CHEMISTRY THE CENTRAL SCIENCE
10 GASES
EXERCISES
VISUALIZING CONCEPTS
10.1 Mars has an average atmospheric pressure of 0.007 atm. Would it be easier or harder to drink from a straw on Mars than on Earth? Explain. [Section 10.2]
10.2 You have a sample of gas in a container with a movable piston, such as the one in the drawing. (a) Redraw the container to show what it might look like if the temperature of the gas is increased from 300 K to 500 K while the pressure is kept constant. (b) Redraw the container to show what it might look like if the external pressure on the piston is increased from 1.0 atm to 2.0 atm while the temperature is kept constant. (c) Redraw the container to show what it might look like if the temperature of the gas decreases from 300 K to 200 K while the pressure is kept constant (assume the gas does not liquefy). [Section 10.3]

10.3 Consider the sample of gas depicted here. What would the drawing look like if the volume and temperature remained constant while you removed enough of the gas to decrease the pressure by a factor of 2? [Section 10.3]

10.4 Imagine that the reaction 2 CO(g) + O2(g) → 2 CO2(g) occurs in a container that has a piston that moves to maintain a constant pressure when the reaction occurs at constant temperature. (a) What happens to the volume of the container as a result of the reaction? Explain. (b) If the piston is not allowed to move, what happens to the pressure as a result of the reaction? [Sections 10.3 and 10.5]
10.5 Suppose you have a fixed amount of an ideal gas at a constant volume. If the pressure of the gas is doubled while the volume is held constant, what happens to its temperature? [Section 10.4]
10.6 The apparatus shown here has two gas-filled containers and one empty container, all attached to a hollow horizontal tube. When the valves are opened and the gases are allowed to mix at constant temperature, what is the distribution of atoms in each container? Assume that the containers are of equal volume and ignore the volume of the connecting tube. Which gas has the greater partial pressure after the valves are opened? [Section 10.6]
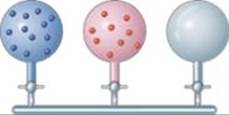
10.7 The accompanying drawing represents a mixture of three different gases. (a) Rank the three components in order of increasing partial pressure. (b) If the total pressure of the mixture is 1.40 atm, calculate the partial pressure of each gas. [Section 10.6]

10.8 On a single plot, qualitatively sketch the distribution of molecular speeds for (a) Kr(g) at –50 °C, (b) Kr(g) at 0 °C, (c) Ar(g) at 0 °C. [Section 10.7]
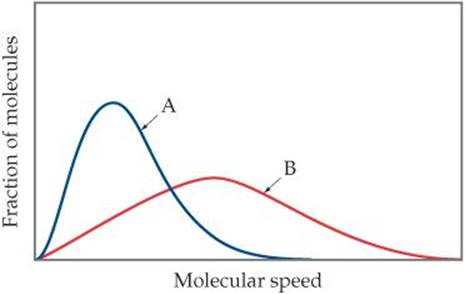
10.9 Consider the following graph. (a) If curves A and B refer to two different gases, He and O2, at the same temperature, which is which? Explain. (b) If A and B refer to the same gas at two different temperatures, which represents the higher temperature? (c) Redraw the graph and put in vertical lines that indicate the approximate positions of the most probable speeds and root-mean-square speeds for each curve. [Section 10.7]
10.10 Consider the following samples of gases:
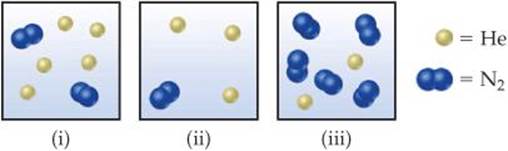
If the three samples are all at the same temperature, rank them with respect to (a) total pressure, (b) partial pressure of helium, (c) density, (d) average kinetic energy of particles. [Section 10.6 and 10.7]
10.11 A thin glass tube 1 m long is filled with Ar gas at 1 atm, and the ends are stoppered with cotton plugs:
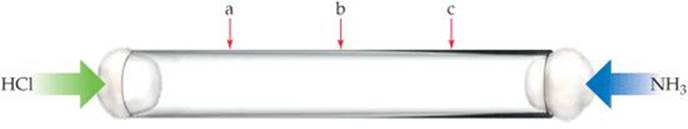
HCl gas is introduced at one end of the tube, and simultaneously NH3 gas is introduced at the other end. When the two gases diffuse through the cotton plugs down the tube and meet, a white ring appears due to the formation of NH4Cl(s). At which location—a, b, or c—do you expect the ring to form? Explain your choice. [Section 10.8]
10.12 Which substances in Table 10.3 would you expect to deviate most from ideal-gas behavior at low temperature and high pressure? Which would deviate least? Explain. [Section 10.9]
GAS CHARACTERISTICS; PRESSURE (sections 10.1 and 10.2)
10.13 How does a gas compare with a liquid for each of the following properties: (a) density, (b) compressibility, (c) ability to mix with other substances of the same phase to form homogeneous mixtures, (d) ability to conform to the shape of its container?
10.14 (a) A liquid and a gas are moved to larger containers. How does their behavior differ once they are in the larger containers? Explain the difference in molecular terms. (b) Although liquid water and carbon tetrachloride, CCl4(l), do not mix, their vapors form a homogeneous mixture. Explain. (c) Gas densities are generally reported in grams per liter, whereas liquid densities are reported in grams per milliliter. Explain the molecular basis for this difference.
______
10.15 Suppose that a woman weighing 130 lb and wearing high-heeled shoes momentarily places all her weight on the heel of one foot. If the area of the heel is 0.50 in.2, calculate the pressure exerted on the underlying surface in (a) kilopascals, (b) atmospheres, and (c) pounds per square inch.
10.16 A set of bookshelves rests on a hard floor surface on four legs, each having a cross-sectional dimension of 3.0 × 4.1 cm in contact with the floor. The total mass of the shelves plus the books stacked on them is 262 kg. Calculate the pressure in pascals exerted by the shelf footings on the surface.
