CHEMICAL BIOLOGY
Marine Natural Products, Chemical Diversity of
Nobuhiro Fusetani, Hokkaido University, Hakodate, japan
doi: 10.1002/9780470048672.wecb371
Oceans provide enormous and diverse habitats for marine life. The distinct feature of marine life is the domination of invertebrates, which account for more than 95% of marine animals. Most marine invertebrates are sessile and soft-bodied and lack obvious physical defenses. Instead, they have evolved to defend by chemical means against predation and overgrowth by other fouling organisms. In fact, their secondary metabolites have unusual structural features and potent biologic activities, many of which are not found in terrestrial natural products. This review focuses on bioactive metabolites isolated mainly from marine invertebrates with a special emphasis on the uniqueness of marine natural products.
The world’s oceans cover more than 70% of the earth’s surface and represent greater than 95% of the biosphere. Species ranging from 3 to 100 million are estimated to inhabit in the oceans. All but 1 of the 35 principal phyla in the animal kingdom are represented in aquatic environments; 8 phyla are exclusively aquatic. Most of them are sessile and soft-bodied organisms, most of which have evolved by chemical means to defend against predators and overgrowth by competing species. As expected, a variety of bioactive metabolites were found in marine invertebrates (see a series of reviews on marine natural products published in Natural Product Reports since 1984).
Bergmann’s revolutionary discovery of arabinose-containing nucleosides [e.g., spongouridine (1)] from the Caribbean marine sponge Cryptothera crypta was a driving force for the birth of a new research field, Marine Natural Products Chemistry, in the early 1970s (1, 2). Nearly 18,000 new compounds, including polyketides, peptides, alkaloids, terpenoids, shikimic acid derivatives, sugars, and a multitude of mixed biogenesis metabolites, have been discovered during the last 40 years according to MarinLit (University of Canterbury, New Zealand). Many marine natural products have structural features previously unreported from terrestrial sources (3). Halo- genated, especially brominated, and sulfated compounds are often encountered as marine natural products. Arsenic compounds, in particular arsenobetaines and arsenosugars, are distributed widely in marine algae and invertebrates. Recently, the first polyarsenic organic compound from Nature, arsenicin A (2) was reported from the sponge Echinochalina bargibanti (4). Also, several highly bioactive compounds with interesting modes of action have been discovered from marine invertebrates. A considerable percentage of these compounds was suggested or found to be derived from microorganisms. Indeed, certain bivalves and ciguateric fishes become poisonous by feeding toxic dinoflagellates or herbivorous ciguateric fish (5), whereas some cytotoxic metabolites of sponges and tunicates are produced by symbiotic microorganisms (6).
This review describes bioactive compounds isolated from marine algae and invertebrates with an emphasis on their uniqueness. Because of limited space, metabolites from bacteria, cyanobacteria, and fungi cannot be included, although some from cyanobacterial and endosymbiotic bacterial origins are described (some reviews on metabolites of marine bacteria, cyanobacteria, and fungi are provided in the “Further Reading” section). Structures and bioactivities are described for compounds that represent natural product classes, but steroids and carotenoids are not included.
Polyketides and Fatty Acid Derivatives
A diverse array of polyketide metabolites are found in marine organisms, which range from simple oxylipins to highly complex polyethers and macrolides. Particularly intriguing are “ladder-shaped” polyethers of dinofragellate origin and sponge macrolides.
Fatty acid derivatives
Oxylipins, which are a major metabolite of fatty acids, are observed regularly in seaweeds and opisthobrachs that feed on seaweeds. Aplydilactone (3), which is an unusual oxylipin isolated from the sea hare Aplysisa kurodai, activates phospholipase A2 (7). More than 50 halogenated C15 acetogenins, which are derived from fatty acids, have been isolated from red algae of the genus Laurencia; laurencin (4) was first isolated from L. nipponica (8). These compounds were reported to be antifeeding and insecticidal. Certain species of soft corals are known to contain prostanoids, of which the most intriguing are punaglandins [punaglandin 1 (5)], the first halogenated prostaglandins isolated from the soft coral Telesto riisei (3). They are antiviral and cytotoxic.
Fatty acid-derived cyclic peroxides are often found in marine sponges of the genus Plakortis; the first example was chondrillin (6). 1,2-Dioxane and 1,2-dioxolane carboxylates were also discovered from Plakortis sponges (9). These cyclic oxides show a range of biologic activities, for example, antimicrobial, cytotoxic, and antimalarial activities.
In addition, a variety of bioactive fatty acid derivatives have been isolated from marine organisms, including glycoceramides (10). Perhaps the most unusual example is a highly chlorinated sulfolipid 7 isolated as a cytotoxic principle from the mussel Mytilus galloprovincialis (10).
Polyacetylenes
Linear polyacetylenes are found frequently in marine sponges of the order Haplosclerida (11). Although the chain lengths vary from C14 to C49, likely they are to be derived from C16 fatty acids. Polyacetylenes show a wide range of bioactivities, including antimicrobial, cytotoxic, antiviral, and enzyme inhibitory. Corticatic acid A (8), which is a C31 polyacetylene carboxylic acid isolated from Petrosia corticata, is not only antimicrobial, but also it inhibits geranylgeranyltransferase I (12).
Polypropionates
The most prominent source of marine polypropionates are mollusks, in particular opisthobranchs (13). Among a variety of polypropionates, the simplest one is possibly siphonarienal (9) isolated from Siphonaria grisea, whereas such unusual pyrone-containing metabolites as siphonarin A (10) were isolated from S. zelandica. Marine polypropionates not only play defensive roles in mollusks, but also they show antimicrobial, antivial, and cytotoxic activities.
Polyethers
Dinoflagellates, which are unique, aquatic photosynthesizing eukaryotes, are classified in the kingdom Protoctista. They produce a variety of unusual polyketides with potent bioactivities (14). The most unique metabolites are “ladder-shaped” polyethers; brevetoxin B (11), which was isolated from Gymnodinium breve (currently, Karenia brevis), was the first example of this group, and more than 15 brevetoxins have been isolated from the same source. Ciguatoxin (12) is a causative agent of ciguatera, a fish poisoning in subtropical and tropical regions, first isolated from a moray eel. It was later found to be originated from the dinoflagellate Gambierdiscus toxicus that contains a more complex polyether, maitotoxin (13), which is also involved in ciguatera (5, 14). G. toxicus produces a variety of polyether compounds, namely highly antifungal gambieric acid (14). Maitotoxin, which is the most toxic natural product (LD50 value of 50 ng/kg in mice), increases membrane permeability to Ca2+, but the detailed mechanism remains unknown. Ciguatoxin is also highly toxic (1/10 potency of maitotoxin) and activates voltage-gated Na+ channels, whereas brevetoxins are much less toxic, although their mode of action is similar to that of ciguatoxin.
