CHEMICAL BIOLOGY
Symbiosis: Topics in Chemical Biology
Daniel Flachshaar and Jorn Piel, University of Bonn, Bonn, Germany
doi: 10.1002/9780470048672.wecb571
The term ''symbiosis'' was defined by Anton de Bary in his monograph ''Die Erscheinung der Symbios'' as ''the living together of unlike organisms'' (1). His studies were based on the formation of lichens, which are the result of an association between a fungus and an alga or cyanobacterium. The definition was coined in the end of the nineteenth century but is regarded by most symbiosis researchers as largely valid today. Accordingly, any specific association between two or more species can be classified as symbiosis. It should be noted that many scientists use symbiosis in a more restricted way to denote a mutually beneficial relationship. This article will give an overview of various biologic manifestations of symbiosis and discuss selected examples, where primary or secondary metabolites play a crucial role in the association. If the partners in a symbiosis differ in size, the larger member is termed ''host'' and the smaller member is termed ''symbiont'' or ''symbiote.'' The more common term ''symbiont'' will be used here. One general way to distinguish between various forms of symbiosis is to identify the location of the attachment of the symbiont to the host. Symbionts that live on the host surface, including internal surfaces like the digestive tube, participate in ectosymbiosis (Greek: εκτος = outside). If a symbiont is localized within the tissues of the host, the association is termed ''endosymbiosis'' (Greek: ενδον = within). Endosymbionts can be found either in the extracellular space or intracellularly.
The term “symbiosis,” from the Greek: σμβιονν = living together, was defined by Anton de Bary in his monograph “Die Erscheinung der Symbios” as “the living together of unlike organisms” (1). His studies were based on the formation of lichens, which are the result of an association between a fungus and an alga or cyanobacterium. The definition was coined in the end of the nineteenth century but is regarded by most symbiosis researchers as largely valid today. Accordingly, any specific association between two or more species can be classified as symbiosis. It should be noted that many scientists use symbiosis in a more restricted way to denote a mutually beneficial relationship. In the widest possible sense of the de Bary definition, symbiosis also refers to pathogenic relationships, although considerable disagreement exists about the inclusion of this type of interaction. For practical reasons, the chemical biology of pathogens will not be covered here.
This article will give an overview of various biologic manifestations of symbiosis and discuss selected examples, where primary or secondary metabolites play a crucial role in the association.
Symbiosis and Its Variants
If the partners in a symbiosis differ in size, the larger member is termed “host” and the smaller member is termed “symbiont” or “symbiote.” The more common term “symbiont” will be used here. One general way to distinguish between various forms of symbiosis is to identify the location of the attachment of the symbiont to the host. Symbionts that live on the host surface, including internal surfaces like the digestive tube, participate in ectosymbiosis (Greek: εκτος = outside). If a symbiont is localized within the tissues of the host, the association is termed “endosymbiosis” (Greek: ενδον = within). Endosymbionts can be found either in the extracellular space or intracellularly.
In addition to a distinction based on microhabitats, variation in symbiont-host relationships gives rise to different terminology. In a mutualism, both the host and the symbiont profit from their relationship. Mutualism, therefore, is defined as the cooperative interaction between different species. Virtually every higher organism is involved in mutual interactions because mitochondria and chloroplasts of eukaryotic cells are descendants of bacteria (see below). Other examples are plants associated with beneficial mycorrhizal fungi (2) or nitrogen-fixing bacteria (3) living on and in their roots. A mutual relationship can consist of partners that benefit from the association but do not depend exclusively on each other for survival. This mutual relationship is called protocooperation (Greek: πρωτος = first; Latin: cooperatio = cooperation). Such a facultative mutualism is known from the association of fungi and photosyn- thesizing microorganisms in lichens (4). In contrast, an obligate mutualism, where host and symbiont cannot survive after separation, is known as eusymbiosis (Greek: εν = good). Examples are mitochondria, chloroplasts, and other bacterial intracellular symbionts of higher organisms. These so-called primary or obligate symbionts usually are ancient. If specialized supracellular host structures exist that harbor such symbionts, they often are termed bacteriosomes. In insects, evidence exists for obligate symbioses that last 30-270 million years, and the symbionts usually coevolve with their hosts (5-7). During this long time of host dependency, the genomes of primary symbionts can undergo massive restructuring, which results in extremely small genome sizes. In addition to primary symbionts, many hosts harbor secondary or facultative symbionts. They participate in an intermediate form of symbiosis, where the symbiont is not strictly necessary for the survival of their host and is not exclusively localized in specific tissues. Because of occasional horizontal transfer, the phylogenetic trees of such organisms often are not congruent with those of their hosts (8). Many secondary symbionts are transmitted maternally and can influence, positively or negatively, various host traits, such as nutrition, survival against natural enemies, or reproduction rates (9, 10).
On the opposite side of mutualism in the interaction spectrum of symbiosis is parasitism (Greek: παρα = beside; σιτος = fatted). Here one species, called the parasite, has physiologic or structural properties that force it to live temporarily or during its whole life on (ectoparasitic) or inside (endoparasitic) another species, the host. Only the parasite benefits from this relationship. Different types of parasitism exist, including biotrophy, in which the host survives during the entire time of the association; perthotrophy, in which the host lives for a short time after contracting a parasite; and necrotrophy, in which the host is killed before the symbiosis starts. Perthotrophs and necrotrophs are known also as parasitoids. This term was coined in 1913 by the entomologist O. M. Reuter to distinguish between true parasites that live at the expense of their hosts without actually causing their death and the parasites that always kill their hosts (11). Among the parasitoids are two categories, which describe the behavior of the parasitoids toward the host. After the initial parasitization, the idiobiont parasitoid prevents any additional development of the host. This parasitization is typical for an immobile host life stage like an egg or a pupa. Most of these parasitoids are endoparasites. Alternately, a koinobiont parasitoid allows the host to continue its development and often does not kill or consume the host until it starts to pupate or become an adult. These parasites, therefore, mostly live in or on an active and mobile host. Some primary parasitoids serve as hosts for yet another parasitoid. The latter is termed “secondary parasitoid” or “hyperparasitoid” and usually kills both the host and the primary parasitoid. Several other commonly used terms exist for different kinds of parasitism. Kleptoparasitism is a relationship in which the parasite steals food that a host has caught or otherwise prepared. An example is the brood parasitism, which is known from many species of cuckoo that use other birds for parenting (12). Social parasites take advantage of interactions between members of a social host species like ants or termites (13). Another special type of parasitism is cheating or exploitation. Here, the parasite uses situations of nonspecific mutualism to its advantage. Some myco-heterotrophic plants, for instance, establish mycorrhiza-like interactions with fungal symbionts and take carbon from the fungi that the fungi obtain from other plants (14). Opportunism is a kind of parasitism where harmless parasites under special circumstances cause diseases or death of their hosts. The fungus Pneumocystis carinii can initiate pneumonia if the host is infected by HIV (15). As in mutualism, obligate and facultative symbionts exist in parasitism. Obligate parasites are not viable without their host, and facultative parasites are free-living organisms that sometimes benefit from their host.
A third form of symbiosis, in addition to mutualism and parasitism, is commensalism (Latin: com = with; mensa = table). In commensal relationships, one symbiotic partner benefits from the association and the other is unaffected. Different variants of such relationships have been described. Inquilinism is a kind of commensalism where the symbiont uses a host for housing, such as birds living in the holes of trees. A more indirect dependency exists if a symbiont uses something its host has created before its death. This commensalism is termed “metabiosis.” A typical metabiont is the hermit crab that uses gastropod shells. In phoresy, the symbiont uses the host for transportation. An example for phoresy is the burdock, a plant with fruits that adhere to fur and are dispersed by the movement of mammals.
