CHEMICAL BIOLOGY
Alkaloid Biosynthesis
Sarah E. O'Connor, Department of Chemistry, Massachusetts Institute of Technology, Cambridge, Massachusetts
doi: 10.1002/9780470048672.wecb004
How nature synthesizes complex secondary metabolites, or natural products, can be studied only by working within the disciplines of both chemistry and biology. Alkaloids are a complex group of natural products with diverse mechanisms of biosynthesis. This article highlights the biosynthesis of four major classes of plant-derived alkaloids. Only plant alkaloids for which significant genetic information has been obtained were chosen for review. Isoquinoline alkaloid, terpenoid indole alkaloid, tropane alkaloid, and purine alkaloid biosynthesis are described here. The article is intended to provide an overview of the basic mechanism of biosynthesis for selected members of each pathway. Manipulation of these pathways by metabolic engineering is highlighted also.
Alkaloids are a highly diverse group of natural products related only by the presence of a basic nitrogen atom located at some position in the molecule. Even among biosynthetically related classes of alkaloids, the chemical structures are often highly divergent. Although some classes of natural products have a recognizable biochemical paradigm that is centrally applied throughout the pathway, for example, the “assembly line” logic of polyketide biosynthesis (1), the biosynthetic pathways of alkaloids are as diverse as the structures. It is difficult to predict the biochemistry of a given alkaloid based solely on precedent, which makes alkaloid biosynthesis a challenging, but rewarding, area of study.
Biologic Background
Hundreds of alkaloid biosynthetic pathways have been studied by chemical strategies, such as isotopic labeling experiments (2, 3). However, modern molecular biology and genetic methodologies have facilitated the identification of alkaloid biosynthetic enzymes. This article focuses on pathways for which a significant amount of genetic and enzymatic information has been obtained. Although alkaloid natural products are produced by insects, plants, fungi, and bacteria, this article focuses on four major classes of plant alkaloids: the isoquinoline alkaloids, the terpenoid indole alkaloids, the tropane alkaloids, and the purine alkaloids.
In general, plant biosynthetic pathways are understood poorly when compared with prokaryotic and fungal metabolic pathways. A major reason for this poor understanding is that genes that express complete plant pathways typically are not clustered together on the genome. Therefore, each plant enzyme often is isolated individually and cloned independently. However, several enzymes involved in plant alkaloid biosynthesis have been cloned successfully, and many more enzymes have been purified from alkaloid-producing plants or cell lines (4-6). Identification and study of the biosynthetic enzymes has a significant impact on the understanding of the biochemistry of the pathway. Furthermore, genetic information also can be used to understand the complicated localization patterns and regulation of plant pathways. This article focuses on the biochemistry responsible for the construction of plant alkaloids and summarizes the biosynthetic genes that have been identified to date. Some of these pathways have been the subject of metabolic engineering studies; the results of these studies are mentioned here also. An excellent, more detailed review that covers the biochemistry and genetics of plant alkaloid biosynthesis up until the late 1990s is available also (7).
Isoquinoline Alkaloids
The isoquinoline alkaloids include the analgesics morphine and codeine as well as the antibiotic berberine (Fig. 1a). Morphine and codeine are two of the most important analgesics used in medicine, and plants remain the main commercial source of the alkaloids (8). Development of plant cell cultures of Eschscholzia californica, Papaver somniferum, and Coptis japonica has aided in the isolation and cloning of many enzymes involved in the biosynthesis of isoquinoline alkaloids (9).
Early steps of isoquinoline biosynthesis
Isoquinoline biosynthesis begins with the substrates dopamine and p-hydroxyphenylacetaldehyde (Fig. 1b). Dopamine is made from tyrosine by hydroxylation and decarboxylation. Enzymes that catalyze the hydroxylation and decarboxylation steps in either order exist in the plant, and the predominant pathway for formation of dopamine from tyrosine is not clear. The second substrate, p-hydroxyphenylacetaldehyde, is generated by transamination and decarboxylation of tyrosine (10, 11).
Condensation of dopamine and p-hydroxyphenylacetaldehyde is catalyzed by norcoclaurine synthase to form (S)-norcoclaurine (Fig. 1b). Two norcoclaurine synthases with completely unrelated sequences were cloned (Thalictrum flavum and C. japonica) and heterologously expressed in E. coli (12-14). One is homologous to iron-dependent diooxygenases, whereas the other is homologous to a pathogenesis-related protein. Undoubtedly, future experiments will shed light on the mechanism of these enzymes and on how two such widely divergent sequences can catalyze the same reaction.


Figure 1. (a) Representative isoquinoline alkaloids. (b) Early biosynthetic steps of the isoquinoline pathway yield the biosynthetic intermediate (S)-reticuline, the central biosynthetic intermediate for all isoquinoline alkaloids. (c) Berberine and sanguinarine biosynthesis pathways. (d) Morphine biosynthesis. NCS, norcoclaurine synthase; 6-OMT, norcoclaurine 6-O-methyltransferase; CNMT, coclaurine N-methyltransferase (Cyp80B); NMTC, N-methylcoclaurine 3'-hydroxylase; 4'-OMT, 3'-hydroxy-N-methylcoclaurine 4'-O-methyltransferase; BBE, berberine bridge enzyme; SOMT, scoulerine 9-O-methyltransferase; CS, canadine synthase; TBO, tetrahydroprotoberberine oxidase; CHS, cheilanthifoline synthase; SYS, stylopine synthase; NMT, N-methyltransferase; NMSH, N-methylstylopine hydroxylase; P6H protopine 6-hydroxylase; DHPO, dihydrobenzophenanthridine oxidase; RO, reticuline oxidase; DHR, dihydroreticulinium ion reductase; STS, salutaridine synthase; SalR, salutaridine reductase; SalAT, salutaridinol acetyltransferase; COR, codeinone reductase.
One of the hydroxyl groups of (S)-norcoclaurine is methylated by a S-adenosyl methionine-(SAM)-dependent O-methyl transferase to yield (S)-coclaurine. This enzyme has been cloned, and the heterologously expressed enzyme exhibited the expected activity (15-17). The resulting intermediate is then N-methylated to yield N-methylcoclaurine, an enzyme that has been cloned recently (18, 19). N-methylcoclaurine, in turn, is hydroxylated by a P450-dependent enzyme (CYP80B), N-methylcoclaurine 3'-hydroxylase, that has been cloned (20, 21). The 4' hydroxyl group then is methylated by the enzyme 3’-hydroxy-N-methylcoclaurine 4’-O-methyltransferase (4’-OMT) to yield (S)-reticuline, the common biosynthetic intermediate for the berberine, benzo(c)phenanthridine, and morphinan alkaloids (Fig. 1b). The gene for this methyl transferase also has been identified (15, 22). These gene sequences also were used to identify the corresponding T. flavum genes that encode the biosynthetic enzymes for reticuline from a cDNA library (23). At this point, the biosynthetic pathway then branches to yield the different structural classes of isoquinoline alkaloids.
Berberine biosynthesis
(S)-reticuline is converted to (S)-scoulerine by the action of a well-characterized flavin-dependent enzyme, berberine bridge enzyme (Fig. 1c). This enzyme has been cloned from several plant species, and the mechanism of this enzyme has been studied extensively (24-28). (S)-scolerine is then O-methylated by scoulerine 9-O-methyltransferase to yield (S)-tetrahydrocolumbamine. Heterologous expression of this gene in E. coli yielded an enzyme that had the expected substrate specificity (29). A variety of O-methyl transferases also have been cloned from Thalictrum tuberosum (30). The substrate-specific cytochrome P450 oxidase canadine synthase (31) that generates the methylene dioxy bridge of (S)-canadine has been cloned recently (32). The final step of berberine biosynthesis is catalyzed by a substrate-specific oxidase, tetrahy- droprotoberberine oxidase, the sequence of which has not been identified yet (33).
Overproduction of berberine in C. japonica cell suspension cultures was achieved by selection of a high-producing cell line (34) with reported productivity of berberine reaching 7 g/L (35). This overproduction is one of the first demonstrations of production of a benzylisoquinoline alkaloid in cell culture at levels necessary for economic production. This cell line has facilitated greatly the identification of the biosynthetic enzymes.
Sanginarine biosynthesis
The biosynthesis of the highly oxidized benzo(c)phenanthidine alkaloid sanguinarine is produced in a variety of plants and competes with morphine production in opium poppy. The pathway to sanguinarine has been elucidated at the enzymatic level (Fig. 1c) (36). Sanguinarine biosynthesis starts from (S)-scoulerine, as in berberine biosynthesis. Methylenedioxy bridge formation then is catalyzed by the P450 cheilanthifoline synthase to yield cheilanthifoline (37). A second P450 enzyme, stylopine synthase, catalyzes the formation of the second methyenedioxy bridge of stylopine (37). Stylopine synthase from E. californica has been cloned recently (38). Stylopine then is N-methylated by (S)-tetrahydroprotoberberine cis-N-methyltransferase to yield (S)-cis-N-methylstylopine, an enzyme that has been cloned recently from opium poppy (39). A third P450 enzyme, (S)-cis-N-methylstylopine hydroxylase, then forms protopine. Protopine is hydroxylated by a fourth P450 enzyme, protopine 6-hydroxylase, to yield an intermediate that rearranges to dihydrosanguinarine (40). This intermediate also serves as the precursor to the benzo(c)phenanthridine alkaloid macarpine (Fig. 1a). The copper-dependent oxidase dihydrobenzophenanthridine oxidase, which has been purified (41, 42), then catalyzes the formation of sanguinarine from dihydrosanguinarine.
Additional enzymes from other benzo(c)phenanthidine alkaloids have been cloned. For example, an O-methyl transferase implicated in palmitine biosynthesis has been cloned recently (43).