A different class of “ladder-shaped” polyethers, yessotoxins [e.g., yessotoxin 1 (15)], was isolated from toxic scallops; again its producer is the dinoflagellate Protoceratium reticulatum (5). The mode of action seems to be different from that of brevetoxins and ciguatoxins.
The sponge Halichondria okadai contains a polyether metabolite named okadaic acid (16) that was also isolated as a causative agent of diarrhetic shellfish poisoning (DSP) from mussels and other bivalves (14). However, the real producers are dinoflagellates of the genus Dinophysis. It is a potent cancer promoter that was found to be caused by inhibition of protein phosphatases 1 and 2A at nanomolar levels. Pectenotoxins are also involved in DSP and are produced by Dinophysis spp.; pectenotoxin 2 (17) inhibits actin polymerization (14).
Azaspiracids [azaspiracid-1 (18)] are another class of highly unusual polyketide polyethers originally isolated from Irish mussels that caused azaspiracid shellfish poisoning (5). They are produced by the dinoflagellate Protoperidinium crassipes. A similar class of polyether toxins named pinnatoxins [pinnatoxin A (19)] were reported from the bivalve Pinna pectinata; a closely related species P. attenuata is known to cause food poisoning in China. Pinnatoxins are likely of dinoflagellate origin and activate Ca2+ channels (15).
Open-chain polyketides
Dinoflagellates of the genus Amphidinium produce highly oxygenated, long-chain polyketides named amphidinols [amphidinol 3 (20)] that are highly antifungal and hemolytic (5). Palytoxin (21), which is found in zoanthids of the genus Palythoa, is an unusual polyketide as toxic as ciguatoxin (15). Its congeners were reported from the dinoflagellate Ostreopsis siamensis. Palytoxin is involved in several seafood poisonings and increases membrane permeability to Na+ by acting on Na+, K+-ATPase.
Discodermolide (22) is a polypropionate-based, unique compound isolated from the Caribbean deep-sea sponge Discodermia dissoluta. It is immunosuppressive as well as highly cytotoxic. More significantly, it stabilizes microtubules more potently than taxol (2).
Macrolides
A wide range of macrolides have been isolated from marine organisms, including dinoflagellates, sponges, bryozoans, and tunicates. Symbiotic dinoflagellates of the genus Amphidinium isolated from flat worms are the prolific source of highly cytotoxic macrolides (16). Amphidinolide N (23), which is a 26-membered macrolide, is the most potent, with an IC50 value of 0.05 ng/mL against L1210 leukemia cells, among the 34 amphidinolides isolated.
Sponges contain a diverse array of macrolides with intriguing activities. The first sponge macrolide, latrunculin A (24), was isolated from the Red Sea collection of Latrunculia magnifica as an ichthyotoxic compound and later was found to inhibit polymerization of G-actin allosterically. Swinholide A (25) is a macrodiolide originally isolated from the Red Sea sponge Theonella swinhoei. It is highly antifungal and cytotoxic, but its primary target is G-actin. Recently, it was discovered from cyanobacteria, although its eubacterial origin was predicted (6). Another class of macrolides that inhibit actin polymerization is the tris-oxazole-containing macrolides, namely kabiramides and mycaolides (1). Kabiramide C (26) was isolated from eggmasses of a nudibranch of the genus Hexabranchus, whereas mycalolides are of sponge origin, which indicates kabiramides were sequestered by nudibranchs from sponges. These macrolides are potently antifungal and cytotoxic. They bind G-actin in a molar ratio of 1:1, which thereby inhibits actin polymerization. Similar macrolides named aplyronines were isolated from the sea hare Aplysis kurodai; aplyronine A (27) shows remarkable antitumor activity (T/C > 500% at 0.08 mg/kg against P388 leukemia cells) as well as a similar mode of action to that of kabiramides (7). Presumably, aplyronines are of cyanobacterial origin.
Halichondrin B (28), which is a polyether macrolide isolated from H. okadai, shows promising antitumor activity and has entered phase I clinical trials (2, 15). It inhibits polymerization of tubulin by binding to the colchicine domain. The macrocyclic portion seems to be essential for the activity (2). Halichondrins have been found in several species of sponges, which indicates their microbial origin (6).
Spongistatins/hyrtiostatins/cinachyrolide A [spongistatin-1(29)] are highly unusual, 35-membered macrolides isolated from several sponges (1). They inhibit growth of tumor cells at sub-nM levels by binding to the vinca domain of tubulin. Their low yields and occurrence in several different sponges suggest their microbial origin. Spirastrellolide B (30) was reported recently as an antimitotic agent from the sponge Spirastrella coccinea. It is actually a potent and selective inhibitor of protein phosphatase 2A (12).
Peloruside A (31), which is a 16-membered, highly oxidized macrolide from the sponge Mycale hentscheli, induces tubulin polymerization (2). 13-Deoxytedanolide (32) isolated from Japanese sponges of the genus Mycale shows promising antitumor activity. It inhibits protein synthesis by binding to a 70 S large subunit of eukaryotic ribosome (17).
Bryostatins are medicinally important macrolides discovered from the cosmopolitan bryozoan Bugula neritina (2). Twenty bryostatins, which all possess a 20-membered ring, are known to date. Bryostatin 1 (33) showed good antitumor activity; it selectively modulates protein kinase C (12).
Phorboxazole A (34) is an unusual oxazole-containing macrolide isolated from a sponge Phorbas sp. (18). It is highly antifungal and cytostatic.
Salicylihalamide A (35) is a salicylic acid-containing macrolide enamide isolated from a sponge Haliclona sp., and it inhibits V-ATPases at a low nM concentration. Members of this family have been isolated from sponges, tunicates, and bacteria (12).



PKS/NRPS metabolites
Bengamides were isolated originally as anthelmintic agents from a sponge Jaspis sp. Later, bengamide A (36) was found to inhibit growth of tumor cells as well as methionine aminopeptidases (2).
Marine natural products of the pederin class (mycalamides, onnamides, and theopederins) isolated from sponges are mixed biogenesis metabolites of polyketide synthase and nonribosomal synthase (1). In fact, biosynthesis gene clusters of this class have been cloned recently using metagenomic techniques from the marine sponge Theonella swinhoei (6). These compounds are potently cytotoxic; theopederin A (37) inhibits protein synthesis in a similar mode of action to that of 13-deoxytedanolide.
Calyculin A (38) is an extraordinary metabolite composed of C28 fatty acid and two γ-amino acids isolated from the sponge Discodermia calyx. It is not only highly antifungal and antitumor but also a potent cancer promoter that was found to be caused by potent inhibition of protein phosphatases 1 and 2A (1). More than 15 calyculin derivatives were isolated from several marine sponges, which indicates the involvement of symbiotic microorganisms in the production of calyculins (6).