Many well-known symbiotic relationships are difficult to categorize into one of the three types, either because distinctions are not clear-cut or because experimental data are hard to obtain. For instance, the question of whether the relationship between humans and some types of their gut flora is commensal or mutual still is unanswered because of the complexity of the multispecies association. The African oxpecker bird is an example where categories are not discrete. The bird picks flies, ticks, and other parasites from the body of a mammal and, in this respect, is a mutualist. However, because blood is its favored food, it keeps wounds open on the body of its host (16). Thus, a mutual symbiont can be a parasite in a separate interaction.
The Chemistry of Symbiotic Associations
Most symbiotic partners exchange low-molecular substances with each other. A chemical interaction can be of crucial importance or even the main determinant for a symbiosis, in which case the term “chemosymbiosis” often is used. A wide spectrum of substances can be involved, ranging from simple inorganic ions to highly complex secondary metabolites. Similarly diverse can be their function, such as nutrition, chemical protection, localization cues, developmental signals, and others. The chemical ecology of symbiotic associations is a relatively unexplored field, and particularly systems that involve microorganisms can be difficult to study because cultivation requirements often are unknown or impossible to reproduce. However, with the advent of culture-independent methods and innovative cultivation techniques, this situation recently has begun to change.
The following sections will give some representative examples where small molecules play an important role in the symbiotic interaction. Cases involving inorganic substances and primary metabolites will be discussed; secondary metabolism will be the subject of the subsequent section.
Inorganic compounds
A best-studied example for a symbiosis between plants and bacteria is the association between leguminous plants and various members of the a-proteobacterial order Rhizobiales (3). The bacteria colonize root nodules of the plants and differentiate morphologically into bacteroids. These bacteroids fix nitrogen from the atmosphere, convert it to ammonia and amino acids, and supply it to the host plant. The bacteria receive organic acids like malate or succinate as a carbon and energy source from the plant. It has to be noted that this symbiosis is not a strict mutualism. The nitrogen fixation is energetically costly to the rhizobia and reduces the resources that could be allocated to their own growth and reproduction (17, 18). Some soils contain rhizobial strains that fix little or no nitrogen (19). Because the strains can coinfect an individual plant together with nitrogen fixers (17), any of these strains could redirect the resources from nitrogen fixation to its own growth and fitness at the expense of its host and the other lineages. This parasitism is a kind of cheating parasitism. Hence, the legume needs ways to guide the evolution of rhizobia toward greater mutualism (20). One mechanism involves reducing the oxygen supply to nodules that fix less nitrogen (21). The frequency of cheating bacteria, thus, is reduced in the next generation. Because rhizobia are not transmitted via the seeds, efficient means of chemical communication are necessary to guarantee a successful infection of each plant. For this purpose, legumes exude low-molecular chemoattractants from their roots, such as betains (22), flavonoids (23), and aldonic acids (24). The substances elicit the production of specific lipooligosaccharides, termed “Nod factors,” in the bacteria (25). After the invasion of a root hair cell, these factors ultimately induce the formation of the root nodules.
Mycorrhiza, an association between soil fungi and plant roots, are among the most widespread symbioses on earth. More than 80% of all land plants carry (vesicular-) arbuscular mycorrhiza, structures generated from an invasion of Glomeromycota fungi into cortical root cells (2, 26). The fungi extract inorganic nutrients, such as phosphate, from the soil and deliver it to the plant. They benefit from this interaction by receiving sugars derived from photosynthesis. Because arbuscular mycorrhizas are at least as old as land plants (27), it has been hypothesized that this interaction helped primitive plants to invade terrestrial habitat before functional roots had evolved. Similar to rhizobial symbiosis, the establishment of mycorrhiza requires the exchange of chemical signals for attraction and differentiation. In Lotus japonicus, terpenes of the strigolactone group have been identified as factors present in root exudates that trigger branching in approaching fungal hyphae (28). A diffusible factor of an as-yet unknown nature also has been demonstrated to be released by fungi and to trigger morphological differentiation in roots (29). Besides arbuscular mycorrhiza, several less frequent mycorrhiza types exist, including the obligate symbiosis between orchids and various fungi (30).
Around deep-sea volcanic vents lives the giant tubeworm Riftiapachyptila in an intimate symbiosis with a sulfur-oxidizing chemoautotrophic bacterium. The two organisms meet extreme living conditions that consist of high and rapidly changing temperatures from 4 °C to 450 °C, an elevated pressure of up to 300 atm, toxic water containing metals and sulfides, and the absence of light (31). The host measures 1-2 meters and completely lacks a digestive tract. Its only tissue with direct contact to the surrounding water is the branchial plume with a highly vascularized surface. It allows an efficient exchange of metabolites and waste products between the worm and the sea water. The major interior tissue of the worm is the trophosome that is localized in a large sac formed by the body wall (32). The cells of the trophosome, termed “bacteriocytes,” are colonized densely by the bacterial endosymbiont (33, 34). Its amount is estimated to reach between 15% and 35% of the whole trophosome (35). The bacteria are supplied with various nutrients by the blood circulation of the worm, which is driven by a heart-like pump. The blood contains an unusual multihemoglobin system, which can transport both sulfide and oxygen (36, 37). The mutual contributions of the bacteria are manifold. One function is to oxidize the transported sulfide into sulfite to produce metabolic energy for both partners (33, 38, 40). Furthermore, the bacteria are indispensable in several assimilation processes by providing various enzymes that the worms lack. An example is the CO2 adsorbed by the worm, which is transported to the bacteria by the blood cycle, either directly (41) or via synthesized malate (42). The bacteria, in turn, employ enzymes of the Calvin-Benson cycle to convert the CO2 to small organic compounds (38, 42, 43). These metabolites then are used by the host for its own metabolism and production of adenosine triphosphate (ATP). The assimilation of ammonia and nitrate also depends on the endosymbiont. Although the glutamine synthetase and glutamate dehydrogenase required for ammonia assimilation are present in both partners (44), the nitrate-reducing enzymes are provided exclusively by the bacteria (43-45). The symbiont also provides the necessary enzymes for a de novo synthesis of pyrimidine but lacks the enzymes of the salvage pathways that allow pyrimidine synthesis from nucleic acid degradation products (45, 46). The host, however, only possesses enzymes that catalyze the final steps of the de novo pathway but provides all enzymes of the salvage pathway (46). The de novo synthesis of pyrimidine, thus, is regulated by the bacterial enzymes. Arginine is metabolized in a similar manner. The worm does not possess arginine decarboxylase or ornithine decarboxylase activities (47), and it is likely that it depends on the bacteria to realize this pathway.
In another gutless marine worm, the oligochaete Olavius algarvensis, three partners participate in an unusual mutualism. The worm harbors two different primary symbionts in immediate proximity below its cuticle between extensions of epidermal cells. By performing comparative 16S rRNA sequencing and fluorescence in situ hybridization, two dominant clone groups of the γ- and δ-subclasses of the proteobacteria were found (48). The larger γ-proteobacterial symbiont possesses a high sequence similarity with endosymbionts from other gutless oligochaetes, like Olavius loisae and Inanidrilus leukodermatus (49, 50), and is thioautotrophic, i.e., possesses a sulfur-oxidizing, CO2-fixing metabolism (48). The smaller δ-proteobacterial symbiont is genetically very similar to free-living sulfate-reducing bacteria, and the sulfate-reducing function also could be demonstrated in O. algarvensis (48). The coexistence of these bacteria indicates that they are engaged in a syntrophic sulfur cycle in which oxidized and reduced sulfur compounds are exchanged back and forth (48). The oligochaete benefits from this arrangement by obtaining de novo-synthesized organic compounds from the symbionts, which in turn assimilate its anaerobic end products. The constant internal source of reduced sulfur compounds allows the worm to colonize new habitats without a requirement of high sulfide concentrations.
