2 + PCH4 = 0.281 atm + 0.841 atm = 1.122 atm
Check A pressure of roughly 1 atm seems right for a mixture of about 0.2 mol O2 and a bit more than 0.5 mol CH4, together in a 15-L volume, because 1 mol of an ideal gas at 1 atm pressure and 0 °C occupies about 22 L.
PRACTICE EXERCISE
What is the total pressure exerted by a mixture of 2.00 g of H2(g) and 8.00 g of N2(g) at 273 K in a 10.0-L vessel?
Answer: 2.86 atm
Partial Pressures and Mole Fractions
Because each gas in a mixture behaves independently, we can relate the amount of a given gas in a mixture to its partial pressure. For an ideal gas, we can write

The ratio n1/nt is called the mole fraction of gas 1, which we denote X1. The mole fraction, X, is a dimensionless number that expresses the ratio of the number of moles of one component in a mixture to the total number of moles in the mixture. Thus, for gas 1 we have
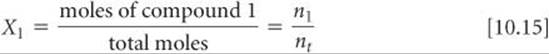
We can combine Equations 10.14 and 10.15 to give

The mole fraction of N2 in air is 0.78—that is, 78% of the molecules in air are N2 (![]() FIGURE 10.14). This means that if the barometric pressure is 760 torr, the partial pressure of N2 is
FIGURE 10.14). This means that if the barometric pressure is 760 torr, the partial pressure of N2 is
![]()
This result makes intuitive sense: Because N2 makes up 78% of the mixture, it contributes 78% of the total pressure.

![]() FIGURE 10.14 Air is approximately 78% nitrogen, 21% oxygen, plus a mixture of many other gases.
FIGURE 10.14 Air is approximately 78% nitrogen, 21% oxygen, plus a mixture of many other gases.
SAMPLE EXERCISE 10.11 Relating Mole Fractions and Partial Pressures
A study of the effects of certain gases on plant growth requires a synthetic atmosphere composed of 1.5 mol percent CO2, 18.0 mol percent O2, and 80.5 mol percent Ar. (a) Calculate the partial pressure of O2 in the mixture if the total pressure of the atmosphere is to be 745 torr. (b) If this atmosphere is to be held in a 121-L space at 295 K, how many moles of O2 are needed?
SOLUTION
Analyze For (a) we need to calculate the partial pressure of O2 given its mole percent and the total pressure of the mixture. For (b) we need to calculate the number of moles of O2 in the mixture given its volume (121 L), temperature (745 torr), and partial pressure from part (a).
Plan We calculate the partial pressures using Equation 10.16, and then use PO2, V, and T in the ideal-gas equation to calculate the number of moles of O2.
Solve
(a) The mole percent is the mole fraction times 100. Therefore, the mole fraction of O2 is 0.180. Equation 10.16 gives
PO2 = (0.180)(745 torr) = 134 torr
(b) Tabulating the given variables and converting to appropriate units, we have

Solving the ideal-gas equation for nO2, we have

Check The units check out, and the answer seems to be the right order of magnitude.
PRACTICE EXERCISE
From data gathered by Voyager 1, scientists have estimated the composition of the atmosphere of Titan, Saturn's largest moon. The pressure on the surface of Titan is 1220 torr. The atmosphere consists of 82 mol percent N2, 12 mol percent Ar, and 6.0 mol percent CH4. Calculate the partial pressure of each gas.
Answer: 1.0 × 103 torr N2, 1.5 × 102 torr Ar, and 73 torr CH4
Collecting Gases over Water
An experiment often run in general chemistry laboratories involves determining the number of moles of gas generated in a reaction. Sometimes the gas is collected over water. For example, solid potassium chlorate, KClO3, can be decomposed by heating it in the arrangement shown in ![]() FIGURE 10.15. The balanced equation for the reaction is
FIGURE 10.15. The balanced equation for the reaction is
![]()
The oxygen gas is collected in a bottle that is initially filled with water and inverted in a water pan.
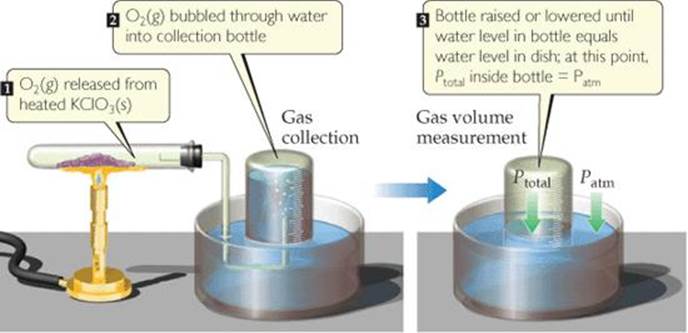
![]() FIGURE 10.15 Collecting a water-insoluble gas over water.
FIGURE 10.15 Collecting a water-insoluble gas over water.
Once the reaction is complete, the volume of gas collected is measured by raising or lowering the bottle as necessary until the water levels inside and outside the bottle are the same. When this condition is met, the pressure inside the bottle is equal to atmospheric pressure. The total pressure inside is the sum of the pressure of gas collected and the pressure of water vapor in equilibrium with liquid water:
![]()
Values for PH2O at various temperatures are listed in Appendix B.
SAMPLE EXERCISE 10.12 Calculating the Amount of Gas Collected over Water
When a sample of KClO3 is partially decomposed in the setup shown in Figure 10.15, the volume of gas collected is 0.250 L at 26 °C and 765 torr total pressure. (a) How many moles of O2 are collected? (b) How many grams of KClO3 were decomposed?
SOLUTION
(a) Analyze We need to calculate the number of moles of O2 gas in a container that also contains water vapor.
Plan We are given values for V and T. To use the ideal-gas equation to calculate the unknown, nO2, we must know the partial pressure of O2 in the system. We can calculate this partial pressure from the total pressure (765 torr) and the vapor pressure of water.
Solve The partial pressure of the O2 gas is the difference between the total pressure and the pressure of the water vapor at 26 °C, 25 torr (Appendix B):
PO2 = 765 torr – 25 torr = 740 torr
We use the ideal-gas equation to calculate the number of moles of O2:

(b) Analyze We need to calculate the number of moles of reactant KClO3 decomposed.
Plan We can use the number of moles of O2 formed and the balanced chemical equation to determine the number of moles of KClO3 decomposed, which we can then convert to grams of KClO3.
Solve From Equation 10.17, we have 2 mol KCl3≏ 3 mol O2. The molar mass of KClO3 is 122.6 g/mol. Thus, we can convert the number of moles of O2 from part (a) to moles of KClO3 and then to grams of KClO3:
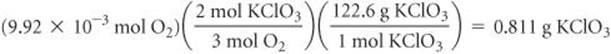
Check The units cancel appropriately in the calculations. The numbers of moles of O2 and KClO3 seem reasonable, given the small volume of gas collected.
PRACTICE EXERCISE
Ammonium nitrite, NH4NO2, decomposes on heating to form N2 gas:
![]()
When a sample of NH4NO2 is decomposed in the apparatus of Figure 10.15, 511 mL of N2 gas is collected over water at 26 °C and 745 torr total pressure. How many grams of NH4NO2 were decomposed?
Answer: 1.26 g