Pateamine (39) is a macrolide isolated from a marine sponge Mycale sp. Its potent cytotoxicity is attributed to inhibition of transcriptional initiation (19).



Aromatic polyketides and enediynes
Aromatic polyketides are rare metabolites in marine invertebrates. Naphthoquinone and anthraquinone derivatives were reported as echinoderm pigments in the earlier stage of marine natural products research. The most interesting aromatic polyketides are the pentacyclic polyketides of the halenaquinone/halenaquinol class. Halenaquinone (40) was first isolated from the sponge Xestospongia exigua. Compounds of this class show a variety of biologic activities, which include inhibition of tyrosine kinase (12, 19).
Only two groups of enediynes have been found from marine organisms; namenamicin (41) was first isolated the Fijian tunicate Polysyncraton lithostrotum, whereas shishijimicins A (42)-C, β-carboline-contining enediynes, were isolated from the Japanese tunicate Didemnum proliferum, along with 41, which thus suggests the involvement of symbiotic microorganisms in Amino acid derivatives their production (1). As expected, these compounds inhibited growth of tumor cells at pM levels.
Nonribosomal Peptides and Amino Acid Derivatives
In addition to ribosomal peptides, some of which exhibit interesting bioactivities as is the case of conotoxins, marine organisms; in particular, sponges and tunicates contain a wide variety of nonribosomal peptides, many of which contain unusual or unprecedented amino acids. It should be noted that these peptides show a range of biological activities.
Amino acid derivatives
Microalgae and macroalgae often contain UV-absorbing amino acids collectively dubbed “mycosporines.” Mycosporine-glycine (43) is most widely distributed in marine organisms that use it for protection from UV irradiation. Domoic acid (44) is not only a helminthic agent originally isolated from a red alga, but also it is a causative agent of amnesic shellfish poisoning (14). It is a potent agonist of glutamate receptors. Dysiherbaine (45), which is a novel betaine isolated from the sponge Dysidea herbacea, is a potent non-MNDA-type agonist with very high affinity for kainate receptors (20). Girolline (46), which is a potent cytotoxin isolated from the sponge Pseudaxinyssa cantharella, inhibits protein synthesis (2).
Bromotyrosine derivatives
Bromotyrosine-derived metabolites are often encountered in marine sponges of the families Aplysinidae and Pseudoceratidae, in particular Pseudoceratina (= Psammaplysilla) purpurea . They show a variety of biological activities, which include antimicrobial, enzyme inhibitory, and antifouling activities. Psammaplysin A (47) is antimicrobial, cytotoxic, and antifouling, whereas psammaplin A (48) is an inhibitor of histone deacetylase (2). The marine sponge Ianthella basta synthesizes at least 25 bastadins that are linear or cyclic peptides composed of four bromotyrosine residues [bastadin 5 (49)] and show antimicrobial, cytotoxic, and enzyme inhibitory activities as well as interaction with Ca2+ channels (21).
Linear peptides
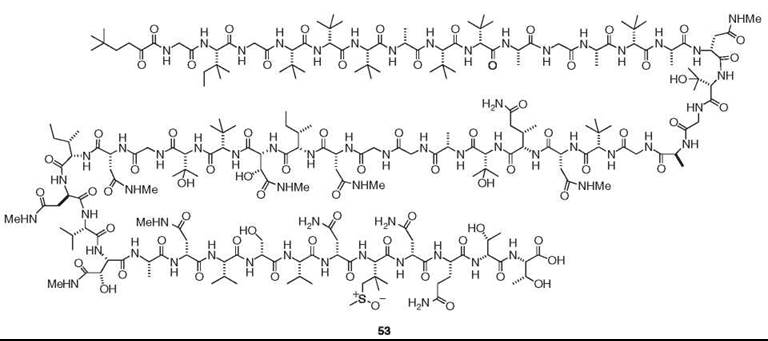
Sponges and tunicates frequently contain unusual linear and cyclic peptides; those from the former source were reviewed in 1993 and 2002 (22, 23). To avoid duplication, this review focuses on bioactive peptides isolated from other sources and new findings. Gymnangiamide (50) is the first described peptide from a hydroid (24). This pentapeptide from Gymnangium regae contains three previously unknown amino acids. Although moderately cytotoxic, its structure is reminiscent of dolastatin 10 (51), which is a powerful antitubulin agent of the sea hare Dolabella auricularia (2). Actually, dolastatins are of cyanobacterial origin. Dysinosin A (52) is a novel inhibitor of factor VIIa and thrombin, which was isolated from a new genus and species of Australian sponge of the family Dysideidae (12). Perhaps the most intriguing linear peptide is polytheonamide B (53), which is a potent cytotoxin discovered from T. swinhoei (25). It is composed of 48 amino acid residues, most of which are unusual amino acids. More significantly, it has the sequence of alternating D- and L-amino acids.



Cyclic peptides
Most cyclic peptides have been isolated from sponges and tunicates. Azumamides A (54) through E, which are cyclic tetrapeptides isolated from the sponge Mycale izuensis, are the most recent addition to the list of cyclic peptides. They strongly inhibit histone deacetylase (12). Dolastatin 11 (55), which is a cyclic depsipeptide isolated from D. auricularia, stabilizes actin filaments as in the case of jaspakinolide/jaspamide. Kahalalide F (56) is a cyclic depsipeptide isolated from the sacoglossan mollusk Elysia rufescens. It shows promising antitumor activity and has entered Phase II clinical trials, but its mode of action is not clear (2). Dolastatins and kahalalides likely are sequestered by the mollusks from cyanobacterial diets. Ascidian peptides often contain thiazole/thiazoline and oxazole/oxazoline amino acids as represented by patellamide A (57) isolated from Lissoclinum patella (3). These cyclic peptides show cytotoxic activity. Probably the most well-known ascidian peptide is didemnin B (58) isolated from Trididemnum solidum. This depsipeptide showed remarkable antitumor activity and entered clinical trials, but it was dropped because of side effects (2). It inhibits protein synthesis. Aplidine (dehydrodidemnin B) (59) isolated from Aplidium albicans is more promising as an anticancer drug, although it shows multiple modes of action (2).
Alkaloids
Marine organisms produce a wide range of alkaloids with potent bioactivities, which include such specific classes as 3-alkylpiperidine, guanidine, indole, polyamine, pyridoacridine, and pyrrole-imidazole alkaloids. Their biological activities vary from antimicrobial to neurological.
3-alkylpiperidines
A variety of 3-alkylpiperidine-derived compounds have been obtained from sponges belonging to five families of the order Haplosclerida (26). They show a range of bioactivities, for example, cytotoxic, antimalarial, and antifouling. It is likely that 3-alkylpiperidines are produced by sponge cells but not by symbiotic microorganisms.