Numerous additional examples exist of intimate symbioses that involve the exchange of inorganic compounds, including cyanobacteria (providing fixed nitrogen to lichens, the water fern Azolla, Gunnera spp. plants, cycads and some mosses, liver worts, and horn worts) (51), clams of the genera Solemya and Codakia (providing sulfide to bacteria) (52, 53), the sponge Haliclona cymiformis (providing ammonia and nitrate to a rhodophyte alga) (54), corals (inorganic carbon to photosynthetic dinoflagellates for carbon fixation) (55, 56), rumen bacteria (providing hydrogen and CO2 to methanogenic archaea) (57), and “Chlorochromatium aggregatum," an association of a sulfate-reducing and a phototrophic sulfide-oxidizing bacterium (58).
Primary organic metabolites
Among the best-investigated chemosymbioses that involve primary metabolites are mutualisms of eukaryotic cells with their mitochondria and chloroplasts. Mitochondria synthesize ATP, as well as heme and steroids, by oxidative phosphorylation, whereas chloroplasts provide plant and algal cells with glucose generated via the Calvin cycle by using the energy of light. According to the endosymbiotic theory, these organelles developed from independent prokaryotic organisms that were absorbed by their host cells (59). Several lines of evidence, such as the phylogeny of plastid DNA sequences and the structure of the photosynthetic apparatus, suggest that chloroplasts developed from cyanobacteria. The origin of the mitochondria has been traced back to an α-proteobacterium. The two associations are typical examples of eusymbioses: The organelles are not viable without the surrounding cell because they have lost much of their original genetic material, some of which now is contained in the cell nucleus; eukaryotic cells, however, strictly depend on the compounds produced by the organelles.
Several animals also have established symbioses with a photosynthetic partner. In contrast to chloroplasts, the photobionts have retained characteristics of their free-living relatives to a much greater extent. Examples for such associations with animals as hosts are the symbioses of corals and other cnidarians as well as some sponges, molluscs, flatworms and protists with zooxanthellae (dinoflagellates; they, in turn, contain chloroplasts derived from cyanobacteria) or zoochlorellae (green algae), ascidians, sponges, and echiuroid worms with cyanobacteria (60).
Many insects harbor one or more highly specific intracellular symbionts (8). An example is the tsetse fly that feeds on vertebrate blood and is a vector of African trypanosomes. These protozoans cause sleeping sickness in animals and humans in Sub-Saharan Africa (61). The fly carries the obligate primary symbiont Wigglesworthia glossinidia and the secondary symbiont Sodalis glossinidius. The primary symbiont resides in the bacteriosome that is located in the gut tissue and consists of differentiated host epithelial cells (61). The secondary symbiont can be detected intra- and extracellularly in the host midgut and the hemolymph (62). Elimination of W. glossidina in the tsetse flies resulted in retarded host growth and decreased egg production and fecundity. Feeding of these aposymbiotic flies with B-complex vitamins partially restored the reproduction rates (63). This finding suggested that the host, feeding on a vitamin-poor diet, obtains these metabolites from its primary symbiont. Phylogenetic characterization of W. glossinidia living in different tsetse flies has shown that their evolution is congruent with that of their hosts (64). According to these investigations, the primary infection event was estimated to have taken place 50-100 million years ago (65). During the long time of intracellular life, a massive reduction of the symbiont genome occurred, which resulted in a genome size of only 698 kilobases (65). Because of the loss of numerous genes, W. glossinidia is expected to be not viable without the host insect. A study on the secondary symbiont S. glossinidius also indicated the existence of mutualism. After selective elimination, the resulting flies showed a significantly reduced longevity, although the underlying mechanism is not well understood (10). In contrast to W. glossinidia, S. glossinidius diverged from its free-living ancestor much more recently. Its genome is 4.2 Mb (66), much larger than that of the primary symbiont, and shows only beginning signs of erosion (67). It is possible to cultivate the bacterium in the absence of host cells (68).
Aphids feed on a plant sap diet rich in carbohydrates but poor in vitamins and amino acids. They possess a bacteriosome that contains up to 80 large cells filled with bacteria of the genus Buchnera (69). They are transmitted maternally through the host generations, and neither of the mutual partners can reproduce independently (70). The association of the aphids with an enterobacterial-like ancestor of the symbiont was established 200-250 million years ago (71). The genome size of Buchnera aphidicola strains measures only 420-640 kb, which is less than one seventh of that of the close relative E. coli (72). During evolution, the symbiont has lost numerous genes, including genes that encode the biosynthesis of cell-surface components, regulatory genes, and genes for its own defense (73). Notably, B. aphidicola lacks several pathways for amino acids that the host can synthesize, although it retains genes for the production of all essential amino acids (7, 73). Evidence exists that the aphid exports synthesized glutamate to the symbiont as a nitrogen source, which allows it to produce its essential amino acids (73, 74). The strain B. aphidicola BCc of the aphid Cinara cedri has an exceptionally small genome of only 422 kb that even lacks essential trypophane and riboflavin genes (75). It has been hypothesized to gradually lose its mutual properties and to be superseded by a secondary symbiont, “Candidatus Serratia symbiotica” (76). The smallest known genome among insect symbionts spans only 160 kb. It belongs to Carsonella ruddii, a symbiont of psyllids that apparently already has reached organelle status. Nevertheless, several essential amino acid pathways have been identified by genomic analysis, which indicates the importance of the metabolic products for the interaction (77).
Termites, which feed on a nutrient-poor diet of cellulose, harbor a complex community of protozoans, bacteria, and archaea in their gut (78). The protozoan converts cellulose to acetate that, in turn, is absorbed by the termite. Another metabolic by-product of some protists is hydrogen and CO2 (79). These by-products are believed to be used by methanogenic archaea and bacteria present in the gut consortium. Another remarkable association in termites is the motility symbiosis between an oxymonad protist and spirochaete bacteria (80). The bacteria are embedded in the host membrane and wave synchronously to assist in host locomotion. Symbioses based on the degradation of organic material are generally common in animals with a diet rich in cellulose, such as ruminants and wood-boring insects. Besides protists, bacterial and fungal cellulose degraders have been identified in their guts. Several insects are known to actively ingest fungi to use their cellulases. This behavior might represent the first stage in the establishment of a symbiosis with such fungi stably residing in the gut (81).
An economically important chemical protocooperation of two different bacteria exists in yogurt preparation. A widely used starter for the production of fermented dairy products is the thermophilic lactid acid bacterium Streptococcus thermophilus. For the manufacturing of yogurt, it is generally used in association with other microorganisms, in particular with Lactobacillus delbrueckii ssp. bulgaricus (L. bulgaricus). To enable the intense and rapid acidification of milk, the fast growing capacity of these bacteria is crucial. The association causes a positive effect on the growth of both species. The growth of L. bulgaricus is stimulated by pyruvic acid, formic acid, and CO2 that are produced by S. thermophilus (82). L. bulgaricus is a producer of amino acids and peptides that stimulate the growth of S. thermophilus (83). The symbiosis additionally results in enhanced aroma formation compared with the pure cultures.
