CHEMISTRY THE CENTRAL SCIENCE
11 LIQUIDS AND INTERMOLECULAR FORCES

BECAUSE THE LEAVES OF THE LOTUS PLANT are highly water repellent, any water on a leaf beads up to minimize contact with the leaf surface.
WHAT'S AHEAD
11.1 A MOLECULAR COMPARISON OF GASES, LIQUIDS, AND SOLIDS
We begin with a comparison of solids, liquids, and gases from a molecular perspective. This comparison reveals the important roles that temperature and intermolecular forces play in determining the physical state of a substance.
11.2 INTERMOLECULAR FORCES
We then examine four intermolecular forces: dispersion forces, dipole–dipole forces, hydrogen bonds, and ion–dipole forces.
11.3 SELECT PROPERTIES OF LIQUIDS
We learn that the nature and strength of the intermolecular forces between molecules are largely responsible for many properties of liquids, including viscosity and surface tension.
11.4 PHASE CHANGES
We explore phase changes—the transitions of matter between the gaseous, liquid, and solid states—and their associated energies.
11.5 VAPOR PRESSURE
We examine the dynamic equilibrium that exists between a liquid and its gaseous state and introduce vapor pressure.
11.6 PHASE DIAGRAMS
We learn how to read phase diagrams, which are graphic representations of the equilibria among the gaseous, liquid, and solid phases.
11.7 LIQUID CRYSTALS
We learn about substances that pass into a liquid crystalline phase, which is an intermediate phase between the solid and liquid states. A substance in the liquid crystalline phase has some of the structural order of a solid and some of the freedom of motion of a liquid.
THE LOTUS PLANT GROWS in aquatic environments. In order to thrive in such an environment the surface of a lotus leaf is highly water repellent. Scientists call surfaces with this property “superhydrophobic.” The superhydrophobic character of the lotus leaf not only allows it to float on water but also causes any water that falls on the leaf to bead up and roll off. The water drops collect dirt as they roll off, keeping the leaf clean, even in the muddy ponds and lakes where lotus plants tend to grow. Because of its self-cleaning properties, the lotus plant is considered a symbol of purity in many Eastern cultures.
What forces cause the lotus leaf to repel water so efficiently? Although this plant's self-cleaning nature has been known for millennia, the effect was not fully understood until the 1970s when scanning electron microscopy images revealed a rough, one might say mountainous, leaf surface (![]() FIGURE 11.1). The rough surface helps minimize contact between water and leaf.
FIGURE 11.1). The rough surface helps minimize contact between water and leaf.
Another important factor contributing to the plant's self-cleaning nature is the contrast between the molecular composition of the leaf and that of the water. The leaf is coated by hydrocarbon molecules that are held together by forces that are different from those that hold the water molecules together. As a result the water molecules preferentially surround themselves with other water molecules, thereby minimizing their contact with the surface.
The lotus effect has inspired scientists to design superhydrophobic surfaces for applications such as self-cleaning windows and water-repellent clothing. To understand the lotus effect and other phenomena involving liquids and solids, we must understand intermolecular forces, the forces that exist between molecules. Only by understanding the nature and strength of these forces can we understand how the composition and structure of a substance are related to its physical properties in the liquid or solid state.

![]() FIGURE 11.1 A microscopic view of a water droplet on the surface of a lotus leaf.
FIGURE 11.1 A microscopic view of a water droplet on the surface of a lotus leaf.
11.1 A MOLECULAR COMPARISON OF GASES, LIQUIDS, AND SOLIDS
As we learned in Chapter 10, the molecules in a gas are widely separated and in a state of constant, chaotic motion. One of the key tenets of kinetic-molecular theory is the assumption that we can neglect the interactions between molecules. ![]() (Section 10.7) The properties of liquids and solids are quite different from gases largely because the intermolecular forces in liquids and solids are much stronger. A comparison of the properties of gases, liquids, and solids is given in
(Section 10.7) The properties of liquids and solids are quite different from gases largely because the intermolecular forces in liquids and solids are much stronger. A comparison of the properties of gases, liquids, and solids is given in ![]() TABLE 11.1.
TABLE 11.1.
In liquids the intermolecular attractive forces are strong enough to hold particles close together. Thus, liquids are much denser and far less compressible than gases. Unlike gases, liquids have a definite volume, independent of the size and shape of their container. The attractive forces in liquids are not strong enough, however, to keep the particles from moving past one another. Thus, any liquid can be poured, and assumes the shape of the container it occupies.
In solids the intermolecular attractive forces are strong enough to hold particles close together and to lock them virtually in place. Solids, like liquids, are not very compressible because the particles have little free space between them. Because the particles in a solid or liquid are fairly close together compared with those of a gas, we often refer to solids and liquids as condensed phases. We will study solids in Chapter 12. For now it is sufficient to know that the particles of a solid are not free to undergo long-range movement, which makes solids rigid.*
TABLE 11.1 • Some Characteristic Properties of the States of Matter