The first 3-alkylpiperidine derivative reported is halitoxin (60), which was isolated from Haliclona sp. Similar polymeric alkylpyridines are also known from several sponges. In addition to the polymers, various types of metabolites of 3-alkylpyridines or 3-alkylpiperidines have been isolated, namely, macrocyclic bis-3-alkylpiperidine [telomerase-inhibitory cyclostellettamine A (61)] (12). Bis-quinolizadine [petrosin A (62)] and bis-1-oxaquinolizadine macrocycles [xestospongin C (63)] isolated from Petrosia seriata and Xestospongia spp., respectively, also belong to this group. The former is ichthyotoxic, whereas the latter is a potent vasodilator as well as an inhibitor of IP3 receptor. Halicyclamine (64), which was isolated from Haliclona sp., is another group of macrocyclic bis-3-alkylpiperidines, whereas sarain A (65), which was isolated from Reniera sarai, has a more complex polycyclic core. These compounds are moderately cytotoxic.
The most well-known group of 3-alkylpiperidine alkaloids is the manzamines. Manzamine A (66), which is the first member of this group, was isolated from an Okinawan Haliclona sp. More than 20 manzamines have been isolated from sponges of eight different genera. Manzamine A is highly cytotoxic, antituberculosis, and antimalarial, but its mode of action remains unknown (27).
Pyrrole-imidazoles
The pyrrole-imidazole alkaloids are found exclusively in marine sponges, in particular in the families Agelasidae, Axinellidae, and Halichondridae (28). Oroidin (67), which was discovered from Agelas oroides, is the building block of about 100 metabolites of this family, which can be divided into those derived from different modes of cyclization, for example, (-)-dibromophakelin (68) isolated from Phakellia flabellate and ageladine A (69) from Agelas nakamurai, and those derived from different modes of dimerization, namely sceptrin (70), which is isolated from Agelas sceptrum, palau’amine (71) from Stylotella aurantium and massadine (72) from Stylissa massa. Quite recently, a dimer of massadine (oridin tetramer) named stylissadine A was isolated from Stylissa caribica. The pyrrole-imidazole alkaloids show a range of bioactivities, which include antimicrobial, cytotoxic, antagonistic to receptors, immnosuppressive, enzyme inhibitory, and antifouling.
Pyridoacridines and related alkaloids
Pyridoacridines are highly colored polycyclic alkaloids mainly isolated from sponges and tunicates (29). They are divided into four groups, the simplest of which is styelsamine D (73), which was isolated from the ascidian Eusynstyela latericius. The pentacyclic pyridoacridines are also classified into two groups as represented by amphimedine (74), which was isolated from a sponge Amphimedon sp., and ascididemin (75), which was isolated from a tunicate Didemnum sp., respectively. Dercitin (76) is a thiazole-containing pentacyclic alkaloid isolated from a sponge Dercitus sp., whereas cyclodercitin (77), which was isolated from a sponge Stelletta sp., is a member of the hex acyclic group. The most complex octacyclic pyridoacridines are represented by eudistone A (78), which was isolated from Eudistoma sp.
Alkaloids related to pyridoacridines are known also from sponges and tunicates. Discorhabdin C (79), which is the first marine pyrroloquinoline alkaloid, was isolated from the sponge Latrunculia cf. bocagei. More than 20 alkaloids of this class are known at moment (30). Additional examples of this family are batzelline A (80) from a deep-sea sponge of the genus Batzella and wakayin (81) from an ascidian Clavelina sp.
Marine pyridoacridines show a wide range of biological activities, namely antimicrobial, antiviral, antiparasitic, insecticidal, antitumor, and enzyme inhibitory (12, 29, 30).
Pyrrole alkaloids
Highly substituted pyrroles are often found in sponges, ascidians, and mollusks (30). Lamellarins A (82)-D are the first metabolites of this group that were reported from a mollusk Lamellaria sp., followed by the discovery of more than 50 alkaloids of this class from sponges and ascidians. Polycitone A (83) was isolated from a tunicate Polysitor sp., whereas storniamide A (84) was isolated from a sponge of the genus Cliona. The most recent addition to this class is dictyodendrins A (85) through E isolated as telomerase inhibitors from the sponge Dictyodendrilla verongiformis (12). Various bioactivities have been reported for lamellarins and related compounds, which include antitumor, antiviral, and enzyme inhibitory.
Indole alkaloids
Many indole-containing metabolites have been reported from marine organisms, some of which were already mentioned. This section focuses on important indole-containing substances that belong to structural classes not mentioned above. These alkaloids show antimicrobial, antiparasitic, antitumor, and enzyme inhibitory activities (27). Dragmacidin (86) is a novel bis -indole isolated from a deep-sea sponge Dragmacidon sp., whereas another deep-sea sponge Spongosorites ruetzleri contains a similar bis -indole, topsentin (87).
β-Carboline-containing metabolites are known from sponges and tunicate. Eudistomins were the first β-carboline alkaloid isolated from marine organisms. Eudistomin K (88), which is a novel oxathiazepine ring containing Pcarboline, was isolated from the tunicate Eudistoma olivaceum, whereas a guanidine-containing grossularine 1 (89) was from the tunicate Dendrodoa grossulatia (27).
Physostigmine alkaloids are often contained in bryozoans. The highly unusual constituent is securine A (90) isolated from Securiflustra securifrons from the North Sea. Another interesting physostigmine is urochordamine A (91) isolated as a larval settlement inducer from the ascidian Ciona savignyi (30). Neosurugatoxin (92), which is a causative agent of “ivory shellfish poisoning” isolated from the gastropod Babylonia japonica, is a reversible nicotinic acetylcholine antagonist (14). Its bacterial origin has been suggested. Finally, diazonamide A (93) is a highly unusual cytotoxic metabolite of the tunicate Diazona angulata. It inhibits tubulin polymerization (2).

Guanidine alkaloids
Many guanidine-containing compounds have been reported from diverse marine organisms (31). No doubt exists that the most well-known guanidine alkaloid is tetrodotoxin (94), which is a causative agent of puffer fish poisoning (14). It is highly toxic because of inhibition of voltage-gated Na+ channels. Production of tetrodotoxin by bacteria of such genera as Pseudoalteromonas and Vibrio was reported. Similarly, saxitoxin (95) causes paralytic shellfish poisoning. Bivalves accumulate the toxin from dinoglagellates, for example, Alexandrium catenella, A. tamarense, and Gymnodinium catenatum. Its mode of action is similar to that of tetrodotoxin. More than 30 saxitoxin derivatives are known to date.