The exchange of primary metabolites also is known from several nonmicrobial symbioses. Carbohydrates play an important role in many of these symbioses. Besides the ubiquitous example of flowering plants offering nectar, several plants are known that produce extrafloral nectar to attract beneficial insects, in particular ants (84). Ants colonizing such myrmecophytes or ant plants protect their nutrient source by efficiently removing herbivores and even pathogenic fungi. Other nutrient sources provided by ant plants are food bodies rich in proteins or lipids. In a well-known functionally related association, ants obtain carbohydrates from honeydew secreted by aphids that they regularly milk and defend against predators (85).
Secondary metabolites
Sessile marine animals, such as sponges, ascidians, and bryozoans, are the source of numerous compounds with diverse pharmacological activities (86, 87). It has long been suspected that many of these natural products are produced, in fact, by symbiotic bacteria because the chemical structures often resemble those of compounds isolated from prokaryonts (87-89). The existence of a bacterial source for an invertebrate-derived natural product should have important biomedical implications, as this ultimately could allow one to create sustainable fermentation systems for drug development and production. Sponges are the most important sources of marine drug candidates. These animals can contain remarkable numbers of diverse microorganisms from various taxa representing up to 40-60% of the total biomass (90, 91). The microflora in most bacteriosponges is highly distinct but shows little variation among sponge species or geographic location, which indicates a long period of coexistence (92). At least a part of the microbial community is transferred vertically via the sponge larvae (93). As with many other symbiotic systems, cultivation attempts have failed in most cases. Therefore, for most sponge-derived natural products, the actual producer still is unknown. Among the few successfully studied examples are the protein biosynthesis inhibitors onnamides (Fig. 1a) and theopederins (Fig. 1b), potent cytotoxic polyketides isolated from Theonella swinhoei (94). A bacterial source was identified by metagenomic techniques, i.e., methods employing the DNA of entire organismic associations without prior separation of species (95, 96). The screening of a large metagenomic library prepared from total T. swinhoei DNA led, in this case, to the isolation of the biosynthetic genes, which where attributed to a bacterial producer (95). Another method employed to identify biosynthetic sources of natural products is mechanical cell separation and subsequent extraction of each cell type. In this way, the likely bacterial sources of several compounds were identified, including the polyketide swinholide A (a unicellular bacterium) (97), the nonribosomal peptide theopalauamide A (a filamentous S-proteobacterium) (97), chlorinated dipeptides (Fig. 2a) (a cyanobacterium) (98, 99) and brominated diphenylethers (Fig. 2b) (a cyanobacterium) (100). One patent exists on the successful cultivation of an actinomycete symbiont from the sponge Acanthostrongylophora sp. producing antiinfective manzamine alkaloids (Fig. 3) (101).

Figure 1. Onnamide A (a), Theopederin A (b) from a symbiont of the sponge Theonella swinhoei.
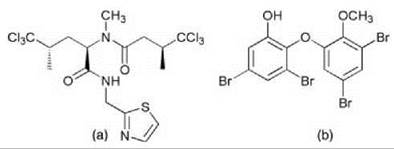
Figure 2. Demethylisodysidenin (a), Brominated diphenylether (b) from cyanobacterial symbionts of sponges.
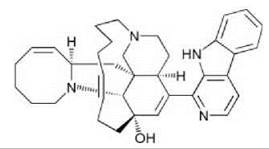
Figure 3. Antiinfective Manzamine A from an actinomycete symbiont of the sponge Acanthostronglyophora sp.
Another pharmacologically relevant group of marine animals is the phylum Ectoprocta, also known as Bryozoa or Polyzoa. They are colonial filter feeders where each member is enclosed in a separate unit called a zooecium. The bryozoan Bugula neritina contains complex polyketides of the bryostatin series (Fig. 4), protein kinase C activators with potent anticancer activity (102, 103). Bryostatin 1 is one of the most promising drug candidates from marine invertebrates and has reached phase II clinical trials for combination therapy (104). The concentration of bryostatins is particularly high in larvae of B. neritina, where it is used as a defense against predators (105). However, in adults the concentration is very low or even undetectable when the animals lack larval brood chambers (105). 16S rRNA analysis and in situ hybridization revealed the presence of a γ-proteobacterium, “Candidatus Endobugula sertula,” in the larvae (105-108). This bacterium was shown to harbor polyketide synthase genes that are good candidates for bryostatin biosynthesis because their presence is correlated strictly with high bryostatin levels (108).

Figure 4. Bryostatin 1, a polyketide isolated from the bryozoan Bugula neritina.
Ascidians are marine filter feeders with a rich natural products chemistry that live commonly associated with symbiotic bacteria (88, 89, 109). A well-studied symbiosis consists of photosynthetic Prochloron spp. cyanobacteria that occur in ascidians of the family Didemnidae (110). Prochloron spp. also can be found in bacterial mat structures of stromatoliths (111) but so far have not been detected outside of such structured environments. From didemnid ascidians, numerous cytotoxic cyclic peptides of the patellamide group (Fig. 5) were isolated (109, 112, 113). Mechanical separation of the Prochloron sp. symbiont from its host Lissoclinum patella and subsequent genome sequencing revealed a set of biosynthetic genes that after transfer into E. coli enabled this bacterium to produce two different patellamides (114). The genes also were identified in an independent study by screening a library of Prochloron sp. DNA constructed in E. coli for the presence of clones that synthesize patellamides (115). The patellamide gene cluster also has been engineered to produce a drug-like, non-natural cyclic peptide (116). So far, the ecological function of the peptides in the natural environment remains unknown.

Figure 5. Patellamide D, a cyclic peptide from a symbiont of didemnid ascidians.
The gorgonian soft coral Pseudopterogorgia elisabethae harbors dinoflagellates of the genus Symbiodinium. The symbiotic function of the dinoflagellate is to contribute to the nutrition of its host by sharing photoassimilates (55, 56). Extracts of the corals contain the pseudopterosins (Fig. 6), a family of diterpene glycosides with antiinflammatory and analgesic properties (117). The substances modulate degranulation and release mechanisms in immune cells (118). They have been included as antiirritants in a widely marketed cosmetic. The amounts and the specific composition of the pseudopterosins vary depending on the location of P. elisabethae (119). Their total amount can be as high as 20% of the dry mass. By applying radioactively labelled geranylgeranyldiphosphate (GGPP) to purified symbiont preparations, it was demonstrated that the biosynthesis of the pseudopterosins occurs in the dinoflagellate (120). Elisabethatriene synthase, the enzyme catalyzing the conversion of GGPP into the first cyclized intermediate elisabethatriene, has been purified and might be used for biotechnological pseudopterosin production (121). Pseudopterosins reduce the inducible oxidative burst of the symbiont dramatically during stress (122). Reactive oxygen species of dinoflagellate origin have been suspected to kill the symbiont and to damage the host cells, which leads to coral bleaching (123). The terpenes, therefore, may have an important function in sustaining a healthy symbiosis.

Figure 6. Pseudopterosine A, from a dinoflagellate symbiont of corals.
Several remarkable natural product symbioses have been reported from terrestrial habitats. Ants of the genera Acromyrmex and Atta are specialized in growing large gardens of fungi (Agaricales, mostly Leucocoprineae) within their colonies and providing them with optimal growth conditions (124). These fungi represent the main food source. This interaction was estimated to have originated more than 45-65 million years ago (125). The cultivated fungi often are infected by fungal pathogens, Escovopsis spp., which results in the destruction of entire colonies (126). The ants use an efficient strategy to prevent such infections with the help of symbiotic actinomycetes of the family Pseudonocardiaceae (127). These bacteria live in specific regions of the ant cuticle and produce as-yet uncharacterized antibiotics that inhibit the pathogenic fungus and promote the growth of the garden fungus (127). The symbiont seems to be exclusively vertically transmitted from the parent to the offspring colony. Transmission is achieved by the queens, which carry the actinomycetes on their cuticle during the nuptial flight.