![]() GO FIGURE
GO FIGURE
For a given substance, do you expect the density of the substance in its liquid state to be closer to the density in the gaseous state or in the solid state?
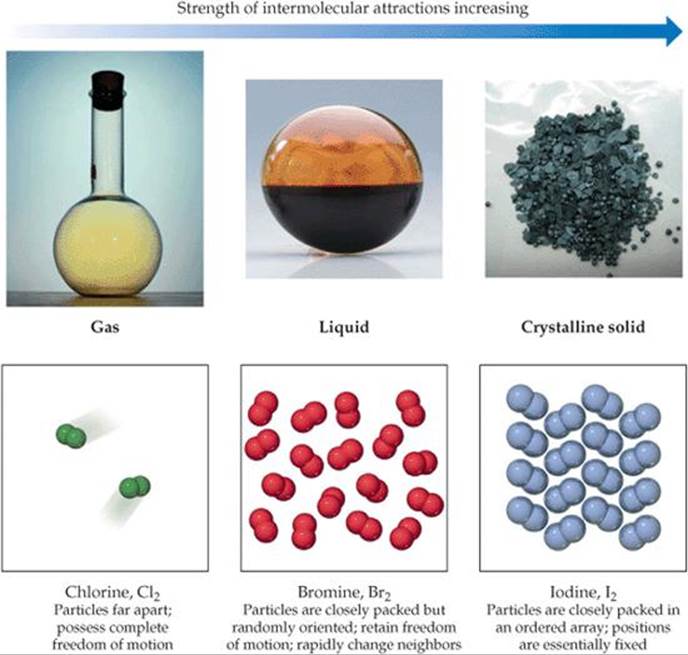
![]() FIGURE 11.2 Gases, liquids, and solids. Chlorine, bromine, and iodine are all made up of diatomic molecules as a result of covalent bonding. However, due to differences in the strength of the intermolecular forces, they exist in three different states at room temperature and standard pressure: Cl2 gaseous, Br2 liquid, I2 solid.
FIGURE 11.2 Gases, liquids, and solids. Chlorine, bromine, and iodine are all made up of diatomic molecules as a result of covalent bonding. However, due to differences in the strength of the intermolecular forces, they exist in three different states at room temperature and standard pressure: Cl2 gaseous, Br2 liquid, I2 solid.
![]() FIGURE 11.2 compares the three states of matter. The state of a substance depends largely on the balance between the kinetic energies of the particles (atoms, molecules or ions) and the interparticle energies of attraction. The kinetic energies, which depend on temperature, tend to keep the particles apart and moving. The interparticle attractions tend to draw the particles together. Substances that are gases at room temperature have much weaker interparticle attractions than those that are liquids; substances that are liquids have weaker interparticle attractions than those that are solids. The different states of matter adopted by the halogens at room temperature—iodine is solid, bromine is a liquid, and chlorine is a gas—is a direct consequence of a decrease in the strength of the intermolecular forces as we move from I2 to Br2 to Cl2.
FIGURE 11.2 compares the three states of matter. The state of a substance depends largely on the balance between the kinetic energies of the particles (atoms, molecules or ions) and the interparticle energies of attraction. The kinetic energies, which depend on temperature, tend to keep the particles apart and moving. The interparticle attractions tend to draw the particles together. Substances that are gases at room temperature have much weaker interparticle attractions than those that are liquids; substances that are liquids have weaker interparticle attractions than those that are solids. The different states of matter adopted by the halogens at room temperature—iodine is solid, bromine is a liquid, and chlorine is a gas—is a direct consequence of a decrease in the strength of the intermolecular forces as we move from I2 to Br2 to Cl2.
We can change a substance from one state to another by heating or cooling, which changes the average kinetic energy of the particles. NaCl, for example, a solid at room temperature, melts at 1074 K and boils at 1686 K under 1 atm pressure, and Cl2, a gas at room temperature, liquefies at 239 K and solidifies at 172 K under 1 atm pressure. As the temperature of a gas decreases, the average kinetic energy of its particles decreases, allowing the attractions between the particles to first draw the particles close together, forming a liquid, and then to virtually lock them in place, forming a solid. Increasing the pressure on a gas can also drive transformations from gas to liquid to solid because the increased pressure brings the molecules closer together, thus making intermolecular forces more effective. For example, propane (C3H8) is a gas at room temperature and 1 atm pressure, whereas liquefied propane (LP) is a liquid at room temperature because it is stored under much higher pressure.