Ptilomycalin A (96) is a novel polycyclic guanidine alkaloid isolated from the sponge Ptilocaulis spiculifer (31). It is highly cytotoxic, antifungal, and antiviral. The related alkaloids were reported from the Mediterranean sponge Crambe crambe that also contains other types of guanidine alkaloids such as crambescin A (97). Batzelladines are a similar class of alkaloids isolated from a sponge Batzella sp.; batzelladine A (98) shows anti-HIV activity.
Variolin B (99), which is a pyridopyrropyrimidine alkaloid isolated from the sponge Kirkpatrichia varialosa, is strongly cytotoxic, antifungal, and antiviral (30). It inhibits cyclin- dependent kinases (12).
Polyamine alkaloids
Stellettadine A (100), which is isolated from a sponge Stelletta sp., represents the first polyamine alkaloid with an arcaine backbone (32). Similar alkaloids [e.g., stellettazole A (101)] are also known from the same source. These compounds show larval settlement-inducing, antifungal, and enzyme inhibitory activities. Isoprenylated polyamines are encountered occasionally as metabolites of soft corals as represented by sinulamide (102), which inhibits H,K-ATPase (12).
Quinolines and isoquinolines
Aaptamine (103), which is a cytotoxic benzonaphthyridine alkaloid isolated from the sponge Aaptos aaptos, induces differentiation in chronic leukemia cells (33). Schulzeine A (104) is a novel dihydroquinoline alkaloids isolated from the sponge Penares schulzei that inhibits glycosidases (12).
Sponges and tunicates contain tetrahydroisoquinoline alkaloids (30). Renieramycin A (105) from a sponge Reniera sp. represents the first example of this particular class of alkaloids isolated from marine organisms. Ecteinascidins, in particular ecteinascidin 743 (106), which was isolated from the tunicate Ecteinascidia turbinata, are promising as anticancer leads. Ecteinascidin 743 cleaves DNA chains and is in clinical trials (2).
Quinolizidines and indolizidines
Only a few compounds of these classes of alkaloids have been reported from marine organisms. Clavepictine B (107), which was isolated from the ascidian Clavelinapicta, is marginally cytotoxic. Stellettamine A (108), which is an indolizidine derivative from a sponge Stelletta sp., is antifungal and cytotoxic. It also inhibits calmodulin (12). Lepadiformine (109), which has a similar structural feature isolated from the tunicate Clavelina inoluccensis, inhibits K+ channels (33).



Steroidal alkaloids
Steroidal alkaloids belong to a rare class of marine natural products. Plakinamine A (110), which is an antimicrobial metabolite from a sponge Plakina sp., is the first steroidal alkaloid isolated from marine organisms. Cephalostatins and ritterazines are unprecedented dimeric steroidal alkaloids isolated from the hemichordate Cephalodiscus grichristii and the tunicate Ritterella tokioka, respectively (1, 30) .They are specific to marine metabolites. Cephalostatin 1 (111) and ritterazine B (112) are highly cytotoxic, but their mode of action remains to be elucidated.
Although unlikely steroids or triterpenoids, zoanthamines found in zoanthids of the genus Zoanthus should be mentioned here. Zoanthamine (113) represents the first example of 10 metabolites of this class. Norzoanthamine has been best studied and found to suppress the production of IL-6 (15).
Terpenoids
Although those similar to terrestrial terpenoids are found commonly in marine organism, in particular in algae, several terpenoids with new or modified skeletons have been isolated frequently from algae, sponges, and cnidarians. Halogenated terpenoids are often contained in algae, whereas sulfated terpenoids and steroids are distributed widely in sponges.
Monoterpenoids and Sesquiterpenoids
Red algae contain highly halogenated monoterpenoids such as 114, which is an antifeeding constituent isolated from Plocamium cartilagineum (34, 35).
Several halogenated sesquiterpenes with various skeletal types were reported from red algae of the genus Laurencia, for example, elatol (115), which is a chamigrane sesquiterpene that has antifeeding and antifouling activities from L. obtusa. A series of linear and cyclic sesquiterpenes with an 1,4-diacetoxybutadiene functionality [e.g. caulerpenyne (116)] have been isolated as antifeeding agents from green algae of the order Caulerpales (35). Coelenterates are a rich source of sesquiterpenes of 20 skeletal types, which include ∆9(12)-capnellane (117), an antifeeding metabolite isolated from Capnella imbricata (36).
Sponges produce furanosesquiterpenes of various skeletal types, such as furodysinin (118) from Dysidea herbacea and nakafuran-8 (119) from D. etheria, the latter of which is antifeeding. Similarly, mixed shikimate-mevalonate metabolites are often encountered as sponge metabolites; the first example is avarol (120), which was isolated from Dysidea avara. Several related compounds have been isolated from dictyoceratid sponges. They show a wide range of bioactivities, for example, antimicrobial, antitumor, anti-inflammatory, and enzyme inhibitory.


Diterpenoids
Brown algae are rich in diterpenoids, which can be classified into three groups, namely “xenicanes,” “extended sesquiterpenes,” and “dolabellanes,” which are represented by dictyotadial (121) from Dictyota crenulata, spatol (122) from Spatoglossum schmittii, and amijidictyol (123) from Dictyota linearis, respectively. These terpenes are involved in chemical defense. Red algae contain brominated diterpenes of several established skeletal types.
Spongian diterpenes are a chemical marker for dictyoceratid and dendroceratid sponges (37). The first example of a spongian diterpene is isoagatholactone (124) isolated from Spongia officinalis. In addition to those possessing a basic spongian skeleton, a wide variety of rearranged spongian diterpenoids have been reported, which include gracilin B (125) from Spongionella gracilis. Many spongian diterepenes are antimicrobial and cytotoxic.
Again, soft corals and gorgonians are a rich source of diterpenoids of 19 structural classes, some of which are specific to them (35, 36). Besides tobacco plants, cembranoid diterpenes are limited to soft corals. Lophotoxin (126) isolated from sea whips of the genus Lophogorgia is a sodium channel inhibitor (3). Xenicin (127) from the soft coral Xenia elongata and briarein A (128) from the gorgonian Briareum asbestinum represent non-cembranolide diterpenes. Diterpenoids of these classes show antimicrobial, cytotoxic, and insecticidal activities. Pseudopterosin A (129), which is a diterpene glycoside isolated from the sea whip Pseudopterogorgia elisabethae, shows anti-inflammatory activity by inhibiting release of leukotriene B2 from leukocytes (2, 35). A more interesting class of diterpenes includes sarcodictyn A (130) and eleutherobin (131), which were isolated from the soft corals Sarcodictyon roseum and Eleutherobia sp., respectively. These diterpenoids show potent cytotoxicity by stabilizing microtubules (2, 35).