______
10.17 (a) How high in meters must a column of water be to exert a pressure equal to that of a 760-mm column of mercury? The density of water is 1.0 g/mL, whereas that of mercury is 13.6 g/mL. (b) What is the pressure, in atmospheres, on the body of a diver if he is 39 ft below the surface of the water when atmospheric pressure at the surface is 0.97 atm?
10.18 The compound 1-iodododecane is a nonvolatile liquid with a density of 1.20 g/mL. The density of mercury is 13.6 g/mL. What do you predict for the height of a barometer column based on 1-iodododecane, when the atmospheric pressure is 749 torr?
______
10.19 Each of the following statements concerns a mercury barometer such as that shown in Figure 10.2. Identify any incorrect statements and correct them. (a) The tube must be 1cm2 in cross-sectional area. (b) At equilibrium the force of gravity per unit area acting on the mercury column equals the force of gravity per unit area acting on the atmosphere. (c) The column of mercury is held up by the vacuum at the top of the column. (d) If you took the mercury barometer with you on a trip from the beach to high mountains, the height of the mercury column would increase with elevation.
10.20 Suppose you make a mercury barometer using a glass tube about 50 cm in length, closed at one end. What would you expect to see if the tube is filled with mercury and inverted in a mercury dish, as in Figure 10.2? Explain.
______
10.21 The typical atmospheric pressure on top of Mt. Everest (29,028 ft) is about 265 torr. Convert this pressure to (a) atm, (b) mm Hg, (c) pascals, (d) bars, (e) psi.
10.22 Perform the following conversions: (a) 0.912 atm to torr, (b) 0.685 bar to kilopascals, (c) 655 mm Hg to atmospheres, (d) 1.323 × 105 Pa to atmospheres, (e) 2.50 atm to psi.
______
10.23 In the United States, barometric pressures are generally re ported in inches of mercury (in. Hg). On a beautiful summer day in Chicago the barometric pressure is 30.45 in. Hg. (a) Convert this pressure to torr. (b) Convert this pressure to atm. (c) A meteorologist explains the nice weather by referring to a “high-pressure area.” In light of your answer to parts (a) and (b), explain why this term makes sense.
10.24 Hurricane Wilma of 2005 is the most intense hurricane on record in the Atlantic basin, with a low-pressure reading of 882 mbar (millibars). Convert this reading into (a) atmospheres, (b) torr, and (c) inches of Hg.
______
10.25 If the atmospheric pressure is 0.995 atm, what is the pressure of the enclosed gas in each of the three cases depicted in the drawing? Assume that the gray liquid is mercury.
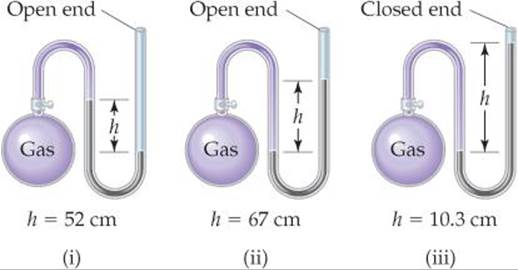
10.26 An open-end manometer containing mercury is connected to a container of gas, as depicted in Sample Exercise 10.2. What is the pressure of the enclosed gas in torr in each of the following situations? (a) The mercury in the arm attached to the gas is 15.4 mm higher than in the one open to the atmosphere; atmospheric pressure is 0.985 atm. (b) The mercury in the arm attached to the gas is 12.3 mm lower than in the one open to the atmosphere; atmospheric pressure is 0.99 atm.
THE GAS LAWS (section 10.3)
10.27 You have a gas confined to a cylinder with a movable piston. What would happen to the gas pressure inside the cylinder if you do the following? (a) Decrease the volume to one-fourth the original volume while holding the temperature constant. (b) Reduce the temperature (in kelvins) to half its original value while holding the volume constant. (c) Reduce the amount of gas to one-fourth while keeping the volume and temperature constant.
10.28 A fixed quantity of gas at 21 °C exhibits a pressure of 752 torr and occupies a volume of 5.12 L. (a) Calculate the volume the gas will occupy if the pressure is increased to 1.88 atm while the temperature is held constant. (b) Calculate the volume the gas will occupy if the temperature is increased to 175 °C while the pressure is held constant.
______
10.29 (a) How is the law of combining volumes explained by Avo-gadro's hypothesis? (b) Consider a 1.0-L flask containing neon gas and a 1.5-L flask containing xenon gas. Both gases are at the same pressure and temperature. According to Avogadro's law, what can be said about the ratio of the number of atoms in the two flasks? (c) Will 1 mol of an ideal gas always occupy the same volume at a given temperature and pressure? Explain.
10.30 Nitrogen and hydrogen gases react to form ammonia gas as follows:
N2(g) + 3 H2(g) → NH3(g)
At a certain temperature and pressure, 1.2 L of N2 reacts with 3.6 L of H2. If all the N2 and H2 are consumed, what volume of NH3, at the same temperature and pressure, will be produced?
THE IDEAL-GAS EQUATION (section 10.4)
10.31 (a) What is an ideal gas? (b) Show how Boyle's law, Charles's law, and Avogadro's law can be combined to give the ideal-gas equation. (c) Write the ideal-gas equation, and give the units used for each term when R = 0.08206 L-atm/mol-K. (d) If you measure pressure in bars instead of atmospheres, calculate the corresponding value of R in L-bar/mol-K.
10.32 (a) What conditions are represented by the abbreviation STP? (b) What is the molar volume of an ideal gas at STP? (c) Room temperature is often assumed to be 25 °C. Calculate the molar volume of an ideal gas at 25 °C and 1 atm pressure.
______
10.33 Suppose you are given two 1-L flasks and told that one contains a gas of molar mass 30, the other a gas of molar mass 60, both at the same temperature. The pressure in flask A is X atm, and the mass of gas in the flask is 1.2 g. The pressure in flask B is 0.5X atm, and the mass of gas in that flask is 1.2 g. Which flask contains the gas of molar mass 30, and which contains the gas of molar mass 60?
10.34 Suppose you are given two flasks at the same temperature, one of volume 2 L and the other of volume 3 L. The 2-L flask contains 4.8 g of gas, and the gas pressure is X atm. The 3-L flask contains 0.36 g of gas, and the gas pressure is 0.1X. Do the two gases have the same molar mass? If not, which contains the gas of higher molar mass?