Morphine biosynthesis
The later steps of morphine biosynthesis have been investigated in P. somniferum cells and tissue. Notably, in morphine biosynthesis, (S)-reticuline is converted to (R)-reticuline, thereby epimerizing the stereocenter generated by norcoclaurine synthase at the start of the pathway (Fig. 1d). (S)-reticuline is converted to (R)-reticuline through a 1,2-dehydroreticuline intermediate. Dehydroreticuline synthase catalyzes the oxidation of (S)-reticuline to 1,2-dehydroreticulinium ion (44). This enzyme has not been cloned but has been purified partially and shown to be membrane-associated. This intermediate then is reduced by dehydroreticuline reductase, an NADPH-dependent enzyme that stereoselectively transfers a hydride to dehydroreti- culinium ion to yield (R)-reticuline. This enzyme has not been cloned yet but has been purified to homogeneity (45).
Next, the key carbon-carbon bond of the morphinan alkaloids is formed by the cytochrome P450 enzyme salutaridine synthase. Activity for this enzyme has been detected in microsomal preparations, but the sequence has not been identified (46). The keto moiety of the resulting product, salutaridine, then is stereoselectively reduced by the NADPH-dependent salutaridine reductase to form salutardinol. The enzyme has been purified (47), and a recent transcript analysis profile of P. sominiferum has resulted in the identification of the clone (48). Salutaridinol acetyltransferase, also cloned, then transfers an acyl group from acetyl-CoA to the newly formed hydroxyl group, which results in the formation of salutaridinol-7-O-acetate (49). This modification sets up the molecule to undergo a spontaneous reaction in which the acetate can act as a leaving group. The resulting product, thebaine, then is demethylated by an as yet uncharacterized enzyme to yield neopinione, which exists in equilibrium with its tautomer codeinone. The NADPH-dependent codeinone reductase catalyzes the reduction of codeinone to codeine and has been cloned (50, 51). Finally, codeine is demethylated by an uncharacterized enzyme to yield morphine.
The localization of isoquinoline biosynthesis has been investigated at the cellular level in intact poppy plants by using in situ RNA hybridization and immunoflouresence microscopy. The localization of 4'-OMT (reticuline biosynthesis), berberine bridge enzyme (saguinarine biosynthesis), salutaridinol acetyltransferase (morphine biosynthesis), and codeinone reductase (morphine biosynthesis) has been probed. 4'-OMT and salutaridinol acetyltransferase are localized to parenchyma cells, whereas codeinone reductase is localized to laticifer cells in sections of capsule (fruit) and stem from poppy plants. Berberine bridge enzyme is found in parenchyma cells in roots. Therefore, this study suggests that two cell types are involved in isoquinoline biosynthesis in poppy and that intercellular transport is required for isoquinoline alkaloid biosynthesis (52). Another study, however, implicates a single cell type (sieve elements and their companion cells) in isoquinoline alkaloid biosynthesis (53, 54). Therefore, it is not clear whether transport of pathway intermediates is required for alkaloid biosynthesis or whether the entire pathway can be performed in one cell type. Localization of enzymes in alkaloid biosynthesis is difficult, and, undoubtedly, future studies will provide more insight into the trafficking involved in plant secondary metabolism.
Metabolic engineering of morphine biosynthesis
In attempts to accumulate thebaine and decrease production of morphine (a precursor to the recreational drug heroine), codeinone reductase in opium poppy plant was down-regulated by using RNAi (8). Silencing of codeinone reductase results in the accumulation of (S)-reticuline but not the substrate codeinone or other compounds on the pathway from (S)-reticuline to codeine. However, the overexpression of codeinone reductase in opium poppy plants did result, in fact, in an increase in morphine and other morphinan alkaloids, such as morphine, codeine, and thebaine, compared with control plants (55). Gene expression levels in low morphine-producing poppy plants have been analyzed also (56). Silencing of berberine bridge enzyme in opium poppy plants also resulted in a change in alkaloid profile in the plant latex (57).
The cytochrome P450 responsible for the oxidation of (S)-N-methylcoclaurine to (S)-3'-hydroxy-N-methylcocluarine has been overexpressed in opium poppy plants, and morphinan alkaloid production in the latex is increased subsequently to 4.5 times the level in wild-type plants (58). Additionally, suppression of this enzyme resulted in a decrease in morphinan alkaloids to 16% of the wild-type level. Notably, analysis of a variety of biosynthetic gene transcript levels in these experiments supports the hypothesis that this P450 enzyme plays a regulatory role in the biosynthesis of benzylisoquinoline alkaloids. Collectively, these studies highlight that the complex metabolic networks found in plants are not redirected easily or predictably in all cases.
Terpenoid Indole Alkaloids
The terpenoid indole alkaloids have a variety of chemical structures and a wealth of biologic activities (Fig. 2a) (59, 60). Terpenoid indole alkaloids are used as anticancer, antimalarial, and antiarrhythmic agents. Although many biosynthetic genes from this pathway remain unidentified, recent studies have correlated terpenoid indole alkaloid production with the transcript profiles of Catharanthus roseus cell cultures (61).



Figure 2. (a) Representative terpenoid indole alkaloids. (b) Early biosynthetic steps of the terpenoid indole alkaloid pathway yield the strictosidine, the central biosynthetic intermediate for all terpenoid indole alkaloids. (c) Ajmaline biosynthesis. (d) Ajmalicine and tetrahydroalstonine biosynthesis. (e) Vindoline biosynthesis from tabersonine. TDC, tryptophan decarboxylase; STR, strictosidine synthase; SGS, strictosidine glucosidase; SB, sarpagan bridge enzyme; PNAE, polyneuridine aldehyde reductase; VS, vinorine synthase; VH, vinorine hydroxylase; VR, vomilenine reductase; DHVR, dihydrovomilenine reductase; AAE, 17-O-acetyl-ajmalanesterase; NMT, norajmaline-N-methyltransferase; T16H, tabersonine-16-hydroxylase; HTOM, 16-hydroxytabersonine-16-O-methyltransferase; NMT, N-methyltransferase; D4H, desacetoxyvindoline-4-hydroxylase; DAT, desacetylvindoline O-acetyltransferase.
Early steps of terpenoid indole alkaloid biosynthesis
All terpenoid indole alkaloids are derived from tryptophan and the iridoid terpene secologanin (Fig. 2b). Tryptophan decarboxylase, a pyridoxal-dependent enzyme, converts tryptophan to tryptamine (62, 63). The enzyme strictosidine synthase catalyzes a stereoselective Pictet-Spengler condensation between tryptamine and secologanin to yield strictosidine. Strictosidine synthase (64) has been cloned from the plants C. roseus (65), Rauwolfia serpentine (66), and, recently, Ophiorrhiza pumila (67). A crystal structure of strictosidine synthase from R. serpentina has been reported (68, 69), and the substrate specificity of the enzyme can be modulated (70).
Strictosidine then is deglycosylated by a dedicated P-glucosidase, which converts it to a reactive hemiacetal intermediate (71-73). This hemiacetal opens to form a dialdehyde intermediate, which then forms dehydrogeissoschizine. The enol form of dehydrogeissoschizine undergoes 1,4 conjugate addition to produce the heteroyohimbine cathenamine (74-76). A variety of rearrangements subsequently act on deglycosylated strictosidine to yield a diversity of indole alkaloid products (77).
Ajmaline biosynthesis
The biosynthetic pathway for ajmaline in R. serpentina is one of the best-characterized terpenoid indole alkaloid pathways. Much of this progress has been detailed in a recent extensive review (78). Like all other terpenoid indole alkaloids, ajmaline, an antiarrhythmic drug with potent sodium channel-blocking properties (79), is derived from deglycosylated strictosidine (Fig. 2c).
A membrane-protein fraction of an R. serpentina extract transforms labeled strictosidine (80, 81) into sarpagan-type alkaloids. The enzyme activity is dependent on NADPH and molecular oxygen, which suggests that sarpagan bridge enzyme may be a cytochrome P450 enzyme. Polyneuridine aldehyde esterase hydrolyzes the polyneuridine aldehyde methyl ester, which generates an acid that decarboxylates to yield epi-vellosamine. This enzyme has been cloned from a Rauwolfia cDNA library, heterologously expressed in E. coli, and subjected to detailed mechanistic studies (82, 83).
In the next step of the ajmaline pathway, vinorine synthase transforms the sarpagan alkaloid epi-vellosamine to the ajmalan alkaloid vinorine (84). Vinorine synthase also has been purified from Rauwolfia cell culture, subjected to protein sequencing, and cloned from a cDNA library (85, 86). The enzyme, which seems to be an acetyl transferase homolog, has been expressed heterologously in E. coli. Crystallization and site-directed mutagenesis studies of this protein have led to a proposed mechanism (87).
Vinorine hydroxylase hydroxylates vinorine to form vomilene (88). Vinorine hydroxylase seems to be a P450 enzyme that requires an NADPH-dependent reductase. This enzyme is labile and has not been cloned yet. Next, the indolenine bond is reduced by an NADPH-dependent reductase to yield 1,2-dihydrovomilenene. A second enzyme, 1,2-dihydrovomilenene reductase, then reduces this product to acetylnorajmaline. Partial protein sequences have been obtained for both of the purified reductases. Although several putative clones that encode these proteins have been isolated, the activity of these clones has not been verified yet (89, 90).
An acetylesterase then hydrolyzes the acetyl link of acetylnorajmaline to yield norajmaline. This esterase has been purified from R. serpentina cell suspension cultures, and a full-length clone has been isolated from a cDNA library. Expression of the gene in tobacco leaves successfully yielded protein with the expected enzymatic activity (91). In the final step of ajmaline biosynthesis, an N-methyl transferase introduces a methyl group at the indole nitrogen of norajmaline. Although this enzymatic activity has been detected in crude cell extracts, the enzyme has not been characterized additionally (92).