Ageladine A (132), which is a 9-methyladenine derivative of diterpene isolated from a sponge Agelas sp., is antimicrobial and inhibitory against Na, K-ATPase (12). A novel, chlorinated diterpenoid, chlorolissoclimide (133), which was isolated from the ascidian Lissoclinum forskalii, inhibits eukaryotic protein synthesis (38).

Isocyanoterpenes and related terpenoids
Isocyanide-containing natural products are rare; they have been reported only from cyanobacteria, Penicillium fungi, marine sponges, and nudibranchs (35, 39). Sesquiterpenoid and diterpenoid isocyanides are found in a limited species of sponges and nudibranchs that prey on these sponges. Axisonitrile-1 (134) isolated from the sponge Axinella cannabina is the first isocyanide-containing marine natural product. Isocyanopupukeanane (135) was isolated originally from the nudibranch Phyllidia verrucosa and later from a sponge Ciocalypta sp. Kalihinol A (136) and 7,20-diisocyanoadocane (137) were isolated from the sponges Acanthella carvenosa and Adocia sp., respectively. Isocyanoterpenes are often accompanied by thiocyanates, isothiocyantes, and formamides. These terpenoids show a wide range of bioactivities, which include antimicrobial, cytotoxic, ichthyotoxic, antifouling, and antimalarial. The carbonimidic dichloride group is considered to be equivalent to isocyanide, and the first sesquiterpenoid that contains this moiety 138 was isolated from the sponge Pseudoaxinyssa pitys. Terpenoids that contain carbonimidic dichloride show similar bioactivities to those of isocyanide counterparts.
Sesterterpenoids, triterpenoids, and steroids
Variabilin (139), which is a C25 tetronic acid isolated from the sponge Ircinia valiabiris, represents the class of furanosesterterpenes derived from sponges (34). Bioactivities include antimicrobial, cytotoxic, antifouling, and antifeedant.
Manoalide (140) is perhaps the most well-known marine sesterterpene isolated from the sponge Luffariella variabilis. It is antimicrobial, anti-inflammatory, and PLA2 inhibitory (2, 12, 35). Several sesterterpenes of the scalarane class [e.g., scalaradial (141)] have been isolated from dyctioceratid sponges, many of which show anti-inflammatory activity (12). Dysidiolide (142), which is an unusual sesterterpene isolated from the sponge Dysidea etheria, is the first natural product inhibitor of cdc25A (12). Mycaperoxide A (143), which is a norsesterterpene peroxide isolated from Mycale sp., showed promising antimalarial activity (9).
Probably one of the most interesting marine triterpenoids is the squalene-derived polyethers found in red algae of the genus Laurencia. Among the nearly 30 metabolites of this class, thyrsiferyl 23-acetate (144) shows the most potent cytotoxicity (40). It selectively inhibits protein phosphatase 2A. The other class of triterpenoid polyethers has been isolated from sponges of the families Halichondridae and Axinellidae (40). Sipholenone B (145), which was isolated from Siphonochalina siphonella, represents the first example of this class. Adociasulfate-2 (146) is a member of the triterpenoid hydroquinone sulfates isolated from a sponge Haliclona (aka Adocia) sp. that is the only known natural product inhibitor of kinesin (12, 19).
Didemnaketal C (147) is an unusual heptaprenoid isolated from an ascidian Didemnum sp.; its methanolysis product, didemnanaketal B (148), inhibited HIV-1 protease (12).
Sugars
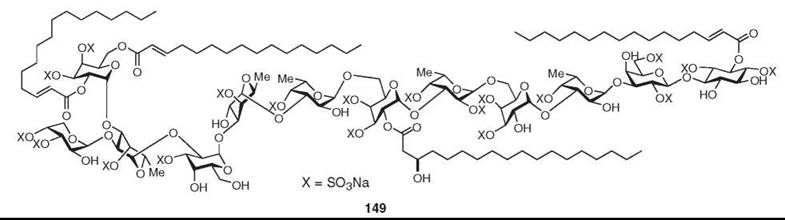
Several unusual polysaccharides have been isolated from sponges (11), among which the most unusual is axinelloside A (149), which has been isolated recently as a potent telomerase inhibiter from Axinella infundibula (12). It resembles bacterial lipopolysaccharides.
Acknowledgments
I would like to thank Otto Hensens for editing the manuscript.
References
1. Fusetani N. Search for drug leads from Japanese marine invertebrates. J. Synth. Org. Chem. Jpn. 2004; 62:1073-1079.
2. Newman DJ, Cragg GM. Marine natural products and related compounds in clinical and advanced preclinical trials. J. Nat. Prod. 2004; 67:1216-1238.
3. Ireland CM, Roll DM, Molinski TF, McKee TC, Zabriskie TM, Swersey JC. Uniqueness of the marine chemical environment: categories of marine natural products from invertebrates. Biomedical Importance of Marine Organisms. Fautin DG, ed. 1988. California Academy of Sciences, San Francisco, CA. pp. 41-57.
4. Mancini I, Guella G, Frostin M, Hnawia E, Laurent D, Debitus C, Pietra F. On the first polyarsenic organic compound from nature: arsenicin A from the New Caledonian marine sponge Echinochalina bargibanti. Chem. Eur. J. 2006; 12:8989-8994.
5. Yasumoto T. Chemistry, etiology, and food chain dynamics of marine toxins. Proc. Japan Acad., Ser. B. 2005; 81:43-51.
6. Piel J. Bacterial symbionts: prospects for the sustainable production of invertebrate-derived pharmaceuticals. Curr. Med. Chem. 2006; 13:39-50.
7. Yamada K, Kigoshi H. Bioactive compounds from the sea hares of two genera: Aplysia and Dolabella. Bull. Chem. Soc. Jpn. 1997; 70:1479-1489.
8. Suzuki M, Vairappan CS. Halogenated secondary metabolites from Japanese species of the red algal genus Laurencia (Rhodomelaceae, Ceramiales). Curr. Topic. Phytochem. 2005; 7:1-34.
9. Casteel DA. Peroxy natural products. Nat. Prod. Rep. 1999; 16:55-73.
10. Costantino V, Fattorusso E, Menna M, Taglialatela-Scafati O. Chemical diversity of bioactive marine natural products: an illustrative case study. Curr. Med. Chem. 2004; 11:1671-1692.
11. Van Soest RWM, Fusetani N, Andersen RJ. Straight-chain acetylenes as chemotaxonomic markers of the marine Haplosclerida. In: Sponge Sciences Multidisciplinary Perspectives. Watanabe Y, Nobuhiro F, eds. 1998. Springer-Verlag, Tokyo. pp. 3-30.