Another symbiosis that involves antibiotics occurs in bee wolves of the genus Philanthus. The adult insects harbor a Streptomyces sp. bacterium in their antennal glands (128, 129). The symbiont is applied to the brood cell, where it is taken up by the larva. At the time of pupation, the bacteria are found in large numbers on the cocoon. It has been shown that the presence of symbiont confers a greater resistance against fungal infection because of the production of an antibiotic (128).
Paederus and Paederidus spp. rove beetles participate in an unusual facultative symbiosis. Within each beetle species, most, but not all, females contain the complex polyketide pederin (Fig. 7) in their hemolymph (130). The substance is highly toxic and is used as a defense against predators (9). Structurally, pederin is highly similar to a range of compounds isolated from marine sponges, such as the onnamides and the theopederins mentioned above (94). Isolation of the genes that encode pederin biosynthesis revealed that the substance is produced by a bacterial symbiont closely related to Pseudomonas aeruginosa (131). The symbiont is present only in pederin-producing females (132) and also can be detected in the eggs (133). P. aeruginosa does not produce pederin, and partial genome sequencing suggests that the symbiont or its free-living ancestor might have acquired the pederin genes by horizontal gene transfer (134). This mechanism also could account for the occurrence of similar compounds in beetles and sponges. Because P. aeruginosa is a pathogen of insects and other organisms (including humans), it is likely that the symbiosis developed after a beetle was infected by a pathogenic ancestor of the symbiont. The protective effect of pederin then might have driven a mutual adaptation of both partners toward symbiosis.

Figure 7. Pederin from a bacterial symbiont of Paederus and Paedericus spp. rove beetles.
Nematodes of the families Steinernematidae and Heterorhabditidae employ Photorhabdus or Xenorhabdus bacteria of the family Enterobacteriaceae in a complicated life cycle (135, 136). In general, both types of bacteria exhibit similar behavior. The nematodes harbor the symbionts during their infective juvenile stage (136-138). They invade various insect larvae via the digestive tract, the respiratory spiracles, or the cuticle and subsequently penetrate into the hemocoel (137, 138), into which they inject the bacteria. Together with the highly virulent bacteria, the host kills the insect within 48 hours (137). Inside the insect carcass, the bacteria grow to stationary phase and create optimal conditions for growth and sexual reproduction of the nematodes. These conditions are reached when the symbiont dominates the microbial flora. The role of the bacteria is to provide essential nutrients for nematode proliferation, to secrete insecticidal proteins, and to inhibit putrefaction of the insect carcass by preventing growth of other microorganisms (137-139). At the end of the development, bacteria and nematodes reassociate, and the nematodes develop into nonfeeding infective juvenile stages that emerge from the carcass and search for new insect hosts. Bacteria of both genera can be cultivated easily. They have been shown to synthesize various broad-spectrum antibiotics, lipases, phospholipases, and proteases that are believed to become secreted into the insect hemolymph when the bacteria enter the stationary phase. From Photorhabdus spp. antibacterial, antifungal, and nematicidal hydroxystilbenes (Fig. 8a) (140), anthraquinone pigments (Fig. 8b) (141) and the antibacterial siderophore photobactin (Fig. 8c) (142) have been isolated. Xenorhabdus spp. synthesize a series of antibacterial pyrrothins known as xenorhabdins (Fig. 9a) (143), the antibacterial xenocoumacins (Fig. 9b) (144), cytotoxic phenethylamides (Fig. 9c) (145), and antibacterial and antifungal 3-substituted indole derivatives (Fig. 9d) (146). In addition to their natural product profiles, Photorhabdus spp. and Xenorhabdus spp. exhibit other characteristic differences. Photorhabdus spp. are associated only with nematodes of the family Heterorhabditidae and have the ability to emit light under stationary phase culture conditions and in the infected host insect (147). Xenorhabdus spp. are associated with nematodes of the family Steinernematidae (148). Thus, this type of symbiosis might have arisen independently by convergent evolution (135).

Figure 8. Examples of natural products from Photorhabdus spp.

Figure 9. Examples of natural products from Xenorhabdus spp.
The Rhizopus-Burkholderia association is another example of a mutual interaction with pathogenic properties against a third species. The antimitotic polyketide rhizoxin (Fig. 10) isolated from the pathogenic fungus Rhizopus microsporus is a crucial factor in the development of rice seedling blight (149). Rhizoxin induces an abnormal swelling of rice seedling roots that is a typical symptom of this disease (150). The compound also arrests mitosis in many other eukaryotic cells, including various tumor cells (151). It, therefore, has reached clinical trials as a potential antitumor drug (152). Rhizoxin was believed for a long time to be produced by the fungus. However, during attempts to clone the biosynthetic genes, an intracellular Burkholderia sp. bacterium was detected in all studied rhizoxin-positive Rhizopus strains. The symbiont was grown in pure culture and shown to produce rhizoxin and several derivatives in significantly greater amounts as compared with the fungus (153). Transfer of the strain to aposymbiotic, nonproducing fungi resulted in the establishment of a rhizoxin-positive chemotype. The availability of an efficient bacterial source also was exploited to generate a chemically modified rhizoxin with 1000-10000 times greater activity than the parent compound (154).
A great diversity of endophytic fungi colonizes the interior of most plants, red algae, and brown algae (155). The fungi often can be isolated and subsequently studied by placing leaf fragments on solid media. In addition to exchanged nutrients, several secondary metabolites have been shown to play an important role in this interaction. Fungus-derived alkaloids, such as peramine (Fig. 11a), lolines (Fig. 11b), ergot alkaloids (Fig. 11c), and lolitrems (Fig. 11d), defend grasses against herbivores (156, 157). Ergot alkaloids also are produced in Convolvulaceae in a symbiosis with a clavicipitaceous fungus that is transmitted via the seeds (158). Numerous additional compounds have been identified from isolated endophytes, which documents their remarkable biosynthetic potential (159). Their exact ecological functions, however, in most cases are poorly understood.

Figure 10. Rhizoxin, a polyketide from bacterial endosymbionts of the pathogenic fungus Rhizopus microsporus.

Figure 11. Peramine (a), Norloline (b), Elymoclavine (c), Lolitreme B (d), alkaloids from endophytic fungi.
References
1. Bary A. Die Erscheinung der Symbios. 1879. Privately printed, Strasborg.
2. Paszkowski U. Mutualism and parasitism: the yin and yang of plant symbioses. Curr. Opin. Plant Biol. 2006; 9:364-370.
3. Lodwig E, Poole P. Metabolism of Rhizobium bacteroids. Crit. Rev. Plant Sci. 2003; 22:37-78.
4. DePriest PT. Early molecular investigations of lichen-forming symbionts: 1986-2001. Annu. Rev. Microbiol. 2004; 58:273-301.
5. Munson MA, Baumann P, Clark MA, Baumann L, Moran NA, Voegtlin DJ, Campbell BC. Evidence for the establishment of aphid-eubacterium endosymbiosis in an ancestor of for aphid families. J. Bacteriol. 1991; 173:6321-6324.
6. Baumann P. Biology of bacteriocyte-associated endosymbionts of plant sap-sucking insects. Annu. Rev. Microbiol. 2005; 59:155-189.
7. Moran NA, Plague GR, Sandstrom JP, Wilcox JL. A genomic perspective on nutrient provisioning by bacterial symbionts of insects. Proc. Natl. Acad. Sci. U.S.A. 2003; 100:14543-14548.