Ajmalicine and tetrahydroalstonine
Ajmalicine (raubasine) affects smooth muscle function and is used to help prevent strokes (93), and tetrahydroalstonine exhibits antipsychotic properties (Fig. 2d) (94). These compounds are found in a variety of plants, including C. roseus and R. serpentina. A partially purified NADPH-dependent reductase isolated from a tetrahydroalstonine that produces a C. roseus cell line was shown to catalyze the conversion of cathenamine, a spontaneous reaction product that results after strictosidine deglycosylation, to tetrahydroalstonine in vitro (95). A second C. roseus cell line contains an additional reductase that produces ajmalicine. Labeling studies performed with crude C. roseus cell extracts in the presence of D2O or NADPD support a mechanism in which the reductase acts on the iminium form of cathenamine (96).
Vindoline
Vindoline, an aspidosperma-type alkaloid produced by C. roseus, is a key precursor for vinblastine, an anticancer drug that is the most important pharmaceutical product of C. roseus. Vindoline, like ajmalicine and ajmaline, is produced from deglycosylated strictosidine. Deglycosylated strictosidine is converted to tabersonine through a series of biochemical steps for which no enzymatic information exists. More details are known about the six steps that catalyze the elaboration of tabersonine to vindoline (Fig. 2e) (97).
Tabersonine-16-hydroxylase, a cytochrome P450, hydroxylates tabersonine to 16-hydroxy-tabsersonine in the first step of this sequence and has been cloned (98, 99). The newly formed hydroxyl group is methylated by a SAM-dependent O-methyl transferase to yield 16-methoxytabersonine; this enzyme (16-hydroxytabersonine-16-O-methyltransferase) has been purified but not cloned (100). In the next step, hydration of a double bond by an uncharacterized enzyme produces 16-methoxy-2,3-dihydro-3-hydroxytabersonine. Transfer of a methyl group to the indole nitrogen by an N-methyl transferase yields desacetoxyvindoline. This methyl transferase activity has been detected only in differentiated plants, not in plant cell cultures (101). The resulting intermediate, deacteylvindoline, is produced by the oxoglutatarate-dependent dioxygenase enzyme desacetylvindoline 4-hydroxylase. This enzyme has been cloned and also is absent from plant cell cultures (102). In the last step, desacteylvindoline is acetylated by desacteylvindoline O-acetyl transferase. This enzyme, also absent from nondifferentiated plant material, has been cloned successfully (103).
As in morphine biosynthesis, the knowledge of the enzyme sequences allows a more detailed understanding of the localization of the enzymes (104). Strictosidine synthase (Fig. 2b) seems to be localized to the vacuole (105), and strictosidine glucosidase is believed to be associated with the membrane of the endoplasmic reticulum (73, 106). Tabersonine-16-hydroxylase is associated with the endoplasmic reticulum membrane (98); N-methyl transferase activity is believed to be associated with the thykaloid, a structure located within the chloroplast (101, 107); and vindoline-4-hydroxylase and desacetylvindoline O-acetyltransferase are believed to be localized to the cytosol (Fig. 2e) (107, 108). Overall, extensive subcellular trafficking of biosynthetic intermediates is required for vindoline biosynthesis.
Aside from subcellular compartmentalization, specific cell types are required for the biosynthesis of some terpenoid alkaloids. Several enzymes involved in the early stages of secologanin biosynthesis seem to be localized to the phloem parenchyma, as evidenced by immunocytochemistry and in situ RNA hybridization studies (109). However, additional studies have suggested that these genes also are observed in the epidermis and laticifers (110). Studies of the localization of vindoline biosynthetic enzymes by using immunocytochemistry and in situ RNA hybridization strongly suggest that the mid-part of the vindoline pathway (tryptophan decarboxylase, strictosidine synthase, and tabersonine-16-hydroxylase) takes place in epidermal cells of leaves and stems. However, the later steps catalyzed by desacetylvindoline 4-hydroxylase and desacetylvindoline O-acetyltransferase take place in specialized cells, the laticifers, and idioblasts (109-112). As with isoquinoline alkaloid biosynthesis, deconvolution of the enzyme localization patterns remains a challenging endeavor.
Vinblastine
Vinblastine is a highly effective anticancer agent currently used clinically against leukemia, Hodgkin’s lymphoma, and other cancers. (113, 114). Vinblastine is derived from dimerization of vindoline and another terpenoid indole alkaloid, catharanthine. The dimerization of catharanthine and vindoline is believed to proceed via the formation of an iminium intermediate with catharanthine (Fig. 2e). This iminium intermediate is reduced to form anhydrovinblastine, a naturally occurring compound in C. roseus plants (115). In support of this mechanism, anhydrovinblastine is incorporated into vinblastine and vincristine in feeding studies (116-119).
Peroxidase containing fractions of plant extracts were found to catalyze the formation of the bisindole dehydrovinblastine from catharanthine and vindoline. (120, 121) A peroxidase from C. roseus leaves has been demonstrated to convert vindoline and catharanthine to anhydrovinblastine in vitro (122, 123). Because the dimerization of these C. roseus alkaloids also can be catalyzed by peroxidase from horseradish in reasonable yields (124), it is interesting to speculate that anhydrovinblastine may be a by-product of isolation; after lysis of the plant material, nonspecific peroxidases are released from the vacuole and may act on vindoline and catharanthine.
Metabolic engineering of terpenoid indole alkaloids
Strictosidine synthase and tryptophan decarboxylase have been overexpressed in C. roseus cell cultures (125, 126). Generally, overexpression of tryptophan decarboxylase does not seem to have a significant impact on alkaloid production, although overexpression of strictosidine synthase does seem to improve alkaloid yields. Overexpression of tryptophan and secologanin biosynthetic enzymes in C. roseus hairy root cultures resulted in modest increases in terpenoid indole alkaloid production (127, 128). Secologanin biosynthesis seems to be the rate-limiting factor in alkaloid production (129). Precursor-directed biosynthesis experiments with a variety of tryptamine analogs suggest that the biosynthetic pathway can be used to produce alkaloid derivatives (130). Strictosidine synthase and strictosidine glucosidase enzymes also have been expressed successfully heterologously in yeast (131); however, efforts to express heterologously terpenoid indole alkaloids currently are limited because the majority of the biosynthetic genes remain uncloned.
Transcription factors that upregulate strictosidine synthase (132), as well as a transcription factor that coordinately upregulates expression of several terpenoid indole alkaloid biosynthetic genes, have been found (133). Several zinc finger proteins that act as transcriptional repressors to tryptophan decarboxylase and strictosidine synthase also have been identified (134). Manipulation of these transcription factors may allow tight control of the regulation of terpenoid indole alkaloid production. Interestingly, expression of a transcription factor from Arabidopsis thaliana in C. roseus cell cultures results in an increase in alkaloid production (135).
Tropane Alkaloids
The tropane alkaloids hyoscyamine and scopolamine (Fig. 3a) function as acetylcholine receptor antagonists and are used clinically as parasympatholytics. The illegal drug cocaine also is a tropane alkaloid. The tropane alkaloids are biosynthesized primarily in plants of the family Solonaceae, which includes Hyoscyamus, Duboisia, Atropa, and Scopolia (136, 137). Nicotine, although perhaps not apparent immediately from its structure, is related biosynthetically to the tropane alkaloids (Fig. 3b).

Figure 3. (a) Representative tropane and nicotine alkaloids. (b) Tropane biosynthesis. ODC, ornithine decarboxylase; PMT, putrescine N-methyltransferase; MPO, diamine oxidase; TR1, tropinone reductase 1; H6H, hyocyamine 6b-hydroxylase.
Tropane alkaloid biosynthesis has been studied at the biochemical level, and several enzymes from the biosynthetic pathway have been isolated and cloned, although the pathway has not been elucidated completely at the genetic level (Fig. 3b) (138). L-arginine is converted to the nonproteogenic amino acid L-ornithine by the urease enzyme arginase. Ornithine decarboxylase then decarboxylates ornithine to yield the diamine putrescine. In Hyoscyamus, Duboisia, and Atropa, putrescine serves as the common precursor for the tropane alkaloids.
Putrescine is N-methylated by a SAM-dependent methyl transferase that has been cloned to yield N-methylputrescine (139, 140). Putrescine N-methyl transferase now has been cloned from a variety of plant species (141-143), and site-directed mutagenesis and homology models have led to insights into the structure function relationships of this enzyme (143). N-methylputrescine then is oxidized by a diamine oxidase to form 4-methylaminobutanal, which then spontaneously cyclizes to form the N-methyl-D-pyrrolinium ion (144, 145). This enzyme, which recently has been cloned, seems to be a copper-dependent amine oxidase (146, 147). Immunoprecipitation experiments suggest that this enzyme associates with the enzyme S-adenosylhomocysteine hydrolase (148). The pyrrolinium ion then is converted to the tropanone skeleton by as yet uncharacterized enzymes (Fig. 3b). Although no enzymatic information is available, chemical labeling studies have indicated that an acetate-derived moiety condenses with the pyridollium ion; one possible mechanism is shown in Fig. 3b (136).
Tropanone then is reduced via an NADPH-dependent reductase to tropine that has been cloned from Hyoscyamus niger (149, 150). All tropane-producing plants seem to contain two tropinone reductases, which create a branch point in the pathway. Tropinone reductase I yields the tropane skeleton (Fig. 3b), whereas tropinone reductase II yields the opposite stereocenter, pseudotropine (151). Tropane is converted to scopolamine or hyoscyamine, whereas the TRII product pseudotropine leads to calystegines (152). These two tropinone reductases have been crystallized, and site-directed mutagenesis studies indicate that the stereoselectivity of the enzymes can be switched (153, 154).