______
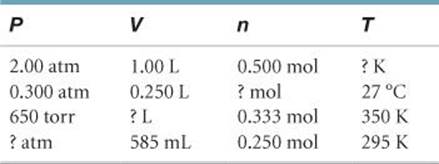
10.35 Complete the following table for an ideal gas:
10.36 Calculate each of the following quantities for an ideal gas: (a) the volume of the gas, in liters, if 1.50 mol has a pressure of 1.25 atm at a temperature of -6 °C; (b) the absolute temperature of the gas at which 3.33 × 10-3 mol occupies 478 mL at 750 torr; (c) the pressure, in atmospheres, if 0.00245 mol occupies 413 mL at 138 °C; (d) the quantity of gas, in moles, if 126.5 L at 54 °C has a pressure of 11.25 kPa.
______
10.37 The Goodyear blimps, which frequently fly over sporting events, hold approximately 175,000 ft3 of helium. If the gas is at 23 °C and 1.0 atm, what mass of helium is in a blimp?
10.38 A neon sign is made of glass tubing whose inside diameter is 2.5 cm and whose length is 5.5 m. If the sign contains neon at a pressure of 1.78 torr at 35 °C, how many grams of neon are in the sign? (The volume of a cylinder is πr2h.)
______
10.39 (a) Calculate the number of molecules in a deep breath of air whose volume is 2.25 L at body temperature, 37 °C, and a pressure of 735 torr. (b) The adult blue whale has a lung capacity of 5.0 × 103 L. Calculate the mass of air (assume an average molar mass 28.98 g/mol) contained in an adult blue whale's lungs at 0.0 °C and 1.00 atm, assuming the air behaves ideally.
10.40 (a) If the pressure exerted by ozone, O3, in the stratosphere is 3.0 × 10-3 atm and the temperature is 250 K, how many ozone molecules are in a liter? (b) Carbon dioxide makes up approximately 0.04% of Earth's atmosphere. If you collect a 2.0-L sample from the atmosphere at sea level (1.00 atm) on a warm day (27 °C), how many CO2 molecules are in your sample?
______
10.41 A scuba diver's tank contains 0.29 kg of O2 compressed into a volume of 2.3 L. (a) Calculate the gas pressure inside the tank at 9 °C. (b) What volume would this oxygen occupy at 26 °C and 0.95 atm?
10.42 An aerosol spray can with a volume of 250 mL contains 2.30 g of propane gas (C3H8) as a propellant. (a) If the can is at 23 °C, what is the pressure in the can? (b) What volume would the propane occupy at STP? (c) The can's label says that exposure to temperatures above 130 °F may cause the can to burst. What is the pressure in the can at this temperature?
______
10.43 Chlorine is widely used to purify municipal water supplies and to treat swimming pool waters. Suppose that the volume of a particular sample of Cl2 gas is 8.70 L at 895 torr and 24 °C. (a) How many grams of Cl2 are in the sample? (b) What volume will the Cl2 occupy at STP?(c) At what temperature will the volume be 15.00 L if the pressure is 8.76 × 102 torr? (d) At what pressure will the volume equal 5.00 L if the temperature is 58 °C?
10.44 Many gases are shipped in high-pressure containers. Consider a steel tank whose volume is 55.0 gallons that contains O2 gas at a pressure of 16,500 kPa at 23 °C. (a) What mass of O2 does the tank contain? (b) What volume would the gas occupy at STP? (c) At what temperature would the pressure in the tank equal 150.0 atm? (d) What would be the pressure of the gas, in kPa, if it were transferred to a container at 24 °C whose volume is 55.0 L?
______
10.45 In an experiment reported in the scientific literature, male cockroaches were made to run at different speeds on a miniature treadmill while their oxygen consumption was measured. In one hour the average cockroach running at 0.08 km/hr consumed 0.8 mL of O2 at 1 atm pressure and 24 °C per gram of insect mass. (a) How many moles of O2 would be consumed in 1 hr by a 5.2-g cockroach moving at this speed? (b) This same cockroach is caught by a child and placed in a 1-qt fruit jar with a tight lid. Assuming the same level of continuous activity as in the research, will the cockroach consume more than 20% of the available O2 in a 48-hr period? (Air is 21 mol percent O2.)
10.46 The physical fitness of athletes is measured by “VO2 max,” which is the maximum volume of oxygen consumed by an individual during incremental exercise (for example, on a treadmill). An average male has a VO2 max of 45 mL O2/kg body mass/min, but a world-class male athlete can have a VO2 max reading of 88.0 mL O2/kg body mass/min. (a) Calculate the volume of oxygen, in mL, consumed in 1 hr by an average man who weighs 185 lbs and has a VO2 max reading of 47.5 mL O2/kg body mass/min. (b) If this man lost 20 lb, exercised, and increased his VO2 max to 65.0 mL O2/kg body mass/min, how many mL of oxygen would he consume in 1 hr?
______
10.47 Mercury is a liquid at room temperature and pressure, but its vapor is present in the atmosphere from natural sources, such as volcanoes, and from human (“anthropogenic”) activities such as coal burning and gold mining. Elemental Hg becomes extremely toxic when oxidized to Hg(I) or Hg(II) compounds. (a) The Environmental Protection Agency has estimated that 119 tons of mercury are currently emitted to Earth's atmosphere annually due to human activities; this has decreased significantly from the mid-1970s before pollution controls were in place. What volume would the 119 tons of mercury occupy if it were a pure vapor at 1.00 atm and 298 K? (b) Geological records suggest that before the Industrial Revolution began in 1750, the baseline level of atmospheric Hg was 35 ppb (parts per billion, by volume, or 35 L Hg for every 109 L of air); current estimates are 245 ppb. For an estimated atmospheric volume of 51 × 1012 m3, calculate the number of moles Hg estimated to be in the atmosphere today.