8. Dale C, Moran NA. Molecular interactions between bacterial symbionts and their hosts: Review. Cell 2006; 126:453-465.
9. Kellner RLL, Dettner K. Differential efficacy of toxic pederin in deterring potential arthropod predators of Paederus (Coleoptera: Staphylinidae) offspring. Oecologia 1996; 107:293-300.
10. Dale C, Welburn SC. The endosymbionts of tsetse flies: manipulating host-parasite interactions. Int. J. Parasitol. 2001;31:628- 631.
11. Reuter OM. Lebensgewohnheiten und Instinkte der Insekten. 1913. Friedlander, Berlin.
12. Winfree R. Cuckoos, cowbirds and the persistence of brood parasitism. Trends Ecol. Evol. 1999; 14:338-343.
13. Lenoir A, D’Ettorre P, Errard C, Hefetz A. Chemical ecology and social parasitism in ants. Annu. Rev. Entomol. 2001; 46:573-599.
14. Leake JR. Myco-heterotroph/epiparasitic plant interactions with ectomycorrhizal and arbuscular mycorrhizal fungi. Curr. Opin. Plant Biol. 2004; 7:422-428.
15. Buzoni-Gatel D, Kasper LH. Opportunism and AIDS: walking the tightrope. Parasitol. Today 1999; 15:45-46.
16. Weeks P. Interactions between red-billed oxpeckers, Buphagus erythrorhynchus, and domestic cattle, Bos taurus, in Zimbabwe. Anim. Behav. 1999; 58:1253-1259.
17. Denison RF. Legume sanctions and the evolution of symbiotic cooperation by rhizobia. Am. Nat. 2000; 156:567-576.
18. West SA, Kiers ET, Simms EL, Denison RF. Sanctions and mutualism stability: when should less beneficial mutualists be tolerated? J. Evol. Biol. 2002; 269:685-694.
19. Burdon JJ, Gibson AH, Searle SD, Woods MJ, Brockwell J. Variation in the effectiveness of symbiotic associations between native rhizobia and temperate Australian Acacia: within-species interactions. J. Appl. Ecol. 1999; 36:398-408.
20. Ferriere R, Bronstein JL, Rinaldi S, Law R, Gauduchon M. Cheating and the evolutionary stability of mutualism. Proc. Biol. Sci. 2002; 269:773-780.
21. Kiers ET, Rousseau RA, West SA, Denison RF. Host sanctions and the legume-rhizobium mutualism. Nature 2003; 425:78-81.
22. Philips DA, Joseph CM, Maxwell CA. Trigonelline and Stachydrine released from Alfalfa seeds activate NodD2 protein in Rhizobium meliloti. Plant Physiol. 1992; 99:1526-1531.
23. Djordjevic MA, Weinman JJ. Factors determining host recognition in the Clover-Rhizobium symbiosis. Aust. J. Plant Physiol. 1991; 18:543-557.
24. Gagnon H, Ibrahim RK. Aldonic acids: a novel family of nod gene inducers of Mesorhizobium loti, Rhizobium lupini, and Sinorhizobium meliloti. Mol. Plant-Microbe Interact. 1998; 11:988-998.
25. D’Haeze W, Holsters M. Nod factor structures, responses, and perception during initiation of nodule development. Glycobiology 2002; 12:79R-105R.
26. Barea JM, Pozo MJ, Azcon R, Azcon-Aguilar C. Microbial co-operation in the rhizosphere. J. Exp. Bot. 2005; 56:1761-1778.
27. Simon L, Bousquet J, Levesque RC, Lalonde M. Origin and diversification of endomycorrhizal fungi and coincidence with vascular land plants. Nature 1993; 363:67-69.
28. Akiyama K, Matsuzaki K, Hayashi H. Plant sesquiterpenes induce hyphal branching in arbuscular mycorrhizal fungi. Nature 2005; 435:824-827.
29. Kosuta S, Chabaud M, Lougnon G, Gough C, Denarie J, Barker DG, Becard G. A diffusible factor from arbuscular mycorrhizal fungi induces symbiosis-specific MtENOD11 expression in roots of Medicago truncatula. Plant Physiol. 2003; 131:952-962.
30. Otero JT, Ackerman JD, Bayman P. Differences in mycorrhizal preferences between two tropical orchids. Mol. Ecol. 2004; 13:2393-2404.
31. Tunnicliffe V. The biology of hydrothermal vents: ecology and evolution. Oceanogr. Mar. Biol. Annu. Rev. 1991; 29:319-407.
32. Gaill F. Aspects of life development at deep sea hydrothermal vents. FASEB J. 1993; 7:558-565.
33. Nelson DC, Fisher CR. Chemoautotrophic and methanotrophic endosymbiotic bacteria at deep-sea vents and seeps. In Microbiology of Deep Sea Hydrothermal Vents. Karl DM, ed. 1995. CRC Press Inc., Boca Raton, FL. pp. 125-167.
34. Hand SC. Trophosome ultrastructure and the characterization of isolated bacteriocytes from invertebrate-sulfur bacteria symbioses. Biol. Bull. 1987; 173:260-276.
35. Powel MA, Somero GN. Adaptations to sulfide by hydrothermal vent animals: sites and mechanisms of detoxification and metabolism. Biol. Bull. 1986; 171:274-290.
36. Arp AJ, Doyle ML, DiCera E, Gill SJ. Oxygenation properties of the two co-occuring hemoglobins of the tube worm Riftia pachyptila. Respir. Physiol. 1990; 80:323-334.
37. Goffredi SK, Childress JJ, Desaulniers NT, Lallier FJ. Sulfide acquisition by the vent worm Riftia pachyptila appears to be via uptake of HS-, rather than H2S. J. Exp. Biol. 1997; 200:2609-2616.
38. Felbeck H, Childress JJ, Somero GN. Calvin-Benson cycle and sulfide oxidation enzymes in animals from sulphide-rich habitats. Nature 1981; 293:291-293.
39. Rau GH. Hydrothermal vent clam and tubeworm 13C/12C. Further evidence of nonphotosynthetic food sources. Science 1981; 213:338-340.
40. Smith DW, Strohl WR. Sulfur-oxidizing bacteria. Variations in Autotrophic Life. Shively JM, Barton LL, eds. 1991. Academic Press, San Diego, CA. pp. 121-146.
41. Childress JJ, Lee RW, Sanders NK, Felbeck H, Oros DR, Toulmond A, Desbruyeres D, Kennicutt MC, Brooks J. Inorganic carbon uptake in hydrothermal vent tubeworms facilitated by high environmental pCO2. Nature 1993; 362:147-149.
42. Felbeck H, Jarchow J. Carbon release from purified chemoau- totrophic bacterial symbionts of the hydrothermal vent tubeworm Riftia pachyptila. Physiol. Zool. 1998; 71:294-302.
43. Hentschel U, Felbeck H. Nitrate respiration in the hydrothermal vent tubeworm Riftia pachyptila. Nature 1993; 366:338-340.
44. Lee RW, Robinson JJ, Cavanaugh CM. Pathways of inorganic nitrogen assimilation in chemoautotrophic bacteria-marine invertebrate symbioses: expression of host and symbiont glutamine synthetase. J. Exp. Biol. 1999; 202:289-300.
45. Minic Z, Simon V, Penverne B, Gaill F, Herve G. Contribution of the bacterial endosymbiont to the biosynthesis of pyrimidine nucleotides in the deep-sea tube worm Riftia pachyptila. J. Biol. Chem. 2001; 276:23777-23784.
46. Minic Z, Pastra-Landis S, Gaill F, Herve G. Catabolism of pyrimidine nucleotides in the deep-sea tube worm Riftia pachyptila. J. Biol. Chem. 2002; 277:127-134.