12. Nakao Y, Fusetani N. Enzyme inhibitors from marine invertebrates. J. Nat. Prod. 2007; 70:689-710.
13. Davies-Coleman MT, Garson MJ. Marine polypropionates. Nat. Prod. Rep. 1998; 15:477-493.
14. Yasumoto T, Murata M. Marine toxins. Chem. Rev. 1993; 93:1897-1909.
15. Uemura D. Bioorganic studies on marine natural products - diverse chemical structures and bioactivities. Chem. Record 2006; 6:235-248.
16. Kobayashi J, Kubota T. Bioactive macrolides and polyketides from marine dinoflagellates of the genus Amphidinium. J. Nat. Prod. 2007; 70:451-460.
17. Nishimura S, Matsunaga S, Yoshida M, Hirota H, Yokoyama S, Fusetani N. 13-Deoxytedanolide, a marine sponge-derived antitumor macrolide, binds to the 60S large ribosomal subunit. Bioorg. Med. Chem. 2005; 13:449-454.
18. Molinski TF. Antifungal compounds from marine organisms. Curr. Med. Chem. Anti-Infective Agents 2004; 3:197-220.
19. Nagle DG, Zhou Y-D, Mora FD, Mohammed KA, Kim Y-P, Mechanism targeted discovery of antitumor marine natural products. Curr. Med. Chem. 2004; 11:1725-1756.
20. Sakai R, Kamiya H, Murata M, Shimamoto K. Dysiherbaine: a new neurotoxic amino acid from the Micronesian marine sponge Dysidea herbacea. J. Am. Chem. Soc. 1997; 119:4112-4116.
21. Couladopuros EA, Pitsinos EN, Moutsos VI, Sarakinos G. A general method for the synthesis of bastaranes and isobastaranes: first total synthesis of bastadins 5, 10, 12, 16, 20, and 21. Chem. Eur. J. 2005; 11:406-421.
22. Fusetani N, Matsunaga S. Bioactive sponge peptides. Chem. Rev. 1993; 93:1793-1806.
23. Matsunaga S, Fusetani N. Nonribosomal peptides from marine sponges. Curr. Org. Chem. 2003; 7:945-966.
24. Milanowski DJ, Gustafson KR, Rashid MA, Rannell LK, McMahon JB, Boyd MR. Gymnangiamide, a cytotoxic pentapeptide from the marine hydroid Gymnangium regae. J. Org. Chem. 2004; 69:3036-3042.
25. Hamada T, Matsunaga S, Yano G, Fusetani N. Polytheonamides A and B, highly cytotoxic, linear polypeptides with unprecedented structural features, from the marine sponge, Theonella swinhoei. J. Am. Chem. Soc. 2005; 127:110-118.
26. Andersen RJ, van Soest RWM, Kong F. 3-Alkylpiperidine alkaloids isolated from marine sponges in the order Haplosclerida. Alakloids: Chem. Biol. Perspect. 1996; 10:301-355.
27. Gul W, Hamann MT. Indole alkaloid marine natural products: an established source of cancer drug leads with considerable promise for the control of parasitic, neurological and other diseases. Life Sci. 2005; 78:442-453.
28. O’Malley DP, Li K, Maue M, Zografos AL, Baran PS. Total synthesis of dimeric pyrrole-imidazole alkaloids: sceptrin, ageliferin, nagelamide E, oxysceptrin, nakamuric acid, and the axinellamine carbon skeleton. J. Am. Chem. Soc. 2007; 129:4762-4775.
29. Delfourne E, Bastide J. Marine pyridoacridine alkaloids and synthetic analogues as antitumor agents. Med. Res. Rev. 2003; 23:234-252.
30. Urban S, Hickford SJH, Blunt JW, Munro MHG. Bioactive marine alkaloids. Curr. Org. Chem. 2000; 4:765-807.
31. Berlinck RGS. Natural guanidine derivatives. Nat. Prod. Rep. 1999; 16:339-365.
32. Bienz S, Bisegger P, Guggisberg A, Hesse M. Polyamine alkaloids. Nat Prod. Rep. 2005; 22:647-658.
33. Folmer F, Houssen WE, Scott RH, Jaspars M. Biomedical research tools from the seabed. Curr. Opin. Drug Discov. Dev. 2007; 10:145-152.
34. Faulkner DJ. Interesting aspects of marine natural products chemistry. Tetrahedron 1977; 33:1421-1443.
35. Gross H, Konig GM. Terpenoids from marine organisms: unique structures and their pharmacological potential. Phytochem. Rev. 2006; 5:115-141.
36. Coll JC. The chemistry and chemical ecology of octocorals (Coe- lenterata, Anthozoa, Octocorallia). Chem. Rev. 1992; 92:613-631.
37. Keyzers RA, Northcote PT, Davies-Coleman MT. Spongian diterpenoids from marine sponges. Nat. Prod. Rep. 2006; 23:321-334.
38. Robert F, Gao HQ, Donia M, Merrick WC, Hamann MT, Pelletier J. Chlorolissoclimides: new inhibitors of eukaryotic protein synthesis. RNA 2006; 12:717-725.
39. Garson MJ, Simpson JS. Marine isocyanides and related natural products - structure, biosynthesis and ecology. Nat. Prod. Rep. 2004; 21:164-179.
40. Fernandez JJ, Souto ML, Norte M. Marine polyether triterpenes. Nat. Prod. Rep. 2000; 17:235-246.
Further Reading
Allingham JS, Klenchin VA, Rayment I. Actin-targeting natural products, Properties and mechanisms of action. Cell. Mol. Life Sci. 2006; 63:2119-2134.
Antunes EM, Copp BR, Davies-Coleman MT, Samaai T. Pyrroloiminoquinone and related metabolites from marine sponges. Nat. Prod. Rep. 2005; 22:62-72.
Bandaranayake WM. The nature and role of pigments of marine invertebrates. Nat. Prod. Rep. 2006; 23:223-255.
Blunt JW, Copp BR, Hu WP, Munro MHG, Northcote PT, Princep MR. Marine natural products. Nat. Prod. Rep. 2007; 24:31-86.
Bowman EJ, Bowman BJ. V-ATPases as drug targets. J. Bioenerget. Biomembr. 2005; 37:431-435.
Bugni TS, Ireland CM. Marine-derived fungi: a chemically and biologically diverse group of microorganisms. Nat. Prod. Rep. 2004; 21:143-163.
Burja AM, Banaigs B, Abou-Mansour E, Burgess JG, Wright PC. Tetrahedron 2001; 57:9347-9377.
Chang CWJ. Naturally occurring isocyano/iothiocyanato and related compounds. Fortsch. Chem. Org. Naturst. 2000; 80:1-186.
Crews P, Hunter LM. The search for antiparasitic agents from marine animals. In: Marine Biotechnology, vol 1. Pharmaceutical and Bioactive Natural Products, vol. 1. Attaway DH, Zaborsky OR, eds. 1993. Plenum Press, New York. pp. 343-390.