The biosynthesis of scopolamine is the best characterized of the tropane alkaloids. After action by tropinone reductase I, tropine is condensed with phenyllactate through the action of a P450 enzyme to form littorine (155). The phenyllactate moiety is believed to derive from an intermediate involved in phenylalanine metabolism (136). Littorine then undergoes rearrangement to form hyoscyamine. The enzyme that catalyzes this rearrangement, which has been purified partially, seems to proceed via a radical mechanism using S-adenysylmethione as the source of an adenosyl radical (156). Labeling studies have been used to examine the mechanism of rearrangement (136, 157-159). Hyoscyamine 6B-hydroxylase (H6H) catalyzes the hydroxylation of hyoscyamine to 6β-hydroxyhyoscyamine as well as the epoxidation to scopolamine (Fig. 3b) (160, 161). H6H, which has been cloned and expressed heterologously (162), is a nonheme, iron-dependent, oxoglutarate-dependent protein. It seems that the epoxidation reaction occurs much more slowly than the hydroxylation reaction. The tropane alkaloids seem to be formed in the roots and then transported to the aerial parts of the plant (163).
Metabolic engineering of tropane alkaloids
Atropa belladonna plants have been transformed with an H6H clone from H. niger. A. belladonna normally produces high levels of hyoscyamine, the precursor for the more pharmaceutically valuable alkaloid scopolamine (Fig. 3b). However, after transformation with the H6H gene, transgenic A. belladonna plants were shown to accumulate scopolamine almost exclusively (164). Additionally, the levels of tropane alkaloid production in a variety of hairy root cultures were altered by overexpression of methyltransferase putrescine-N-methyltransferase and H6H. Overexpression of both of these enzymes in a hairy root cell culture resulted in significant increases in scopolamine production (164, 165). Fluorinated phenyllactic acid substrates could be incorporated into the pathway (166), and several substrates derived from putrescine analogs were turned over by the enzymes of several Solonaceae species (167).
Purine Alkaloids
Caffeine biosynthesis
Caffeine, a purine alkaloid, is one of the most widely known natural products. Caffeine is ingested as a natural component of coffee, tea, and cocoa, and the impact of caffeine on human health has been studied extensively. The biosynthetic pathway of caffeine has been elucidated recently on the genetic level. Caffeine biosynthesis has been studied most widely in the plant species Coffea (coffee) and Camellia (tea) (168, 169).
Xanthosine, which is derived from purine metabolites, is the first committed intermediate in caffeine biosynthesis (Fig. 4). Xanthosine can be formed from de novo purine biosynthesis, S-adenosylmethione (SAM) cofactor, the adenylate pool, and the guanylate pool (169). De novo purine biosynthesis and the adenosine from SAM are believed to be the most important sources of xanthosine (168, 170).
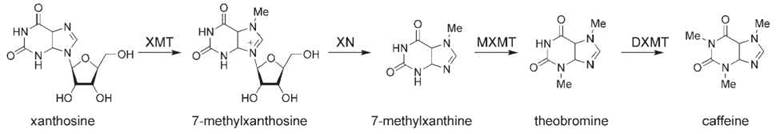
Figure 4. Caffeine biosynthesis. XMT, xanthosine N-methyltransferase (also called 7-methylxanthosine synthase); XN, methylxanthosine nucleotidase; MXMT, 7-methylxanthine-N-methyltransferase (also called theobromine synthase); DXMT, dimethylxanthine-N-methyltransferase (also called caffeine synthase).
The biosynthesis of caffeine begins with the methylation of xanthosine to yield N-methylxanthosine by the enzyme xanthosine N-methyltransferase (XMT) (also called 7-methylxanthosine synthase) (171-173). N-methylxanthosine is converted to N-methylxanthine by methylxanthine nucleosidase, an enzyme that has not been cloned yet (174). N-methylxanthine is converted to theobromine by 7-methylxanthine-N-methyltransferase (MXMT) (also called theobromine synthase), a second N-methyltransferase (171, 175). Theobromine is converted to caffeine by a final N-methyltransferase, dimethylxanthine- N-methyltransferase (DXMT) (also called caffeine synthase) (171).
Coffee and tea plants seem to contain a variety of N-methyltransferase enzymes that have varying substrate specificity (168, 169). For example, a caffeine synthase enzyme isolated from tea leaves catalyzes both the N-methylation of N-methylxanthine and theobromine (176). The substrate specificity of the methyltransferases can be changed by site-directed mutagenesis (177), and the crystal structure of two of the N-methyltransferases has been reported recently (178).
Metabolic engineering of caffeine biosynthesis
Caffeine may act as a natural insecticide in plants. When the three N-methyltransferase genes were overexpressed in tobacco, the resulting increase in caffeine production improved the tolerance of the plants to certain pests (179). Conversely, coffee beans with low caffeine levels would be valuable commercially, given the demand for decaffeinated coffee. Because of the discovery of these N-methyltransferase genes, genetically engineered coffee plants with reduced caffeine content now can be constructed (180, 181). For example, a 70% reduction in caffeine content in Coffea was obtained by downregulating MXMT (theobromine synthase) using RNAi (182). Additionally, the promoter of one of the N-methyltransferases has been discovered recently, which may allow transcriptional gene silencing (183).
References
1. Fischbach MA, Walsh CT. Assembly-line enzymology for polyketide and nonribosomal Peptide antibiotics: logic, machinery, and mechanisms. Chem. Rev. 2006;106:3468-3496.
2. Herbert RB. The Biosynthesis of Secondary Metabolites. 2nd edition. 1989. Chapman and Hall, London.
3. Dewick PM. Medicinal Natural Products: A Biosynthetic Approach. 2nd edition. 2002. John Wiley and Sons, Ltd., New York.
4. Hashimoto T, Yamada Y. New genes in alkaloid metabolism and transport. Curr. Opin. Biotech. 2003; 14:163-168.
5. de Luca V, Laflamme PT. The expanding universe of alkaloid biosynthesis. Curr. Opin. Plant Biol. 2001; 4:225-233.
6. Facchini PJ. Alkaloid biosynthesis in plants: biochemistry, cell biology, molecular regulation and metabolic engineering applications. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001; 52:29-66.
7. Kutchan TM. Molecular genetics of plant alkaloid biosynthesis. In: The Alkaloids: Chemistry and Biology, vol. 50. Cordell GA, ed. 1998. Academic Press, San Diego, CA. pp. 257-316.
8. Allen RS, Millgate AG, Chitty JA, Thisleton J, Miller JAC, Fist AJ, Gerlach WL, Larkin PJ. RNAi-mediated replacement of morphine with the nonnarcotic alkaloid reticuline in opium poppy. Nat. Biotechnol. 2004; 22:1559-1566.
9. Facchini PJ, Bird DA, Bourgault R, Hagel JM, Liscombe DK, MacLeod BP, Zulak KG. Opium poppy: a model system to investigate alkaloid biosynthesis in plants. Can. J. Bot. 2005; 83:1189-1206.
10. Battersby AR, Binks R, Francis RJ, McCaldin DJ, Ramuz H. Alkaloid biosynthesis. IV. 1-Benzylisoquinolines as precursors of thebaine, codeine, and morphine. J. Chem. Soc. 1964:3600-3610.
11. Rueffer M, Zenk MH. Distant precursors of benzylisoquinoline alkaloids and their enzymatic formation. Naturforsch. 1987;42c: 319-332.
12. Samanani N, Facchini PJ. Purification and characterization of norcoclaurine synthase. J. Biol. Chem. 2002; 277:33878-33883.
13. Samanani N, Liscombe DK, Facchini PJ. Molecular cloning and characterization of norcoclaurine synthase, an enzyme catalyzing benzylisoquinoline alkaloid biosynthesis. Plant J. 2004; 40: 302-313.
14. Minami H, Dubouzet E, Iwasa K, Sato F. Functional analysis of norcoclaurine synthase in Coptis japonica. J. Biol. Chem. 2007; 282:6274-6282.
15. Morishige T, Tsujita T, Yamada Y, Sato F. Molecular characterization of the S-adenosyl-L-methionine:3’-hydroxy-N-methylcoclaurine 4’-O- methyltransferase involved in isoquinoline alkaloid biosynthesis in Coptis japonica. J. Biol. Chem. 2000; 275:23398-23405.
16. Ounaroon A, Decker G, Schmidt J, Lottspeich F, Kutchan TM. (R,S)-reticuline 7-O-methyltransferase and (R,S)-norcoclaurine 6-O-methyltransferase of Papaver somniferum - cDNA cloning and characterization of methyl transfer enzymes of alkaloid biosynthesis in opium poppy. Plant J. 2003; 36:808-819.
17. Sato F, Tsujita T, Katagiri Y, Yoshida S, Yamada Y. Purification and characterization of S-adenosyl-L-methionine: norcoclaurine 6-O-methyltransferase from cultured Coptis japonica cells. Eur. J. Biochem. 1994; 225:125-131.
18. Choi K-B, Morishige T, Shitan N, Yazaki K, Sato F. Molecular cloning and characterization of coclaurine N- methyltransferase from cultured cells of Coptis japonica. J. Biol. Chem. 2002; 277:830-835.
19. Choi K-B, Morishige T, Sato F. Purification and characterization of coclaurine N-methyltransferase from cultured Coptis japonica cells. Phytochemistry 2001; 56:649-655.
20. Huang F-C, Kutchan TM. Distribution of morphinan and benzo-(c)phenanthridine alkaloid gene transcript accumulation in Pa- paver somniferum. Phytochemistry 2000; 53:555-564.
21. Pauli HH, Kutchan TM. Molecular cloning and functional heterologous expression of two alleles encoding (S)-N-methylcoclaurine 3;-hydroxylase (CYP80B1), a new methyl jasmonateinducible cytochrome P-450-dependent mono-oxygenase of benzylisoquinoline alkaloid biosynthesis. Plant J. 1998; 13:793-801.
22. Ziegler J, Diaz-Chavez ML, Kramell R, Ammer C, Kutchan TM. Comparative macroarray analysis of morphine containing Papaver somniferum and eight morphine free Papaver species identifies an O- methyltransferase involved in benzylisoquinoline biosynthesis. Planta 2005; 222:458-471.