10.48 After the large eruption of Mount St. Helens in 1980, gas samples from the volcano were taken by sampling the downwind gas plume. The unfiltered gas samples were passed over a gold-coated wire coil to absorb mercury (Hg) present in the gas. The mercury was recovered from the coil by heating it and then analyzed. In one particular set of experiments scientists found a mercury vapor level of 1800 ng of Hg per cubic meter in the plume at a gas temperature of 10 °C. Calculate (a) the partial pressure of Hg vapor in the plume, (b) the number of Hg atoms per cubic meter in the gas, (c) the total mass of Hg emitted per day by the volcano if the daily plume volume was 1600km3.
FURTHER APPLICATIONS OF THE IDEAL-GAS EQUATION (section 10.5)
10.49 Which gas is most dense at 1.00 atm and 298 K: CO2, N2O, or Cl2? Explain.
10.50 Rank the following gases from least dense to most dense at 1.00 atm and 298 K: SO2, HBr, CO2. Explain.
10.51 Which of the following statements best explains why a closed balloon filled with helium gas rises in air?
(a) Helium is a monatomic gas, whereas nearly all the molecules that make up air, such as nitrogen and oxygen, are diatomic.
(b) The average speed of helium atoms is higher than the average speed of air molecules, and the higher speed of collisions with the balloon walls propels the balloon upward.
(c) Because the helium atoms are of lower mass than the average air molecule, the helium gas is less dense than air. The mass of the balloon is thus less than the mass of the air displaced by its volume.
(d) Because helium has a lower molar mass than the average air molecule, the helium atoms are in faster motion. This means that the temperature of the helium is higher than the air temperature. Hot gases tend to rise.
10.52 Which of the following statements best explains why nitrogen gas at STP is less dense than Xe gas at STP?
(a) Because Xe is a noble gas, there is less tendency for the Xe atoms to repel one another, so they pack more densely in the gas state.
(b) Xe atoms have a higher mass than N2 molecules. Because both gases at STP have the same number of molecules per unit volume, the Xe gas must be denser.
(c) The Xe atoms are larger than N2 molecules and thus take up a larger fraction of the space occupied by the gas.
(d) Because the Xe atoms are much more massive than the N2 molecules, they move more slowly and thus exert less upward force on the gas container and make the gas appear denser.
______
10.53 (a) Calculate the density of NO2 gas at 0.970 atm and 35 °C. (b) Calculate the molar mass of a gas if 2.50 g occupies 0.875 L at 685 torr and 35 °C.
10.54 (a) Calculate the density of sulfur hexafluoride gas at 707 torr and 21 °C. (b) Calculate the molar mass of a vapor that has a density of 7.135 g/L at 12 °C and 743 torr.
______
10.55 In the Dumas-bulb technique for determining the molar mass of an unknown liquid, you vaporize the sample of a liquid that boils below 100 °C in a boiling-water bath and determine the mass of vapor required to fill the bulb (see drawing, next page). From the following data, calculate the molar mass of the unknown liquid: mass of unknown vapor, 1.012 g; volume of bulb, 354cm3; pressure, 742 torr; temperature, 99 °C.
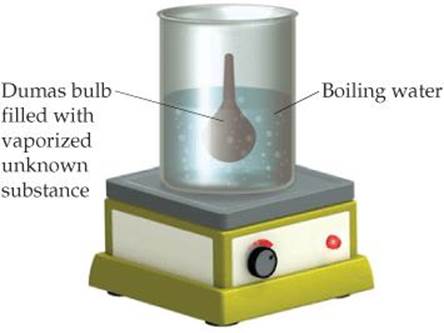
10.56 The molar mass of a volatile substance was determined by the Dumas-bulb method described in Exercise 10.55. The unknown vapor had a mass of 0.846 g; the volume of the bulb was 354 cm3, pressure 752 torr, and temperature 100 °C. Calculate the molar mass of the unknown vapor.
______
10.57 Magnesium can be used as a “getter” in evacuated enclosures to react with the last traces of oxygen. (The magnesium is usually heated by passing an electric current through a wire or ribbon of the metal.) If an enclosure of 0.452 L has a partial pressure of O2 of 3.5 × 10-6 torr at 27 °C, what mass of magnesium will react according to the following equation?
2 Mg(s) + O2(g) → MgO(s)
10.58 Calcium hydride, CaH2, reacts with water to form hydrogen gas:
CaH2(s) + 2 H2O(l) → Ca(OH)2(aq) + 2 H2(g)
This reaction is sometimes used to inflate life rafts, weather balloons, and the like, when a simple, compact means of generating H2 is desired. How many grams of CaH2 are needed to generate 145 L of H2 gas if the pressure of H2 is 825 torr at 21 °C?
______
10.59 The metabolic oxidation of glucose, C6H12O6, in our bodies produces CO2, which is expelled from our lungs as a gas:
C6H12O6(aq) + 6 CO2(g) → 6 CO2(g) + 6 H2O(l)
(a) Calculate the volume of dry CO2 produced at body temperature (37 °C) and 0.970 atm when 24.5 g of glucose is consumed in this reaction. (b) Calculate the volume of oxygen you would need, at 1.00 atm and 298 K, to completely oxidize 50.0 g of glucose.
10.60 Both Jacques Charles and Joseph Louis Guy-Lussac were avid balloonists. In his original flight in 1783, Jacques Charles used a balloon that contained approximately 31,150 L of H2. He generated the H2 using the reaction between iron and hy drochloric acid:
Fe(s) + 2 HCl(aq) → FeCl2(aq) + H2(g)
How many kilograms of iron were needed to produce this volume of H2 if the temperature was 22 °C?
______
10.61 Hydrogen gas is produced when zinc reacts with sulfuric acid:
Zn(s) + H2SO4(aq) ZnSO4(aq) + H2(g)
If 159 mL of wet H2 is collected over water at 24 °C and a barometric pressure of 738 torr, how many grams of Zn have been consumed? (The vapor pressure of water is tabulated in Appendix B.)
10.62 Acetylene gas, C2H2(g), can be prepared by the reaction of calcium carbide with water:
CaC2(s) + 2 H2O(l) → Ca(OH)2(s) + C2H2(g)
Calculate the volume of C2H2 that is collected over water at 23 °C by reaction of 1.524 g of CaC2 if the total pressure of the gas is 753 torr. (The vapor pressure of water is tabulated in Appendix B.)