D’Auria MV, Minale L, Riccio R. Polyoxygenated steroids of marine origin. Chem. Rev. 1993; 93:1839-1895.
Daranas AH, Norte M, Fernandez JJ. Toxic marine microalgae. Toxicon 2001; 39:1101-1132.
Dembitsky VM, Levitsky DO. Arsenolipids. Prog. Lipid Res. 2004; 43:403-448.
Donia M, Hamann MT. Marine natural products and their potential applications as anti-infective agents. Lancet Infect. Dis. 2003; 3:338-348.
Faulkner DJ. Marine natural products. Nat. Prod. Rep. 2002; 19:1-48, and earlier reviews cited within.
Fenical W, Jensen PR. Developing a new resource for drug discovery: marine actinomycete bacteria. Nat. Chem. Biol. 2006; 2:666-673.
Gerwick WH, Bernart MW. Eicosanoids and related compounds from marine algae. In: Marine Biotechnology, vol 1. Pharmaceutical and Bioactive Natural Products, vol 1. Attaway DH, Zaborsky OR, eds. 1993. Plenum Press, New York. pp. 101-152.
Gerwick WH, Nagle DG, Proteau PJ. Oxylipins from marine invertebrates. Topics Curr. Chem. 1993; 167:117-180
Gribble GW. The diversity of naturally occurring organobromine compounds. Chem. Soc. Rev. 1999; 28:335-346.
Heys L, Moore CG, Murphy PJ. The guanidine metabolites of Ptilocaulis spiculifer and related compounds; isolation and synthesis. Chem. Soc. Rev. 2000; 29:57-67.
Ireland CM, Copp BR, Foster MP, McDonald LA, Radisky DC, Swersey JC. Biomedical potential of marine natural products. In: Marine Biotechnology, vol 1. Pharmaceutical and Bioactive Natural Products, vol 1. Attaway DH, Zaborsky OR, eds. 1993. Plenum Press, New York. pp. 1-76.
Janin YL. Peptides with anticancer use or potential. Amino Acids 2003; 25:1-40.
Jensen PR, Fenical W. Marine microorganisms and drug discovery: current status and future potential. In: Drugs from the Sea. Fusetani N, ed. 2000. Karger, Basel pp. 6-29.
Kobayashi J, Ishibshi M. Marine natural products and marine chemical ecology. Comp. Nat. Prod. Chem. 1999; 8:415-649.
Kornprobst JM, Sallenave C, Barnathan G. Sulfated compounds from marine organisms. Comp. Biochem. Physiol. 1998; 119B:1-51.
Lewis RJ, Garcia ML. Therapeutic potential of venom peptides. Nat. Rev. Drug Discov. 2003; 2:790-802.
Llewellyn LE. Saxitoxin, a toxic marine natural product that targets a multitude of receptors. Nat. Prod. Rep. 2006; 23:200-222.
Marshall KM, Barrows LR. Biological activities of pyridoacridines. Nat. Prod. Rep. 2004; 21:731-751.
Minale L, Cimino G, de Stefano S, Sodano G. Natural products from Porifera. Fortsch. Chem. Org. Naturst. 1976; 33:1-72.
Minale L, Riccio R, Zollo F. Steroidal oligosaccharides and polyhydorxylated steroids from echinoderms. Fortsch. Chem. Org. Naturst. 1993; 62:75-308.
Moore BS. Biosynthesis of marine natural products: macroorganisms (Part B). Nat. Prod. Rep. 2006; 23:615-629.
Mourabit AA, Potier P. Sponge’s molecular diversity through the ambivalent reactivity of 2-aminoimidazole: a universal chemical pathway to the oroidin-based pyrrole-imidazole alkaloids and their palau’amine congeners. Eur. J. Org. Chem. 2001; 2:237-243.
Newman DJ, Cragg GM. Natural products from marine invertebrates and microbes as modulators of antitumor targets. Curr. Drug Target. 2006; 7:279-304.
Northcote PT, Blunt JW, Munro MHG. Pateamine: a potent cytotoxin from the New Zealand marine sponge, Mycale sp. Tetrahedron Lett. 1991; 32:6511-6414.
Pettit GR. The dolastatins. Forsch. Chem. Org. Naturst. 1997; 70:1-79.
Piel J. Metabolites from symbiotic bacteria. Nat. Prod. Rep. 2004; 21:519-538.
Pietra F. Biodiversity and Natural Product Diversity. 2002. Pergamon, Amsterdam.
Pindur U, Lemster T. Advances in marine natural products of the indole and annelated indole series: chemical and biological aspects. Curr. Med. Chem. 2001; 8:1681-1698.
Rinehart KL, Shield LS, Cohen-Parsons M. Antiviral substances. In: Marine Biotechnology, vol 1. Pharmaceutical and Bioactive Natural Products, vol. 1. Attaway DH, Zaborsky OR, eds. 1993. Plenum Press, New York. pp. 309-342.
Sarma AS, Daum T, Muller WEG, eds. Secondary Metabolites from Marine Sponges. 1993. Ullstein Mosby, Berlin.
Scheuer PJ, ed. Marine Natural Products. Chemical and Biological Perspectives, vol. I-V. 1978, 1978, 1980, 1981, 1983. Academic Press, New York.
Scheuer PJ. Chemistry of Marine Natural Products. 1973. Academic Press, New York.
Schmitz FJ, Bowden BF, Toth SI. Antitumor and cytotoxic compounds from marine organisms. In: Marine Biotechnology, vol 1. Pharmaceutical and Bioactive Natural Products, vol. 1. Attaway DH, Zaborsky OR, eds. 1993. Plenum Press, New York. pp. 197-308.
Scott JD, Williams RM. Chemistry and biology of the tetrahydroisoquinoline antitumor antibiotics. Chem. Rev. 2002; 102:1669-1730.
Shimuzu Y. Dinoflagellates as sources of bioactive molecules. In: Marine Biotechnology, vol 1. Pharmaceutical and Bioactive Natural Products, vol. 1 Attaway DH, Zaborsky OR, eds. 1993. pp. 391-410.
Skyler D, Heathcock CH. The pyridoacridine family tree: a useful scheme for designing synthesis and predicting undiscovered natural products. J. Nat. Prod. 2002; 65:1573-1581.
Terracciano S, Aquino M, Rodriguez M, Monti MC, Casapullo A, Riccio R, Gomez-Paloma L. Chemistry and biology of anti-inflammatory marine natural products: molecules interfering with cyclooxygenase, NF-kB and other unidentified targets. Curr. Med. Chem. 2006; 13:1947-1969.
See Also
Natural Product Discovery
Natural Products in Microbes
Natural Products in Plants
Natural Products, Common Biological Targets for
Natural Products: an Overview