23. Samanani N, Park S-U, Facchini PJ. Cell type-specific localization of transcripts encoding nine consecutive enzymes involved in protoberberine alkaloid biosynthesis. Plant Cell. 2005; 17:915-926.
24. Dittrich H, Kutchan TM. Molecular cloning, expression, and induction of berberine bridge enzyme, an enzyme essential to the formation of benzophenanthridine alkaloids in the response of plants to pathogenic attack. Proc. Natl. Acad. Sci. 1991; 88:9969-9973.
25. Facchini PJ, Penzes C, Johnson AG, Bull D. Molecular characterization of berberine bridge enzyme genes from opium poppy. Plant Physiol. 1996; 112:1669-1677.
26. Hauschild K, Pauli HH, Kutchan TM. Isolation and analysis of a gene bbe1 encoding the berberine bridge enzyme from the California poppy Eschscholzia californica. Plant Mol. Biol. 1998; 36:473-478.
27. Kutchan TM, Dittrich H. Characterization and mechanism of the berberine bridge enzyme, a covalently flavinylated oxidase of benzophenanthridine alkaloid biosynthesis in plants. J. Biol. Chem. 1995; 270:24475-24481.
28. Winkler A, Hartner F, Kutchan TM, Glieder A, Macheroux P. Biochemical evidence that berberine bridge enzyme belongs to a novel family of flavoproteins containing a bi-covalently attached FAD cofactor. J. Biol. Chem. 2006; 281:21276-21285.
29. Takeshita N, Fujiwara H, Mimura H, Fitchen JH, Yamada Y, Sato F. Molecular cloning and characterization of S-adenosyl-L-methionine:scoulerine-9-O-methyltransferase from cultured cells of Coptis japonica. Plant Cell Physiol. 1995; 36:29-36.
30. Frick S, Kutchan TM. Molecular cloning and functional expression of O-methyltransferasese common to isoquinoline alkaloid and phenylpropanoid biosynthesis. Plant J. 1999; 17:329-339.
31. Galneder E, Rueffer M, Wanner G, Tabata M, Zenk MH. Alternative final steps in Coptis japonica cell cultures. Plant Cell Rep. 1988; 7:1-4.
32. Ikezawa N, Tanaka M, Nagayoshi M, Shinkyo R, Sakaki T, Inouye K, Sato F. Molecular cloning and characterization of CYP719, a methylenedioxy bridge-forming enzyme that belongs to a novel P450 family, from cultured Coptis japonica cells. J. Biol. Chem. 2003; 278:38557-38565.
33. Amann M, Nagakura N, Zenk MH. Purification and properties of (S)-tetrahydroprotoberberine oxidase from suspension cultures of Berberis wilsoniae. Eur. J. Biochem. 1988; 175:17-25.
34. Sato F, Yamada Y. High berberine-producing cultures of Coptis-japonica cells. Phytochemistry 1984; 23:281-285.
35. Matsubara K, Kitani S, Yoshioka T, Morimoto T, Fujita Y. High-density culture of Coptis-japonica cells increases berberine production. J. Chem. Technol. Biotechnol. 1989; 46:61-69.
36. Zenk MH. The formation of benzophenanthridine alkaloids. Pure Appl. Chem. 1994;66:2023-2028.
37. Bauer W, Zenk MH. Two methylenedioxy bridge forming cytochrome P-450 dependent enzymes are involved in (S)-stylopine biosynthesis. Phytochemistry 1991; 30:2953-2961.
38. Ikezawa N, Iwasa K, Sato F. Molecular cloning and characterization of methylenedioxy bridge-forming enzymes involved in stylopine biosynthesis in E. californica. FEBS J. 2007; 274: 1019-1035.
39. Liscombe DK, Facchini PJ. Molecular cloning and characterization of tetrahydroprotoberberine cis-N-methyltransferase, an enzyme involved in alkaloid biosynthesis in opium poppy. J. Biol. Chem. 2007; 282:14741-14751.
40. Tanahashi T, Zenk MH. Elicitor induction and characterization of microsomal protopine 6-hydroxylase, the central enzyme in benzophenanthridine alkaloid biosynthesis. Phytochemistry 1990; 29:1113-1122.
41. Schumacher HM, Zenk MH. Partial purification and characterization of dihydrophenanthridine oxidase from Eschscholtzia californica cell suspension cultures. Plant Cell Rep. 1988; 7:43-46.
42. Arakawa H, Clark WG, Psenak M, Coscia CJ. Purification and characterization of dihydrobenzophenanthridine oxidase from elicited Sanguinaria canadensis cell cultures. Arch. Biochem. Biophys. 1992; 299:1-7.
43. Morishige T, Dubouzet E, Choi K-B, Yazaki K, Sato F. Molecular cloning of columbamine O- methyltransferase from cultured Coptis japonica cells. Eur. J. Biochem. 2002; 269:5659-5667.
44. Hirata K, Poeaknapo C, Schmidt J, Zenk MH. 1,2-Dehydroreticuline synthase, the branch point enzyme opening the morphinan biosynthetic pathway. Phytochemistry 2004; 65:1039-1046.
45. De-Eknamkul W, Zenk MH. Purification and properties of 1,2-dehydroreticuline reductase from Papaver somniferum seedlings. Phytochemistry 1992; 31:813-821.
46. Gerardy R, Zenk MH. Formation of salutaridine from (R)-reticuline by a membrane-bound cytochrome P-450 enzyme from Papaver somniferum. Phytochemistry 1992; 32:79-86.
47. Gerardy R, Zenk MH. Purification and characterization of salutaridine:NADPH 7-oxidoreductase from Papaver somniferum. Phytochemistry 1993;34:125-132.
48. Ziegler J, Voigtlaender S, Schmidt J, Kramell R, Miersch O, Ammer C, Gesell A, Kutchan TM. Comparative transcript and alkaloid profiling in Papaver species identifies a short chain dehydrogenase/ reductase involved in morphine biosynthesis. Plant J. 2006; 48:177-192.
49. Grothe T, Lenz R, Kutchan TM. Molecular characterization of the salutaridinol 7-O-acetyltransferase involved in morphine biosynthesis in opium poppy Papaver somniferum. J. Biol. Chem. 2001; 276:30717-30723.
50. Lenz R, Zenk MH. Stereoselective reduction of codeinone, the penultimate enzymic step during morphine biosynthesis in Papaver somniferum. Tetrahedron Lett. 1995; 36:2449-2452.
51. Unterlinner B, Lenz R, Kutchan TM. Molecular cloning and functional expression of codeinone reductase. Plant J. 1999; 18:465-475.
52. Weid M, Ziegler J, Kutchan TM. The roles of latex and the vascular bundle in morphine biosynthesis in the opium poppy, Papaver somniferum. Proc. Natl. Acad. Sci. U.S.A. 2004; 101:13957-13962.
53. Samanani N, Alcantara J, Bourgault R, Zulak KG, Facchini PJ. The role of phloem sieve elements and laticifers in the biosynthesis and accumulation of alkaloids in opium poppy. Plant J. 2006; 47:547-563.
54. Bird DA, Franceschi VR, Facchini PJ. A tale of three cell types: alkaloid biosynthesis is localized to sieve elements in opium poppy. Plant Cell 2003; 15:2626-2635.
55. Larkin PJ, Miller JAC, Allen RS, Chitty JA, Gerlach WL, Frick S, Kutchan TM, Fist AJ. Increasing morphinan alkaloid production by overexpressing codeinone reductase in transgenic Papaver somniferum. Plant Biotechnol. J. 2007; 5:26-37.
56. Millgate AG, Pogson G, Wilson IW, Kutchan TM, Zenk MH, Gerlach WL, Fist AJ, Larkin PJ. Analgesia: morphine-pathway block in topi poppies. Nature 2004; 431:413-414.
57. Frick S, Chitty J, Kramell R, Schmidt J, Allen R, Larkin P, Kutchan T. Transformation of opium poppy with antisense berberine bridge enzyme gene via somatic embryogenesis results in an altered ratio of alkaloids in latex but not in roots. Transgenic Res. 2004; 13:607-613.
58. Frick S, Kramell R, Kutchan TM. Metabolic engineering with a morphine biosynthetic P450 in opium poppy surpasses breeding. Met. Eng. 2007; 9:169-176.
59. Leonard J. Recent progress in the chemistry of monoterpenoid indole alkaloids derived from secologanin. Nat. Prod. Rep. 1999; 16:319-338.
60. Van der Heijden R, Jacobs DI, Snoeijer W, Hallard DVR. The Catharanthus alkaloids: pharmacognosy and biotechnology. Curr. Med. Chem. 2004; 11:607-628.
61. Rischer H, Oresic M, Seppanen-Laakso T, Katajamaa M, Lammertyn F, Ardiles-Diaz W, Van Montagu MCE, Inze D, Oksman-Caldentey K-M, Goossens A. Gene-to-metabolite networks for terpenoid indole alkaloid biosynthesis in Catharanthus roseus cells. Proc. Natl. Acad. Sci. U.S.A. 2006; 103:5614-5619.
62. de Luca V, Marineau C, Brisson N. Molecular cloning and analysis of a cDNA encoding a plant tryptophan decarboxylase. Proc. Natl. Acad. Sci. U.S.A. 1989; 86:2582-2586.
63. Facchini PJ, Huber-Allanach KL, Tari LW. Plant aromatic L-amino acid decarboxylases: evolution, biochemistry, regulation, and metabolic engineering applications. Phytochemistry 2000; 54:121-138.
64. Kutchan TM. Strictosidine: from alkaloid to enzyme to gene. Phytochemistry 1993; 32:493-506.
65. McKnight TD, Roessner CA, Devagupta R, Scott AI, Nessler C. Nucleotide sequence of a cDNA encoding the vacuolar protein strictosidine synthase from Catharanthus roseus. Nuc. Acids Res. 1990; 18:4939.