PARTIAL PRESSURES (section 10.6)
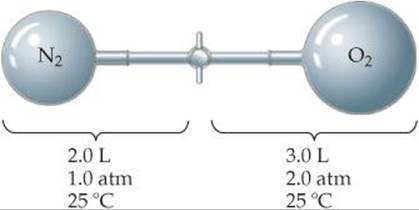
10.63 Consider the apparatus shown in the following drawing. (a) When the valve between the two containers is opened and the gases allowed to mix, how does the volume occupied by the N2 gas change? What is the partial pressure of N2 after mixing? (b) How does the volume of the O2 gas change when the gases mix? What is the partial pressure of O2 in the mixture? (c) What is the total pressure in the container after the gases mix?
10.64 Consider a mixture of two gases, A and B, confined in a closed vessel. A quantity of a third gas, C, is added to the same vessel at the same temperature. How does the addition of gas C affect the following: (a) the partial pressure of gas A, (b) the total pressure in the vessel, (c) the mole fraction of gas B?
______
10.65 A mixture containing 0.765 mol He(g), 0.330 mol Ne(g), and 0.110 mol Ar(g) is confined in a 10.00-L vessel at 25 °C. (a) Calculate the partial pressure of each of the gases in the mixture. (b) Calculate the total pressure of the mixture.
10.66 A deep-sea diver uses a gas cylinder with a volume of 10.0 L and a content of 51.2 g of O2 and 32.6 g of He. Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 19 °C
______
10.67 The atmospheric concentration of CO2 gas is presently 390 ppm (parts per million, by volume; that is, 390 L of every 106 L of the atmosphere are CO2). What is the mole fraction of CO2 in the atmosphere?
10.68 A plasma-screen TV contains thousands of tiny cells filled with a mixture of Xe, Ne, and He gases that emits light of specific wavelengths when a voltage is applied. A particular plasma cell, 0.900 mm × 0.300 mm × 10.0 mm, contains 4% Xe in a 1:1 Ne:He mixture at a total pressure of 500 torr. Calculate the number of Xe, Ne, and He atoms in the cell and state the assumptions you need to make in your calculation.
______
10.69 A piece of dry ice (solid carbon dioxide) with a mass of 5.50 g is placed in a 10.0-L vessel that already contains air at 705 torr and 24 °C. After the carbon dioxide has totally vaporized, what is the partial pressure of carbon dioxide and the total pressure in the container at 24 °C?
10.70 A sample of 5.00 mL of diethylether (C2H5OC2H5, density = 0.7134 g/mL) is introduced into a 6.00-L vessel that already contains a mixture of N2 and O2, whose partial pressures are PN2 = 0.751 atm and PO2 = 0.208 atm. The temperature is held at 35.0 °C, and the diethylether totally evaporates. (a) Calculate the partial pressure of the diethylether. (b) Calculate the total pressure in the container.
______
10.71 A mixture of gases contains 0.75 mol N2, 0.30 mol O2, and 0.15 mol CO2. If the total pressure of the mixture is 2.15 atm, what is the partial pressure of each component?
10.72 A mixture of gases contains 10.25 g of N2, 1.83 g of H2, and 7.95 g of NH3. If the total pressure of the mixture is 1.85 atm, what is the partial pressure of each component?
______
10.73 At an underwater depth of 250 ft, the pressure is 8.38 atm. What should the mole percent of oxygen be in the diving gas for the partial pressure of oxygen in the mixture to be 0.21 atm, the same as in air at 1 atm?
10.74 (a) What are the mole fractions of each component in a mixture of 15.08 g of O2, 8.17 g of N2, and 2.64 g of H2? (b) What is the partial pressure in atm of each component of this mixture if it is held in a 15.50-L vessel at 15 °C?
______
10.75 A quantity of N2 gas originally held at 5.25 atm pressure in a 1.00-L container at 26 °C is transferred to a 12.5-L container at 20 °C. A quantity of O2 gas originally at 5.25 atm and 26 °C in a 5.00-L container is transferred to this same container. What is the total pressure in the new container?
10.76 A sample of 3.00 g of SO2(g) originally in a 5.00-L vessel at 21 °C is transferred to a 10.0-L vessel at 26 °C. A sample of 2.35 g N2(g) originally in a 2.50-L vessel at 20 °C is transferred to this same 10.0-L vessel. (a) What is the partial pressure of SO2(g) in the larger container?(b) What is the partial pressure of N2(g) in this vessel? (c) What is the total pressure in the vessel?
KINETIC-MOLECULAR THEORY OF GASES; EFFUSION AND DIFFUSION (sections 10.7 and 10.8)
10.77 What change or changes in the state of a gas bring about each of the following effects? (a) The number of impacts per unit time on a given container wall increases. (b) The average energy of impact of molecules with the wall of the container decreases. (c) The average distance between gas molecules increases. (d) The average speed of molecules in the gas mixture is increased.
10.78 Indicate which of the following statements regarding the kinetic-molecular theory of gases are correct. For those that are false, formulate a correct version of the statement. (a) The average kinetic energy of a collection of gas molecules at a given temperature is proportional to m½.(b) The gas molecules are assumed to exert no forces on each other. (c) All the molecules of a gas at a given temperature have the same kinetic energy. (d) The volume of the gas molecules is negligible in comparison to the total volume in which the gas is contained. (e) All gas molecules move with the same speed if they are at the same temperature.
______
10.79 What property or properties of gases can you point to that support the assumption that most of the volume in a gas is empty space?
10.80 Newton had an incorrect theory of gases in which he assumed that all gas molecules repel one another and the walls of their container. Thus, the molecules of a gas are statically and uniformly distributed, trying to get as far apart as possible from one another and the vessel walls. This repulsion gives rise to pressure. Explain why Charles's law argues for the kinetic-molecular theory and against Newton's model.
______
10.81 Explain the difference between average speed and root-mean-square speed. Which is larger for a given gas sample at a fixed temperature?
[10.82] You have an evacuated container of fixed volume and known mass and introduce a known mass of a gas sample. Measuring the pressure at constant temperature over time, you are surprised to see it slowly dropping. You measure the mass of the gas-filled container and find that the mass is what it should be—gas plus container—and the mass does not change over time, so you do not have a leak. Suggest an explanation for your observations.