47. Minic Z, Herve G. Arginine metabolism in the deep sea tube worm Riftia pachyptila and its bacterial endosymbiont. J. Biol. Chem. 2003; 278:40527-40533.
48. Dubilier N, Mulders C, Ferdelman T, de Beer D, Pernthaler A, Klein M, Wagner M, Erseus C, Thiermann F, Krieger J, Giere O, Amann R. Endosymbiotic sulphate-reducing and sulphide-oxidizing bacteria in an oligochaete worm. Nature 2001; 411:298-302.
49. Dubilier N, Amann R, Erseus C, Muyzer G, Park SY, Giere O, Cavanaugh CM. Phylogenetic diversity of bacterial endosym- bionts in the gutless marine oligochete Olavius loisae (Annelida). Mar. Ecol. Prog. Ser. 1999; 178:271-280.
50. Dubilier N, Giere O, Distel DL, Cavanaugh CM. Characterization of chemoautotrophic bacterial symbionts in a gutless marine worm (Oligochaeta, Annelida) by phylogenetic 16S rRNA sequence analysis and in situ hybridization. Appl. Environ. Microbiol. 1995; 61:2346-2350.
51. Stewart WD, Rowell P, Rai AN. Cyanobacteria-eukaryotic plant symbioses. Ann. Microbiol. 1983; 134B:205-228.
52. Stewart FJ, Cavanaugh CM. Bacterial endosymbioses in Solemya (Mollusca: Bivalvia)—model systems for studies of symbiont- host adaptation. Antonie Van Leeuwenhoek. 2006; 90:343-360.
53. Distel DL, Lane DJ, Olsen GJ, Giovannoni SJ, Pace B, Pace NR, Stahl DA, Felbeck H. Sulfur-oxidizing bacterial endosymbionts: analysis of phylogeny and specificity by 16S rRNA sequences. J. Bacteriol. 1988; 170:2506-2510.
54. Davy SK, Trautman DA, Borowitzka MA, Hinde R. Ammonium excretion by a symbiotic sponge supplies the nitrogen requirements of its rhodophyte partner. J. Exp. Biol. 2002; 205:3505- 3511.
55. Muscatine L, Porter JW. Reef corals: mutualistic symbiosis adapted to nutrient poor environments. Bioscience 1977; 27:454- 460.
56. Schmitz K, Kremer BP. Carbon fixation and analysis of assimilates in a coral-dinoflagellate symbiosis. Mar. Biol. 1977; 42:305-313.
57. Vogels GD, Hoppe WF, Stumm CK. Association of methanogenic bacteria with rumen ciliates. Appl. Environ. Microbiol. 1980; 40:608-612.
58. Overmann J, van Gemerden H. Microbial interactions involving sulfur bacteria: implications for the ecology and evolution of bacterial communities. FEMS Microbiol. Rev. 2000; 24:591-599.
59. Margulis L. Symbiotic theory of the origin of eukaryotic organelles; criteria for proof. Symp. Soc. Exp. Biol. 1975; 29:21-38.
60. Smith DC, Bernays EA. Why do so few animals form endosymbiotic associations with photosynthetic microbes?. Phil. Trans. R. Soc. Lond. B 1991; 333:225-230.
61. Aksoy S. Tsetse: a haven for microorganisms. Parasitol. Today 2000; 16:114-118.
62. Cheng Q, Aksoy S. Tissue tropism, transmission and expression of foreign genes in vivo in midgut symbionts of tsetse flies. Insect Mol. Biol. 1999; 8:125-132.
63. Nogge G. Significance of symbionts for the maintenance of an optional nutritional state for successful reproduction in hematophagous arthropods. Parasitology 1981; 82:101-104.
64. Chen X, Song L, Aksoy S. Concordant evolution of a symbiont with its host insect species: Molecular phylogeny of genus Glossina and its bacteriome-associated endosymbiont, Wigglesworthia glossinidia. J. Mol. Evol. 1999; 48:49-58.
65. Akman L, Yamashita A, Watanabe H, Oshima K, Shiba T, Hattori M, Aksoy S. Genome sequence of the endocellular obligate symbiont of tsetse flies, Wigglesworthia glossinidia. Nat. Genet. 2002; 32:402-407.
66. Toh H, Weiss BL, Perkin SA, Yamashita A, Oshima K, Hattori M, Aksoy S. Massive genome erosion and functional adaptations provide insights into the symbiotic lifestyle of Sodalis glossini- dius in the tsetse host. Genome Res. 2006; 16:149-156.
67. Akman L, Rio RV, Beard CB, Aksoy S. Genome size determination and coding capacity of Sodalis glossinidius, an enteric symbiont of tsetse flies, as revealed by hybridization to Escherichia coli gene arrays. J. Bacteriol. 2001; 183:4517-4525.
68. Beard CB, O’Neill SL, Mason P, Mandelco L, Woese CR, Tesh RB, Richards FF, Aksoy S. Genetic transformation and phylogeny of bacterial symbionts from tsetse. Insect Mol. Biol. 1993; 1:123-131.
69. Munson MA, Baumann P, Kinsey MG. Buchnera, new genus and Buchnera aphidicola, new species, a taxon consisting of the mycetocyte-associated, primary endosymbionts of aphids. Int. J. Syst. Bacteriol. 1991; 41:566-568.
70. Ishikawa H. Biochemical and molecular aspects of endosymbiosis in insects. Int. Rev. Cytol. 1989; 116:1-45.
71. Moran NA, Munson MA, Baumann P, Ishikawa H. A molecular clock in endosymbiotic bacteria is calibrated using the insect hosts. Proc. R. Soc. Lond. B 1993; 253:167-171.
72. Charles H, Ishikawa H. Physical and genetic map of the genome of Buchnera, the primary endosymbiont of the pea aphid Acyrthosiphon pisum. J. Mol. Evol. 1999; 48:142-150.
73. Shigenobu S, Watanabe H, Hattori M, Sakaki Y, Ishikawa H. Genome sequence of the endocellular bacterial symbiont of aphids Buchnera sp. APS. Nature 2000; 407:81-86.
74. Rahbe Y, Digilio MC, Febvay G, Guillaud J, Fanti P, Pennacchio F. Metabolic and symbiotic interactions in amino acid pools of the pea aphid, Acyrthosiphon pisum, parasitized by the braconid Aphidius ervi. J. Insect Physiol. 2002; 48:507-516.
75. Perez-Brocal V, Gil R, Ramos S, Lamelas A, Postigo M, Michelena JM, Silva FJ, Moya A, Latorre A. A small microbial genome: the end of a long symbiotic relationship? Science 2006; 314:312-313.
76. Moran NA, Russell JA, Koga R, Fukatsu T. Evolutionary relationships of three new species of Enterobacteriaceae living as symbionts of aphids and other insects. Appl. Environ. Microbiol. 2005; 71:3302-3310.
77. Nakabachi A, Yamashita A, Toh H, Ishikawa H, Dunbar HE, Moran NA, Hattori M. The 160-kilobase genome of the bacterial endosymbiont Carsonella. Science 2006; 314:267.
78. Dillon RJ, Dillon VM. The gut bacteria of insects: nonpathogenic interactions. Annu. Rev. Entomol. 2004; 49:71-92.
79. Yang H, Peng JX, Liu KY, Hong HZ. Diversity and function of symbiotic microbes in the gut of lower termites. Wei Sheng Wu Xue Bao. 2006; 46:496-499.
80. Iida T, Ohkuma M, Ohtoko K, Kudo T. Symbiotic spirochetes in the termite hindgut: phylogenetic identification of ectosymbiotic spirochetes of oxymonad protists. FEMS Microbiol. Ecol. 2000; 34:17-26.