66. Kutchan TM, Hampp N, Lottspeich F, Beyreuther K, Zenk MH. The cDNA clone for strictosidine synthase from Rauvolfia serpentina. DNA sequence determination and expression in Escherichia coli. FEBS Lett. 1988; 237:40-44.
67. Yamazaki Y, Sudo H, Yamazaki M, Aimi N, Saito K. Camp- tothecin biosynthetic genes in hairy roots of Ophiorrhiza pumila: cloning, characterization and differential expression in tissues and by stress compounds. Plant Cell Physiol. 2003; 44:395-403.
68. Ma X, Panjikar S, Koepke J, Loris E, Stockigt J. The structure of Rauvolfia serpentina strictosidine synthase is a novel six-bladed beta-propeller fold in plant proteins. Plant Cell 2006; 18:907-920.
69. Koepke J, Ma X, Fritzsch G, Michel H, Stoeckigt J. Crystallization and preliminary X-ray analysis of strictosidine synthase and its complex with the substrate tryptamine. Acta Crystallographica D. 2005;D61:690-693.
70. Chen S, Galan MC, Coltharp C, O’Connor SE. Redesign of a central enzyme in alkaloid biosynthesis. Chem. Biol. 2006; 13:1137-1141.
71. Gerasimenko I, Sheludko Y, Ma X, Stockigt J. Heterologous expression of a Rauvolfia cDNA encoding strictosidine glucosidase, a biosynthetic key to over 2000 monoterpenoid indole alkaloids. Eur. J. Biochem. 2002; 269:2204-2213.
72. Brandt V, Geerlings A, Tits M, Delaude C, Van der Heijden R, Verpoorte R, Angenot L. New strictosidine b-glucosidase from Strychnos mellodora. Plant Physiol. Biochem. 2000; 38:187-192.
73. Geerlings A, Ibanez MM-L, Memelink J, Van der Heijden R, Verpoorte R. Molecular cloning and analysis of strictosidine b-D-glucosidase, an enzyme in terpenoid indole alkaloid biosynthesis in Catharanthus roseus. J. Biol. Chem. 2000; 275: 3051-3056.
74. Kan-Fan C, Husson HP. Isolation and biomimetic conversion of 4,21-dehydrogeissoschizine. J. Chem. Soc. Chem. Comm. 1979:1015-1018.
75. El-Sayed M, Choi YH, Frederich M, Roytrakul S, Verpoorte R. Alkaloid accumulation in Catharanthus roseus cell suspension cultures fed with stemmadenine. Biotech. Lett. 2004; 26:793-798.
76. Heinstein P, Hofle G, Stockigt J. Involvement of cathenamine in the formation of N-analogues of indole akaloids. Planta Med. 1979; 37:349-357.
77. Lounasmaa M, Hanhinen P. Biomimetic formation and interconversion in the heteroyohimbine series. Heterocycles 1998; 48: 1483-1492.
78. Ruppert M, Ma X, Stoeckigt J. Alkaloid biosynthesis in Rauvolfia-cDNA cloning of major enzymes of the ajmaline pathway. Curr. Org. Chem. 2005; 9:1431-1444.
79. Brugada J, Brugada P, Brugada R. The ajmaline challenge in Brugada syndrome. A useful tool or misleading information? European Heart J. 2003; 24:1085-1086.
80. Stockigt J. Enzymatic biosynthesis of monoterpenoid indole alkaloids: ajmaline, sarpagine and vindoline. Studies Organ. Chem. 1986; 26:497-511.
81. Schmidt D, Stockigt J. Enzymatic formation of the sarpagan bridge: a key step in the biosynthesis of sarpagine and ajmaline type alkaloids. Planta Med. 1995; 61:254-258.
82. Mattern-Dogru E, Ma X, Hartmann J, Decker H, Stockigt J. Potential active site residues in polyneuridine aldehyde esterase a central enzyme of indole alkaloid biosynthesis by modelling and site directed mutagenesis. Eur. J. Biochem. 2002; 269:2889-2896.
83. Dogru E, Warzecha H, Seibel F, Haebel S, Lottspeich F, Stockigt J. The gene encoding polyneuridine aldehyde esterase of monoterpenoid indole alkaloid biosynthesis in plants is an ortholog of the a/b hydrolase super family. Eur. J. Biochem. 2000; 267:1397-1406.
84. Pfitzner A, Stoeckigt J. Biogenetic link between sarpagine and ajmaline type alkaloids. Tetrahedron Lett. 1983; 24:5197-5200.
85. Gerasimenko I, Ma X, Sheludko Y, Mentele R, Lottspeich F, Stockigt J. Purification and partial amino acid sequences of the enzyme vinorine synthase involved in a crucial step of ajmaline biosynthesis. Bioorg. Med. Chem. 2004; 12:2781-2786.
86. Bayer A, Ma X, Stockigt J. Acetyltransfer in natural product biosynthesis functional cloning and molecular analysis of vinorine synthase. Bioorg. Med. Chem. 2004; 12:2787-2795.
87. Ma X, Koepke J, Panjikar S, Fritzsch G, Stoeckigt J. Crystal estructure of vinorine synthase, the first representative of the BAHD superfamily. J. Biol. Chem. 2005; 280:13576-13583.
88. Falkenhagen H, Polz L, Takayama H, Kitajima M, Sakai S, Aimi N, Stockigt J. Substrate specificity of vinorine hydroxylase, a novel membrane bound key enzyme of Rauwolfia indole alkaloid biosynthesis. Heterocycles 1995; 41:2683-2690.
89. Gao S, von Schumann G, Stockigt J. A newly detectedreductase from Rauvolfia closes a gap in the biosynthesis of the antiarrhythmic alkaloid ajmaline. Planta Med. 2002; 68:906-911.
90. von Schumann G, Gao S, Stockigt J. Vomilenine reductase: a novel enzyme catalyzing a crucial step in the biosynthesis of the therapeutically applied antiarrhythmic alkaloid ajmaline. Bioorg. Med. Chem. 2002; 10:1913-1918.
91. Ruppert M, Woll J, Giritch A, Genady E, Ma X, Stoeckigt J. Functional expression of an ajmaline pathway-specific esterase from Rauvolfia in a novel plant-virus expression system. Planta 2005; 222:888-898.
92. Stockigt J, Pfitzner A, Keller PI. Enzymic formation of ajmaline. Tetrahedron Lett. 1983; 244:2485-2486.
93. Li S, Long J, Ma Z, Xu Z, Li J, Zhang Z. Assessment of the therapeutic activity of a combination of almitrine and raubasine on functional rehabilitation following ischaemic stroke. Curr. Med. Res. Opin. 2004; 20:409-415.
94. Costa-Campos L, Iwu M, Elisabetsky E. Lack of pro-convulsant activity of the antipsychotic alkaloid alstonine. J. Ethnopharma- col. 2004; 93:307-310.
95. Hemscheidt T, Zenk MH. Partial purification and characterization of a NADPH dependent tetrahydroalstonine synthase from Catharanthus rosues cell suspension cultures. Plant Cell Rep. 1985; 4:216-219.
96. Stockigt J, Hemscheidt T, Hofle G, Heinstein P, Formacek V. Steric cours of hydrogen transfer during enzymatic formation of 3a-heteroyohimbine alkaloids. Biochemistry 1983; 22:3448-3452.
97. de Luca V. Biochemistry and molecular biology of indole alkaloid biosynthesis: the implication of recent discoveries. Rec. Adv. Phytochem. 2003; 37:181-202.
98. St. Pierre B, de Luca V. A cytochrome P-450 monooxygenase catalyzes the first step in the conversion of tabersonine to vindoline in Catharanthus roseus. Plant Physiol. 1995; 109:131-139.
99. Schroder G, Unterbusch E, Kaltenbach M, Schmidt J, Strack D, de Luca V, Schroder J. Light induced cytochrome P450 dependent enzyme in indole alkaoid biosynthesis: tabersonine 16-hydroxylase. FEBS Lett. 1999; 458:97-102.
100. Cacace S, Schroder G, Wehinger E, Strack D, Schmidt J, Schroder J. A flavonol O-methyltransferase from Catharanthus roseus performing two sequential methylations. Phytochemistry 2003; 62:127-137.
101. Dethier M, de Luca V. Partial purification of an N-methyltransferase involved in vindoline biosynthesis in Catharanthus roseus. Phytochemistry 1993; 32:673-678.
102. Vazquez-Flota F, de Carolis E, Alarco A, de Luca V. Molecular cloning and characterization of desacetoxyvindoline-4-hydroxylase, a 2-oxoglutarate dependent-dioxygenase involved in the biosynthesis of vindoline in Catharanthus roseus (L.) G. Don. Plant Mol. Biol. 1997; 34:935-948.
103. St. Pierre B, Laflamme P, Alarco A, de Luca V. The terminal O-acetyltransferase involved in vindoline biosynthesis defines a new class of proteins responsible for coenzyme A-dependent acyl transfer. Plant J. 1998; 14:703-713.
104. de Luca V, St. Pierre B. The cell and developmental biology of alkaloid biosynthesis. Trends Plant Sci. 2000; 5:168-173.
105. McKnight TD, Bergey DR, Burnett RJ, Nessler C. Expression of enzymatically active and correctly targeted strictosidine synthase in transgeneic tobacco plants. Planta 1991; 185:148-152.
106. Stevens LH, Blom TJM, Verpoorte R. Subcellular localization of tryptophan decarboxylase, strictosidine synthase and strictosidine glucosidase in suspension cultured cells of Catharanthus roseus and Tabernamontana divaricata. Plant Cell Rep. 1993; 12:573-576.