______
10.83 The temperature of a 5.00-L container of N2 gas is increased from 20 °C to 250 °C. If the volume is held constant, predict qualitatively how this change affects the following: (a) the average kinetic energy of the molecules; (b) the root-mean-square speed of the molecules; (c) the strength of the impact of an average molecule with the container walls; (d) the total number of collisions of molecules with walls per second.
10.84 Suppose you have two 1-L flasks, one containing N2 at STP, the other containing CH4 at STP. How do these systems compare with respect to (a) number of molecules, (b) density, (c) average kinetic energy of the molecules, (d) rate of effusion through a pinhole leak?
______
10.85 (a) Place the following gases in order of increasing average molecular speed at 25 °C: Ne, HBr, SO2, NF3, CO. (b) Calculate the rms speed of NF3 molecules at 25 °C. (c) Calculate the most probable speed of an ozone molecule in the stratosphere, where the temperature is 270 K.
10.86 (a) Place the following gases in order of increasing average molecular speed at 300 K: CO, SF6, H2S, Cl2, HBr. (b) Calculate and compare the rms speeds of CO and Cl2 molecules at 300 K. (c) Calculate and compare the most probable speeds of CO and Cl2 molecules at 300 K.
______
10.87 Explain the difference between effusion and diffusion.
[10.88] At constant pressure, the mean free path (λ) of a gas molecule is directly proportional to temperature. At constant temperature, λ is inversely proportional to pressure. If you compare two different gas molecules at the same temperature and pressure, λ is inversely proportional to the square of the diameter of the gas molecules. Put these facts together to create a formula for the mean free path of a gas molecule with a proportionality constant (call it Rmfp, like the ideal-gas constant) and define units for Rmfp.
______
10.89 Hydrogen has two naturally occurring isotopes, 1H and 2H. Chlorine also has two naturally occurring isotopes, 35Cl and 37Cl. Thus, hydrogen chloride gas consists of four distinct types of molecules: 1H35Cl, 1H37Cl, 2H35Cl, and 2H37Cl. Place these four molecules in order of increasing rate of effusion.
10.90 As discussed in the “Chemistry Put to Work” box in Section 10.8, enriched uranium can be produced by gaseous diffusion of UF6. Suppose a process were developed to allow diffusion of gaseous uranium atoms, U(g). Calculate the ratio of diffusion rates for 235U and 238U, and compare it to the ratio for UF6 given in the essay.
______
10.91 Arsenic(III) sulfide sublimes readily, even below its melting point of 320 °C. The molecules of the vapor phase are found to effuse through a tiny hole at 0.28 times the rate of effusion of Ar atoms under the same conditions of temperature and pressure. What is the molecular formula of arsenic(III) sulfide in the gas phase?
10.92 A gas of unknown molecular mass was allowed to effuse through a small opening under constant-pressure conditions. It required 105 s for 1.0 L of the gas to effuse. Under identical experimental conditions it required 31 s for 1.0 L of O2 gas to effuse. Calculate the molar mass of the unknown gas. (Remember that the faster the rate of effusion, the shorter the time required for effusion of 1.0 L; that is, rate and time are inversely proportional.)
NONIDEAL-GAS BEHAVIOR (section 10.9)
10.93 (a) List two experimental conditions under which gases deviate from ideal behavior. (b) List two reasons why the gases deviate from ideal behavior. (c) Explain how the function PV/RT can be used to show how gases behave nonideally.
10.94 The planet Jupiter has a surface temperature of 140 K and a mass 318 times that of Earth. Mercury (the planet) has a surface temperature between 600 K and 700 K and a mass 0.05 times that of Earth. On which planet is the atmosphere more likely to obey the ideal-gas law? Explain.
______
10.95 Based on their respective van der Waals constants (Table 10.3), is Ar or CO2 expected to behave more nearly like an ideal gas at high pressures? Explain.
10.96 Briefly explain the significance of the constants a and b in the van der Waals equation.
10.97 In Sample Exercise 10.16, we found that one mole of Cl2 confined to 22.41 L at 0 °C deviated slightly from ideal behavior. Calculate the pressure exerted by 1.00 mol Cl2 confined to a smaller volume, 5.00 L, at 25 °C. (a) First use the ideal-gas equation and (b) then use the van der Waals equation for your calculation. (Values for the van der Waals constants are given in Table 10.3.) (c) Why is the difference between the result for an ideal gas and that calculated using the van der Waals equation greater when the gas is confined to 5.00 L compared to 22.4 L?
10.98 Calculate the pressure that CCl4 will exert at 40 °C if 1.00 mol occupies 33.3 L, assuming that (a) CCl4 obeys the ideal-gas equation; (b) CCl4 obeys the van der Waals equation. (Values for the van der Waals constants are given in Table 10.3.) (c) Which would you expect to deviate more from ideal behavior under these conditions, Cl2 or CCl4? Explain.
______
[10.99] Table 10.3 shows that the van der Waals b parameter has units of L/mol. This implies that we can calculate the size of atoms or molecules from b. Using the value of b for Xe, calculate the radius of a Xe atom and compare it to the value found in Figure 7.6, 1.30 Å. Recall that the volume of a sphere is (4/3)πr3.
[10.100] Table 10.3 shows that the van der Waals b parameter has units of L/mol. This means that we can calculate the sizes of atoms or molecules from the b parameter. Refer back to the discussion in Section 7.3. Is the van der Waals radius we calculate from the b parameter of Table 10.3 more closely associated with the bonding or nonbonding atomic radius discussed there? Explain.
ADDITIONAL EXERCISES
10.101 A gas bubble with a volume of 1.0 mm3 originates at the bottom of a lake where the pressure is 3.0 atm. Calculate its volume when the bubble reaches the surface of the lake where the pressure is 730 torr, assuming that the temperature doesn't change.
10.102 A 15.0-L tank is filled with helium gas at a pressure of 1.00 × 102 atm. How many balloons (each 2.00 L) can be inflated to a pressure of 1.00 atm, assuming that the temperature remains constant and that the tank cannot be emptied below 1.00 atm?
