CHEMISTRY THE CENTRAL SCIENCE
10 GASES
CHAPTER SUMMARY AND KEY TERMS
SECTION 10.1 Substances that are gases at room temperature tend to be molecular substances with low molar masses. Air, a mixture composed mainly of N2 and O2, is the most common gas we encounter. Some liquids and solids can also exist in the gaseous state, where they are known asvapors. Gases are compressible; they mix in all proportions because their component molecules are far apart from each other.
SECTION 10.2 To describe the state or condition of a gas, we must specify four variables: pressure (P), volume (V), temperature (T), and quantity (n). Volume is usually measured in liters, temperature in kelvins, and quantity of gas in moles. Pressure is the force per unit area. It is expressed in SI units as pascals, Pa (1 Pa = 1 N/m2). lated unit, the bar, equals 105 Pa. In chemistry, standard atmospheric pressure is used to define the atmosphere (atm) and the torr (also called the millimeter of mercury). One atmosphere of pressure equals 101.325 kPa, or 760 torr. A barometer is often used to measure the atmospheric pressure. A manometer can be used to measure the pressure of enclosed gases.
SECTIONS 10.3 AND 10.4 Studies have revealed several simple gas laws: For a constant quantity of gas at constant temperature, the volume of the gas is inversely proportional to the pressure (Boyle's law). For a fixed quantity of gas at constant pressure, the volume is directly proportional to its absolute temperature (Charles's law). Equal volumes of gases at the same temperature and pressure contain equal numbers of molecules (Avogadro's hypothesis). For a gas at constant temperature and pressure, the volume of the gas is directly proportional to the number of moles of gas (Avogadro's law). Each of these gas laws is a special case of the ideal-gas equation.
The ideal-gas equation, PV = nRT, is the equation of state for an ideal gas. The term R in this equation is the gas constant. We can use the ideal-gas equation to calculate variations in one variable when one or more of the others are changed. Most gases at pressures less than 10 atm and temperatures near 273 K and above obey the ideal-gas equation reasonably well. The conditions of 273 K (0 °C) and 1 atm are known as the standard temperature and pressure (STP). In all applications of the ideal-gas equation we must remember to convert temperatures to the absolute-temperature scale (the Kelvin scale).
SECTIONS 10.5 AND 10.6 Using the ideal-gas equation, we can relate the density of a gas to its molar mass: M = dRT/P. We can also use the ideal-gas equation to solve problems involving gases as reactants or products in chemical reactions.
In gas mixtures the total pressure is the sum of the partial pressures that each gas would exert if it were present alone under the same conditions (Dalton's law of partial pressures). The partial pressure of a component of a mixture is equal to its mole fraction times the total pressure:P1 = X1Pt. The mole fraction is the ratio of the moles of one component of a mixture to the total moles of all components. In calculating the quantity of a gas collected over water, correction must be made for the partial pressure of water vapor in the gas mixture.
SECTION 10.7 The kinetic-molecular theory of gases accounts for the properties of an ideal gas in terms of a set of statements about the nature of gases. Briefly, these statements are as follows: Molecules are in continuous chaotic motion. The volume of gas molecules is negligible compared to the volume of their container. The gas molecules neither attract nor repel each other. The average kinetic energy of the gas molecules is proportional to the absolute temperature and does not change if the temperature remains constant.
The individual molecules of a gas do not all have the same kinetic energy at a given instant. Their speeds are distributed over a wide range; the distribution varies with the molar mass of the gas and with temperature. The root-mean-square (rms) speed, u rms, varies in proportion to the square root of the absolute temperature and inversely with the square root of the molar mass: ![]() . The most probable speed of a gas molecule is given by
. The most probable speed of a gas molecule is given by ![]() .
.
SECTION 10.8 It follows from kinetic-molecular theory that the rate at which a gas undergoes effusion (escapes through a tiny hole) is inversely proportional to the square root of its molar mass (Graham's law). The diffusion of one gas through the space occupied by a second gas is another phenomenon related to the speeds at which molecules move. Because molecules undergo frequent collisions with one another, the mean free path—the mean distance traveled between collisions—is short. Collisions between molecules limit the rate at which a gas molecule can diffuse.
SECTION 10.9 Departures from ideal behavior increase in magnitude as pressure increases and as temperature decreases. The extent of nonideality of a real gas can be seen by examining the quantity PV = RT for one mole of the gas as a function of pressure; for an ideal gas, this quantity is exactly 1 at all pressures. Real gases depart from ideal behavior because the molecules possess finite volume and because the molecules experience attractive forces for one another. The van der Waals equation is an equation of state for gases that modifies the ideal-gas equation to account for intrinsic molecular volume and intermolecular forces.
KEY SKILLS
• Convert between pressure units with an emphasis on torr and atmospheres. (Section 10.2)
• Calculate P, V, n, or T using the ideal-gas equation. (Section 10.4)
• Understand how the gas laws relate to the ideal-gas equation and apply the gas laws in calculations. (Sections 10.3 and 10.4)
• Calculate the density or molecular weight of a gas. (Section 10.5)
• Calculate the volume of gas consumed or formed in a chemical reaction. (Section 10.5)
• Calculate the total pressure of a gas mixture given its partial pressures or given information for calculating partial pressures. (Section 10.6)
• Describe the kinetic-molecular theory of gases and how it explains the pressure and temperature of a gas, the gas laws, and the rates of effusion and diffusion. (Sections 10.7 and 10.8)
• Explain why intermolecular attractions and molecular volumes cause real gases to deviate from ideal behavior at high pressure or low temperature. (Section 10.9)
KEY EQUATIONS
![]()
Ideal-gas equation

The combined gas law, showing how P, V, and T are related for a constant n
![]()
Calculating the density or molar mass of a gas
![]()
Relating the total pressure of a gas mixture to the partial pressures of its components (Dalton's law of partial pressures)
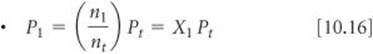
Relating partial pressure to mole fraction

Definition of the root-mean-square (rms) speed of gas molecules

Relating the relative rates of effusion of two gases to their molar masses