107. de Luca V, Cutler AJ. Subcellular localization of enzymes involved in indole alkaloid biosynthesis in Catharanthus roseus. Plant Physiol. 1987; 85:1099-1102.
108. de Carolis E, Chan F, Balsevich J, de Luca V. Isolation and characterization of a 2-oxoglutarate dependent dioxygenase involved in the second to last step in vindoline biosynthesis. Plant Physiol. 1990; 94:1323-1329.
109. Burlat V, Oudin A, Courtois M, Rideau M, St. Pierre B. Coexpression of three MEP pathway genes and geraniol 10 hydroxylase in internal phloem parenchyma of Catharanthus roseus implicates multicellular translocation of intermediates during the biosynthesis of monoterpene indole alkaloids and isoprenoid derived primary metabolites. Plant J. 2004; 38:131-141.
110. Murata J, de Luca V. Localization of tabersonine 16-hydroxylase and 16-OH tabersonine 16-O-methyl transferase to leaf epidermal cells defines them as a major site of precursor biosynthesis in the vindoline pathway in Catharanthus roseus. Plant J. 2005; 44:581-594.
111. Irmler S, Schroder G, St. Pierre B, Crouch NP, Hotze M, Schmidt J, Strack D, Matern U, Schroder J. Indole alkaloid biosynthesis in Catharanthus roseus: new enzyme activities and identification of cytochrome P450 CYP72A1 as secologanin synthase. Plant J. 2000; 24:797-804.
112. St. Pierre B, Vazquez-Flota F, de Luca V. Multicellular compart- mentation of Catharanthus roseus alkaloid biosynthesis predicts intercellular translocation of a pathway intermediate. Plant Cell 1999; 11:887-900.
113. Islam MN, Iskander MN. Microtubulin binding sites as target for developing anticancer agents. Mini-Rev. Med. Chem. 2004; 4:1077-1104.
114. Beckers T, Mahboobi S. Natural, semisynthetic and synthetic microtubule inhibitors for cancer therapy. Drugs Future 2003; 28:767-785.
115. Scott AI, Gueritte F, Lee SL. Role of anhydrovinblastine in the biosynthesis of the antitumor dimeric indole alkaloids. J. Am. Chem. Soc. 1978; 100:6253-6255.
116. Stuart KL, Kutney JP, Honda T, Worth BR. Intermediacy of 3’-4’-dehydrovinblastine in the biosynthesis of vinblastine type alkaloids. Heterocycles 1978; 9:1419-1426.
117. Baxster RI, Dorschel CA, Lee SL, Scott AI. Biosynthesis of the antitumor catharanthus alkaloids. Conversion of anhydrovinblastine into vinblastine. J. Chem. Soc. Chem. Comm. 1979:257-259.
118. Gueritte F, Bac NV, Langlois Y, Potier P. Biosynthesis of antitumour alkaloids from Catharanthus roseus. Conversion of 20’deoxyleurosidine into vinblastine. J. Chem. Soc. Chem. Comm. 1980:452-453.
119. Sottomayor M, Cardoso IL, Pereira LG, Barcel AR. Peroxidase and the biosynthesis of terpenoid indole alkaloids in the medicinal plant Catharanthus roseus (L.) G. Don. Phytochem. Rev. 2004; 3:159-171.
120. Endo T, Goodbody A, Vukovic J, Misawa M. Enzymes from Catharanthus roseus cell suspension cultures that couple vindoline and catharanthine to form 3’,4’-anhydrovinbastine. Phytochemistry 1988; 27:2147-2149.
121. Smith JL, Amouzou E, Yamagushi A, McLean S, DiCosmo F. Peroxidase from bioreactor cultivated Catharanthus roseus cell cultures mediate biosynthesis of a-3’,4’-anhydrovinblastine. Biotechnol. Appl. Bioeng. 1988; 10:568-575.
122. Hillou F, Costa M, Almeida I, Lopes Cardoso I, Leech M, Ros Barcelo A, Sottomayor M. Cloning of a peroxidase enzyme involved in the biosynthesis of pharmaceutically aciveterpenoid indole alkaloids in Catharanthus roseus. In: Proceedings of the VI International Plant Peroxidase Symposium. Acosta M, Rodriguez-Lopez JN, Pedreno MA, eds. 2002. pp. 152-158.
123. Sottomayor M, Lopez-Serrano M, DiCosmo F, Ros Barcelo A. Purification and characterization of anhydrovinblastine synthase (peroxidase like) from Catheranthus roseus. FEBS Lett. 1998; 428:299-303.
124. Goodbody AE, Endo T, Vukovic J, Kutney JP, Choi LSL, Misawa M. Enzymic coupling of catharanthine and vindoline to form 3/,4/-anhydrovinblastine by horseradish peroxidase. Planta Med. 1988; 54:136-140.
125. Canel C, Lopes-Cardoso MI, Whitmer S, Van der Fits L, Pasquali G, Van der Heijden R, Hoge JHC, Verpoorte R. Effects of over-expression of strictosidine synthase and tryptophan decarboxylase on alkaloid production by cell cultures of Catharanthus roseus. Planta 1998; 205:414-419.
126. Di Fiore S, Hoppmann V, Fischer R, Schillberg S. Transient gene expression of recombinant terpenoid indole alkaloid enzymes in Catharanthus roseus leaves. Plant Mol. Biol. Rep. 2004; 22:15-22.
127. Ayora-Talavera T, Chappell J, Lozoya-Gloria E, Loyola-Vargas VM. Overexpression in Catharanthus roseus hairy roots of a truncated hamster 3-hydroxy-3-methylglutaryl-CoA reductase gene. Appl. Biochem. Biotechnol. 2002; 97:135-145.
128. Peebles CAM, Hong S-B, Gibson SI, Shanks JV, San K-Y. Transient effects of overexpressing anthranilate synthase a and b subunits in Catharanthus roseus hairy roots. Biotech. Prog. 2005; 21:1572-1576.
129. Hedhili S, Courdavault V, Gigioli-Guivarc’h N, Gantet P. Regulation of the terpene moiety biosynthesis of Catharanthus roseus alkaloids. Phytochem. Rev. 2007; 6:341-351.
130. McCoy E, O’Connor SE. Directed biosynthesis of alkaloid analogues in the medicinal plant periwinkle. J. Am. Chem. Soc. 2006; 128:14276-14277.
131. Geerlings A, Redondo FJ, Contin A, Memelink J, Van der Heijden R, Verpoorte R. Biotransformation of tryptamine and secologanin into plant terpenoid indole alkaloids by transgenic yeast. Appl. Microbiol. Biotechnol. 2001; 56:420-424.
132. Menke FLH, Champion A, Kijne JW, Memelink J. A novel jasmonate- and elicitor-responsive element in the periwinkle secondary metabolite biosynthetic gene Str interacts with a jasmonate- and elicitor-inducible AP2-domain transcription factor, ORCA2. EMBO J. 1999; 18:4455-4463.
133. Van der Fits L, Memelink J. ORCA3, a Jasmonate-responsive transcriptional regulator of plant primary and secondary metabolism. Science 2000; 289:295-297.
134. Pauw B, Hilliou FAO, Martin VS, Chatel G, de Wolf CJF, Champion A, Pre M, Van Duijn B, Kijne JW, Van der Fits L, Memelink J. Zinc finger proteins act as transcriptional repressors of alkaloid biosynthesis genes in Catharanthus roseus. J. Biol. Chem. 2004; 279:52940-52948.
135. Montiel G, Breton C, Thiersault M, Burlat V, Jay-Allemand C, Gantet P. Transcription factor Agamous-like 12 from Arabidopsis promotes tissue-like organization and alkaloid biosynthesis in Catharanthus roseus suspension cells. Met. Eng. 2007; 9:125-132.
136. Humphrey AJ, O’Hagan D. Tropane alkaloid biosynthesis: a century old problem unresolved. Nat. Prod. Rep. 2001; 18:494-502.
137. Hemscheidt T. Tropane and related alkaloids. Top. Curr. Chem. 2000; 209:175-206.
138. Oksman-Caldenty KM, Arroo R. Regulation of tropane alkaloid metabolism in plants and plant cell cultures. In: Metabolic Engineering of Plant Secondary Metabolism. Verpoorte R, Alfermann AW, eds. 2000. Kluwer Academic Publishers, Dordrecht, the Netherlands. pp. 253-281.
139. Hibi N, Higashiguchi S, Hashimoto T, Yamada Y. Gene expression in tobacco low-nicotine mutants. Plant Cell 1994; 6:723-735.
140. Hibi N, Fujita T, Hatano M, Hashimoto T, Yamada Y. Putrescine N-methyltransferase in cultured roots of Hyoscyamus albus. Plant Physiol. 1992; 100:826-835.
141. Hashimoto T, Shoji T, Mihara T, Oguri H, Tamaki K, Suzuki KI, Yamada Y. Intraspecific variability of the tandem repeats in Nicotiana putrescine N-methyltransferases. Plant Mol. Biol. 1998; 37:25-37.
142. Suzuki K, Yamada Y, Hashimoto T. Expression of Atropa belladonna putrescine N-methyltransferase gene in root pericycle. Plant Cell Physiol. 1999; 40:289-297.
143. Teuber M, Azemi ME, Namjoyan F, Meier A-C, Wodak A, Brandt W, Drager B. Putrescine N-methyltransferases—a structure-function analysis. Plant Mol. Biol. 2007; 63:787-801.
144. McLauchlan WR, McKee RA, Evans DM. The purification and immunocharacterization of N-methylputrescine oxidase from transformed root cultures of Nicotinia tabacum. Planta 1993; 191: 440-445.
145. Haslam SC, Young TW. Purification of N-methylputrescine oxidase from Nicotiana rustica. Phytochemistry 1992; 31:4075-4079.
146. Katoh A, Shoji T, Hashimoto T. Molecular cloning of N-methylputrescine oxidase from tobacco. Plant Cell Physiol. 2007; 48:550-554.