10.103 To minimize the rate of evaporation of the tungsten filament, 1.4 × 10–5 mol of argon is placed in a 600-cm3 lightbulb. What is the pressure of argon in the lightbulb at 23 °C?
10.104 Carbon dioxide, which is recognized as the major contributor to global warming as a “greenhouse gas,” is formed when fossil fuels are combusted, as in electrical power plants fueled by coal, oil, or natural gas. One potential way to reduce the amount of CO2 added to the atmosphere is to store it as a compressed gas in underground formations. Consider a 1000-megawatt coal-fired power plant that produces about 6 × 106 tons of CO2 per year. (a) Assuming ideal-gas behavior, 1.00 atm, and 27 °C, calculate the volume of CO2 produced by this power plant. (b) If the CO2 is stored underground as a liquid at 10 °C and 120 atm and a density of 1.2 g/cm3, what volume does it possess? (c) If it is stored underground as a gas at 36 °C and 90 atm, what volume does it occupy?
10.105 Propane, C3H8, liquefies under modest pressure, allowing a large amount to be stored in a container. (a) Calculate the number of moles of propane gas in a 110-L container at 3.00 atm and 27 °C. (b) Calculate the number of moles of liquid propane that can be stored in the same volume if the density of the liquid is 0.590 g/mL. (c) Calculate the ratio of the number of moles of liquid to moles of gas. Discuss this ratio in light of the kinetic-molecular theory of gases.
[10.106] Nickel carbonyl, Ni(CO)4, is one of the most toxic substances known. The present maximum allowable concentration in laboratory air during an 8-hr workday is 1 ppb (parts per billion) by volume, which means that there is one mole of Ni(CO)4 for every 109 moles of gas. Assume 24 °C and 1.00 atm pressure. What mass of Ni(CO)4 is allowable in a laboratory room that is 12 ft × 20 ft × 9 ft?
10.107 When a large evacuated flask is filled with argon gas, its mass increases by 3.224 g. When the same flask is again evacuated and then filled with a gas of unknown molar mass, the mass increase is 8.102 g. (a) Based on the molar mass of argon, estimate the molar mass of the unknown gas. (b) What assumptions did you make in arriving at your answer?
10.108 Consider the arrangement of bulbs shown in the drawing. Each of the bulbs contains a gas at the pressure shown. What is the pressure of the system when all the stopcocks are opened, assuming that the temperature remains constant? (We can neglect the volume of the capillary tubing connecting the bulbs.)
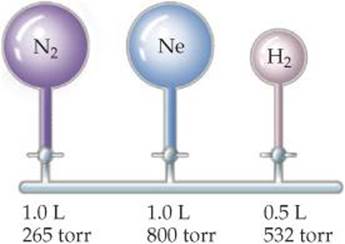
10.109 Assume that a single cylinder of an automobile engine has a volume of 524cm3. (a) If the cylinder is full of air at 74 °C and 0.980 atm, how many moles of O2 are present? (The mole fraction of O2 in dry air is 0.2095.) (b) How many grams of C8H18 could be combusted by this quantity of O2, assuming complete combustion with formation of CO2 and H2O?
10.110 Assume that an exhaled breath of air consists of 74.8% N2, 15.3% O2, 3.7% CO2, and 6.2% water vapor. (a) If the total pressure of the gases is 0.985 atm, calculate the partial pressure of each component of the mixture. (b) If the volume of the exhaled gas is 455 mL and its temperature is 37 °C, calculate the number of moles of CO2 exhaled. (c) How many grams of glucose (C6H12O6) would need to be metabolized to produce this quantity of CO2? (The chemical reaction is the same as that for combustion of C6H12O6. See Section 3.2 and Problem 10.59.)
10.111 A 1.42-g sample of helium and an unknown mass of O2 are mixed in a flask at room temperature. The partial pressure of the helium is 42.5 torr, and that of the oxygen is 158 torr. What is the mass of the oxygen?
10.112 A gaseous mixture of O2 and Kr has a density of 1.104 g/L at 355 torr and 400 K. What is the mole percent O2 in the mixture?
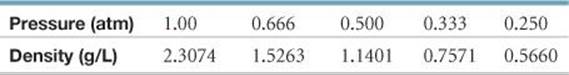
10.113 The density of a gas of unknown molar mass was measured as a function of pressure at 0 °C, as in the table that follows. (a) Determine a precise molar mass for the gas. [Hint: Graph d/P versus P.] (b) Why is d/P not a constant as a function of pressure?
10.114 A glass vessel fitted with a stopcock valve has a mass of 337.428 g when evacuated. When filled with Ar, it has a mass of 339.854 g. When evacuated and refilled with a mixture of Ne and Ar, under the same conditions of temperature and pressure, it has a mass of 339.076 g. What is the mole percent of Ne in the gas mixture?
10.115 You have a sample of gas at -33 °C. You wish to increase the rms speed by a factor of 2. To what temperature should the gas be heated?
10.116 Consider the following gases, all at STP: Ne, SF6, N2, CH4. (a) Which gas is most likely to depart from the assumption of the kinetic-molecular theory that says there are no attractive or repulsive forces between molecules? (b) Which one is closest to an ideal gas in its behavior?(c) Which one has the highest root-mean-square molecular speed at a given temperature? (d) Which one has the highest total molecular volume relative to the space occupied by the gas? (e) Which has the highest average kinetic-molecular energy? (f) Which one would effuse more rapidly than N2? (g) Which one would have the largest van der Waals b parameter?
10.117 Does the effect of intermolecular attraction on the properties of a gas become more significant or less significant if (a) the gas is compressed to a smaller volume at constant temperature; (b) the temperature of the gas is increased at constant volume?
10.118 Which of the noble gases other than radon would you expect to depart most readily from ideal behavior? Use the density data in Table 7.8 to show evidence in support of your answer.
10.119 It turns out that the van der Waals constant b equals four times the total volume actually occupied by the molecules of a mole of gas. Using this figure, calculate the fraction of the volume in a container actually occupied by Ar atoms (a) at STP, (b) at 200 atm pressure and 0 °C. (Assume for simplicity that the ideal-gas equation still holds.)