147. Heim WG, Sykes KA, Hildreth SB, Sun J, Lu RH, Jelesko JG. Cloning and characterization of a Nicotiana tabacum methylpu- trescine oxidase transcript. Phytochemistry 2007; 68:454-463.
148. Heim WG, Jelesko JG. Association of diamine oxidase and S-adenosylhomocysteine hydrolase in Nicotiana tabacum extracts. Plant Mol. Biol. 2004; 56:299-308.
149. Hashimoto T, Nakajima K, Ongena G, Yamada Y. Two tropinone reductases with distinct stereospecificities from cultured roots of Hyoscyamus niger. Plant Physiol. 1992; 100:836-845.
150. Rocha P, Stenzel O, Parr A, Walton NJ, Christou P, Drager B, Leech MJ. Functional expression of tropinone reductase I and hyoscyaine-6b-hydroxylase from Hyoscyamus niger in Nicotiana tabacum. Plant Sci. 2002; 162:905-913.
151. Nakajima K, Yamashita A, Akama H, Nakatsu T, Kato H, Hashimoto T, Oda J, Yamada Y. Crystal structures of two tropinone reductases: different reaction stereospecificities in the same protein fold. Proc. Natl. Acad. Sci. 1998; 95:4876-4881.
152. Drager B. Chemistry and biology of calystegines. Nat. Prod. Rep. 2004; 21:211-223.
153. Nakajima K, Kato H, Oda J, Yamada Y, Hashimoto T. Site directed mutagenesis of putative substrate binding residues reveals a mechanism controlling different substrate specificities of two tropinone reductases. J. Biol. Chem. 1999; 274:16563-16568.
154. Yamashita A, Kato H, Wakatsuki S, Tomizaki T, Nakatsu T, Nakajima K, Hashimoto T, Yamada Y, Oda J. Structure of tropinone reductase-II with NADP + and pseudotropine at 1.9A resolution: implication for stereospecific substrate binding and catalysis. Biochemistry 1999; 38:7630-7637.
155. Li R, Reed DW, Liu E, Nowak J, Pelcher LE, Page JE, Covello PS. Functional genomic analysis of alkaloid biosynthesis in Hyoscyamus niger reveals a cytochrome P450 involved in littorine rearrangement. Chem. Biol. 2006; 13:513-520.
156. Oliagnier S, Kervio E, Retey J. The role and source of 5’-de-oxyadenosylradical in a carbon skeleton rearrangement catalyzed by a plant enzyme. FEBS Lett. 1998; 437:309-312.
157. Lanoue A, Boitel-Conti M, Portais JC, Laberche JC, Barbotin JN, Christen P, Sangwan-Norreel B. Kinetic study of littorine rearrangement in Datura innoxia hairy roots by 13C NMR spectroscopy. J. Nat. Prod. 2002; 65:1131-1135.
158. Patterson S, O’Hagan D. Biosynthetic studies on the tropane alkaloid hyoscyamine in Datura stramonium; hyoscyamine is stable to in vivooxidtion and is not derived from littorine via a vicinal interchange process. Phytochemistry 2002; 61:323-329.
159. Duran-Patron R, O’Hagan D, Hamilton JTG, Wong CW. Biosynthetic studies on the tropane ring system of the tropane alakloids from Datura stramonium. Phytochemistry 2000; 53:777-784.
160. Yamada Y, Hashimoto T. Substrate specificity of the hyoscyamine 6β-hydroxylase from cultured roots of Hyoscyamus niger. Proc. Japan Acad. B 1989; 65:156-159.
161. Hashimoto T, Kohno J, Yamada Y. 6b-Hydroxyhyoscyamine epoxidase from cultured roots of Hyoscyamus niger. Phytochemistry 1989; 28:1077-1082.
162. Matsuda J, Okabe S, Hashimoto T, Yamada Y. Molecular cloning of hyoscyamine 6β-hydroxylase, a 2-oxoglutarate-dependent dioxygenase, from cultured roots of Hyoscyamus niger. J. Biol. Chem. 1991; 266:9460-9464.
163. Hashimoto T, Yamada Y. Scopolamine production in suspension- cultures and redifferentiated roots of Hyoscyamus niger. Planta Med. 1983; 47:195-199.
164. Yun DJ, Hashimoto T, Yamada Y. Metabolic engineering of medicinal plants: transgenic Atropa belladonna with an improved alkaloid composition. Proc. Natl. Acad. Sci. 1992; 89:11799-11803.
165. Sato F, Hashimoto T, Hachiya A, Tamura K, Choi K-B, Morishige T, Fujimoto H, Yamada Y. Metabolic engineering of plant alkaloid biosynthesis. Proc. Natl. Acad. Sci. U.S.A. 2001; 98:367-372.
166. O’Hagan D, Robins RJ, Wilson M, Wong CW, Berry M. Fluorinated tropane alkaloids generated by directed biosynthesis in transformed root cultures of Datura stramonium. J. Chem. Soc. Perkins Trans. 1. 1999:2117-2120.
167. Boswell HD, Drager B, McLauchlan WR, Portseffen A, Robins DJ, Robins RJ, Walton NJ. Specificities of the enzymes of N-alkyltropane biosynthesis in Brugmansia and Datura. Phytochemistry 1999; 52:871-878.
168. Ashihara H, Crozier A. Caffeine: a well known but little mentioned compound in plant science. Trends Plant Sci. 2001; 6:407-413.
169. Crozier A, Ashihara H. The cup that cheers. Biochemist 2006; 28: 23-26.
170. Koshiishi C, Kato A, Yama S, Crozier A, Ashihara H. A new caffeine biosynthetic pathway in tea leaves: utilisation of adenosine released from the S-adenosyl-L-methionine cycle. FEBS Lett. 2001; 499:50-54.
171. Uefuji H, Ogita S, Yamaguchi Y, Koizumi N, Sano H. Molecular cloning and characterization of three distinct N-methyltransferases involved in the caffeine biosynthetic pathway in coffee plants. Plant Physiol. 2003; 132:372-380.
172. Mizuno K, Kato M, Irino F, Yoneyama N, Fujimura T, Ashihara H. The first committed step reaction of caffeine biosynthesis: 7-methylxanthosine synthase is closely homologous to caffeine synthase in coffee. FEBS Lett. 2003; 547:56-60.
173. Waldhauser SSM, Gillies FM, Crozier A, Baumann TW. Separation of the N-methyltransferase the key enzyme in caffeine biosynthesis. Phytochemistry 1997; 45:1407-1414.
174. Stoychev G, Kierdaszuk B, Shugar D. Xanthosine and xanthine: substrate properties with purine nucleoside phosphorylases and relevance to other enzyme systems. Eur. J. Biochem. 2002; 269:4048-4057.
175. Ogawa M, Herai Y, Koizumi N, Kusano T, Sano H. 7-methyl- xanthine methyltransferase of coffee plants. Gene isolation and enzymatic properties. J. Biol. Chem. 2001; 276:8213-8218.
176. Kato M, Mizuno K, Crozier A, Fumjimura T, Ashihara H. Plant biotechnology: caffeine synthase gene from tea leaves. Nature 2000; 406:956-957.
177. Yoneyama N, Morimoto H, Ye CX, Ashira H, Mizuno K, Kato M. Substrate specificity of N-methyltransferase involved in purine alkaloid synthesis is dependent on one amino acid residue of the enzyme. Mol. Gen. Genomics 2006; 275:125-135.
178. McCarthy AA, McCarthy JG. The structure of two N-methyltransferases from the caffeine biosynthetic pathway. Plant Physiol. 2007; 144:879-889.
179. Kim YS, Uefuji H, Ogita S, Sano H. Transgenic tobacco plants producing caffeine: a potential new strategy for insect pest control. Transgenic Res. 2006; 15:667-672.
180. Ashira H, Zheng XQ, Katahira R, Morimoto M, Ogita S, Sano H. Caffeine biosynthesis and adenine metabolism in transgenic Coffea canephora plants with reduced expression of N-methyltransferase genes. Phytochemistry 2006; 67:882-886.
181. Ogita S, Uefuji H, Morimoto H, Sano H. Application of RNAi to confirm theobromine as the major intermediate for caffeine biosynthesis in coffee plants with potential for construction of decaffeinated varieties. Plant Mol. Biol. 2004; 54:931-941.
182. Ogita S, Uefuji H, Yamaguchi Y, Koizumi N, Sano H. Producing decaffeinated coffee plants. Nature 2003; 423:823.
183. Satyanarayana KV, Kumar V, Chandrasheker A, Ravishankar GA. Isolation of promoter for N-methyltransferase gene associated with caffeine biosynthesis in Coffea canephora. J. Biotech- nol. 2005; 119:20-25.
Further Reading
Ashihara H, Crozier A., Caffeine: a well known but little mentioned compound in plant science. Trends Plant Sci. 2001; 6:407-413.
de Luca V, Laflamme PT. The expanding universe of alkaloid biosynthesis. Curr. Opin. Plant Biol. 2001; 4:225-233.
Hemscheidt T. Tropane and related alkaloids. Top. Curr. Chem. 2000; 209:175-206.
Humphrey AJ, O’Hagan D. Tropane alkaloid biosynthesis: a century old problem unresolved. Nat. Prod. Rep. 2001; 18:494-502.
Kutchan TM. Molecular genetics of plant alkaloid biosynthesis. In: The Alkaloids: Chemistry and Biology, vol. 50. Cordell GA, ed. 1998. Academic Press: San Diego, CA. pp. 261-263.
O’Connor SE, Maresh JM. Chemistry and biology of terpene indole alkaloid biosynthesis. Nat. Prod. Rep. 2006; 23:532-547.
See Also
Chemical Diversity of Natural Products in Plants
Enzymatic Synthesis of Biomolecules
Metabolic Engineering
Metabolic Profiling
Natural Products: An Overview