[10.120] Large amounts of nitrogen gas are used in the manufacture of ammonia, principally for use in fertilizers. Suppose 120.00 kg of N2(g) is stored in a 1100.0-L metal cylinder at 280 °C. (a) Calculate the pressure of the gas, assuming ideal-gas behavior. (b) By using data in Table 10.3, calculate the pressure of the gas according to the van der Waals equation. (c) Under the conditions of this problem, which correction dominates, the one for finite volume of gas molecules or the one for attractive interactions?
INTEGRATIVE EXERCISES
10.121 Cyclopropane, a gas used with oxygen as a general anesthetic, is composed of 85.7% C and 14.3% H by mass. (a) If 1.56 g of cyclopropane has a volume of 1.00 L at 0.984 atm and 50.0 °C, what is the molecular formula of cyclopropane? (b) Judging from its molecular formula, would you expect cyclopropane to deviate more or less than Ar from ideal-gas behavior at moderately high pressures and room temperature? Explain. (c) Would cyclopropane effuse through a pinhole faster or more slowly than methane, CH4?
[10.122] Consider the combustion reaction between 25.0 mL of liquid methanol (density = 0.850 g/mL) and 12.5 L of oxygen gas measured at STP. The products of the reaction are CO2(g) and H2O(g). Calculate the volume of liquid H2O formed if the reaction goes to completion and you condense the water vapor.
10.123 An herbicide is found to contain only C, H, N, and Cl. The complete combustion of a 100.0-mg sample of the herbicide in excess oxygen produces 83.16 mL of CO2 and 73.30 mL of H2O vapor at STP. A separate analysis shows that the sample also contains 16.44 mg of Cl. (a)Determine the percent composition of the substance. (b) Calculate its empirical formula. (c) What other information would you need to know about this compound to calculate its true molecular formula?
10.124 A 4.00-g sample of a mixture of CaO and BaO is placed in a 1.00-L vessel containing CO2 gas at a pressure of 730 torr and a temperature of 25 °C. The CO2 reacts with the CaO and BaO, forming CaCO3 and BaCO3. When the reaction is complete, the pressure of the remaining CO2 is 150 torr. (a) Calculate the number of moles of CO2 that have reacted. (b) Calculate the mass percentage of CaO in the mixture.
[10.125] Ammonia and hydrogen chloride react to form solid ammonium chloride:
NH3(g) + HCl(g) → NH4Cl(s)
Two 2.00-L flasks at 25 °C are connected by a valve, as shown in the drawing on the next page. One flask contains 5.00 g NH3(g), and the other contains 5.00 g HCl(g). When the valve is opened, the gases react until one is completely consumed. (a) Which gas will remain in the system after the reaction is complete? (b) What will be the final pressure of the system after the reaction is complete? (Neglect the volume of the ammonium chloride formed.) (c) What mass of ammonium chloride will be formed?

10.126 The “Chemistry Put to Work” box on pipelines in Section 10.5 mentions that the total deliverability of natural gas (methane, CH4) to the various regions of the United States is on the order of 2.7 × 1012 L per day, measured at STP. Calculate the total enthalpy change for combustion of this quantity of methane. (Note: Less than this amount of methane is actually combusted daily. Some of the delivered gas is passed through to other regions.)
10.127 Chlorine dioxide gas (ClO2) is used as a commercial bleaching agent. It bleaches materials by oxidizing them. In the course of these reactions, the ClO2 is itself reduced. (a) What is the Lewis structure for ClO2? (b) Why do you think that ClO2 is reduced so readily? (c) When a ClO2 molecule gains an electron, the chloriteion, ClO2, forms. Draw the Lewis structure for ClO2. (d) Predict the O —Cl — O bond angle in the ClO2 ion. (e) One method of preparing ClO2 is by the reaction of chlorine and sodium chlorite:
Cl2(g) + 2 NaClO2(s) → 2 ClO2(g) + 2 NaCl(s)
If you allow 15.0 g of NaClO2 to react with 2.00 L of chlorine gas at a pressure of 1.50 atm at 21 °C, how many grams of ClO2 can be prepared?
10.128 Natural gas is very abundant in many Middle Eastern oil fields. However, the costs of shipping the gas to markets in other parts of the world are high because it is necessary to liquefy the gas, which is mainly methane and has a boiling point at atmospheric pressure of —164 °C. One possible strategy is to oxidize the methane to methanol, CH3OH, which has a boiling point of 65 °C and can therefore be shipped more readily. Suppose that 10.7 × 109 ft3 of methane at atmospheric pressure and 25 °C is oxidized to methanol. (a) What volume of methanol is formed if the density of CH3OH is 0.791 g/mL? (b) Write balanced chemical equations for the oxidations of methane and methanol to CO2(g) and H2O(l). Calculate the total enthalpy change for complete combustion of the 10.7 × 109 ft3 of methane just described and for complete combustion of the equivalent amount of methanol, as calculated in part (a). (c) Methane, when liquefied, has a density of 0.466 g/mL; the density of methanol at 25 °C is 0.791 g/mL. Compare the enthalpy change upon combustion of a unit volume of liquid methane and liquid methanol. From the standpoint of energy production, which substance has the higher enthalpy of combustion per unit volume?
[10.129] Gaseous iodine pentafluoride, IF5, can be prepared by the reaction of solid iodine and gaseous fluorine:
I2(s) + 5 F2(g) → 2 IF5(g)
A 5.00-L flask containing 10.0 g I2 is charged with 10.0 g F2, and the reaction proceeds until one of the reagents is completely consumed. After the reaction is complete, the temperature in the flask is 125 °C. (a) What is the partial pressure of IF5 in the flask? (b) What is the mole fraction of IF5 in the flask (c) Draw the Lewis structure of IF5. (d) What is the total mass of reactants and products in the flask?
[10.130] A 6.53-g sample of a mixture of magnesium carbonate and calcium carbonate is treated with excess hydrochloric acid. The resulting reaction produces 1.72 L of carbon dioxide gas at 28 °C and 743 torr pressure. (a) Write balanced chemical equations for the reactions that occur between hydrochloric acid and each component of the mixture. (b) Calculate the total number of moles of carbon dioxide that forms from these reactions. (c) Assuming that the reactions are complete, calculate the percentage by mass of magnesium carbonate in the mixture.