81. Martin MM. The evolution of cellulose digestion in insects. Phil. Trans. R. Soc. Lond. B 1991; 333:281-288.
82. Zourari A, Accolas JP, Desmazeaud MJ. Metabolism and biochemical characteristics of yogurt bacteria. A review. Lait 1992; 72:1-34.
83. Radke-Mitchell L, Sandine WE. Associative growth and differential enumeration of Streptococcus thermophilus and Lactobacillus bulgaricus, a review. J. Food Prot. 1984; 47:245-248.
84. Elias TS. The Biology of Nectaries. Bentley B, Elias TS, eds. 1983. Columbia Univ. Press, New York. pp. 174-203.
85. Heil M, McKey D. Protective ant-plant interactions as model systems in ecological and evolutionary research. Annu. Rev. Ecol. Evol. Syst. 2003; 34:425-553.
86. Mayer AM, Hamann MT. Marine pharmacology in 1999: compounds with antibacterial, anticoagulant, antifungal, anthelmintic, anti-inflammatory, antiplatelet, antiprotozoal and antiviral activities affecting the cardiovascular, endocrine, immune and nervous systems, and other miscellaneous mechanisms of action. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2002; 132:315-339.
87. Bewley CA, Faulkner DJ. Lithistid Sponges: star performers or hosts to the stars. Angew. Chem. Int. Ed. 1998; 37:2162-2178.
88. Faulkner DJ. Highlights of marine natural products chemistry (1972-1999). Nat. Prod. Rep. 2000; 17:1-6.
89. Proksch P, Edrada RA, Ebel R. Drugs from the sea—current status and microbiological implications. Appl. Microbiol. Biotechnol. 2002; 59:125-134.
90. Hentschel U, Hopke J, Horn M, Friedrich AB, Wagner M, Hacker J, Moore BS. Molecular evidence for a uniform microbial community in sponges from different oceans. Appl. Environ. Microbiol. 2002; 68:4431-4440.
91. Webster NS, Wilson KJ, Blackall LL, Hill RT. Phylogenetic diversity of bacteria associated with the marine sponge Rhopaloeides odorabile. Appl. Environ. Microbiol. 2001; 67:434-444.
92. Lafi FF, Garson MJ, Fuerst JA. Culturable bacterial symbionts isolated from two distinct sponge species (Pseudoceratina clavata and Rhabdastrella globostellata) from the Great Barrier Reef display similar phylogenetic diversity. Microb. Ecol. 2005; 50:213-220.
93. Kaye HR. Sexual reproduction in four Caribbean commercial sponges. 11. Oogenesis and transfer of bacterial symbionts. Invert. Reprod. Dev. 1991; 19:13-24.
94. Narquizian R, Kocienski PJ. The pederin family of antitumor agents: structures, synthesis and biological activity. In The Role of Natural Products in Drug Discovery, Vol. 32. Mulzer R, Bohlmann R, eds. 2000. Springer, Heidelberg, Germany. pp. 25-56.
95. Piel J, Hui D, Wen G, Butzke D, Platzer M, Fusetani N, Matsunaga S. Antitumor polyketide biosynthesis by an uncultivated bacterial symbiont of the marine sponge Theonella swinhoei. Proc. Natl. Acad. Sci. U.S.A. 2004; 101:16222-16227.
96. Hrvatin S, Piel J. Rapid isolation of rare clones from highly complex DNA libraries by PCR analysis of liquid gel pools. J. Microbiol. Methods. e-pub 2006.
97. Bewley CA, Holland ND, Faulkner DJ. Two classes of metabolites from Theonella swinhoei are localized in distinct populations of bacterial symbionts. Experientia 1996; 52:716-722.
98. Unson MD, Faulkner DJ. Cyanobacterial symbiont biosynthesis of chlorinated metabolites from Dysidea herbacea (Porifera). Experientia 1993; 49:349-353.
99. Flowers AE, Garson MJ, Webb RI, Dumdei EJ, Charan RD. Cellular origin of chlorinated diketopiperazines in the dictyoceratid sponge Dysidea herbacea (Keller). Cell Tissue Res. 1998; 292:597-607.
100. Unson MD, Holland ND, Faulkner DJ. A brominated secondary metabolite synthesized by the cyanobacterial symbiont of a marine sponge and accumulation of the crystalline metabolite in the sponge tissue. Mar. Biol. 1994; 119:1-11.
101. Enticknap JJ, Kelly M, Peraud O, Hill RT. Characterization of a culturable alphaproteobacterial symbiont common to many marine sponges and evidence for vertical transmission via sponge larvae. Appl. Environ. Microbiol. 2006; 72:3724-3732.
102. Correale P, Caraglia M, Fabbrocini A, Guarrasi R, Pepe S, Patella V, Marone G, Pinto A, Bianco AR, Tagliaferri P. Bryostatin 1 enhances lymphokine activated killer sensitivity and modulates the beta 1 integrin profile of cultured human tumor cells. Anticancer Drugs 1995; 6:285-290.
103. Sung SJ, Lin PS, Schmidt-Ullrich R, Hall CE, Walters JA, McCrady C, Grant S. Effects of the protein kinase C stimulant bryostatin 1 on the proliferation and colony formation of irradiated human T-lymphocytes. Int. J. Radiat. Biol. 1994; 66:775-783.
104. Kortmansky J, Schwartz GK. Bryostatin-1: a novel PKC inhibitor in clinical development. Cancer Invest. 2003; 21:924-936.
105. Lopanik N, Lindquist N, Targett N. Potent cytotoxins produced by a microbial symbiont protect host larvae from predation. Oecologia 2004; 139:131-139.
106. Davidson SK, Allen SW, Lim GE, Anderson CM, Haygood MG. Evidence for the biosynthesis of bryostatins by the bacterial symbiont “Candidatus Endobugula sertula” of the bryozoan Bugula neritina. Appl. Environ. Microbiol. 2001; 67:4531-4537.
107. Haygood MG, Davidson SK. Small-subunit rRNA genes and in situ hybridization with oligonucleotides specific for the bacterial symbionts in the larvae of the bryozoan Bugula neritina and proposal of “Candidatus Endobugula sertula”. Appl. Environ. Microbiol. 1997; 63:4612-4616.
108. Hildebrand M, Waggoner LE, Liu H, Sudek S, Allen S, Anderson C, Sherman DH, Haygood MG. bryA: An unusual modular polyketide synthase gene from the uncultivated bacterial symbiont of the bryozoan Bugula neritina. Chem. Biol. 2004; 11:1543-1552.
109. Sings HL, Rinehart KL. Compounds produced from potential tunicate-blue-green algal symbiosis: A review. J. Ind. Microbiol. Biotechnol. 1996; 17:385-396.
110. Lewin RA, Cheng L, eds. Prochloron: A Microbial Enigma. 1989. Chapman and Hall, New York.
111. Burns BP, Goh F, Allen M, Neilan BA. Microbial diversity of extant stromatolites in the hypersaline marine environment of Shark Bay, Australia. Environ. Microbiol. 2004; 6:1096-1101.
112. Degnan BM, Hawkins CJ, Lavin MF, McCaffrey EJ, Parry DL, van den Brenk AL, Watters DJ. New cyclic peptides with cytotoxic activity from the ascidian Lissoclinumpatella. J. Med. Chem. 1989; 32:1349-1354.
113. Fu X, Do T, Schmitz FJ, Andrusevich V, Engel MH. New cyclic peptides from the ascidian Lissoclinum patella. J. Nat. Prod. 1998; 61:1547-1551.
114. Schmidt EW, Nelson JT, Rasko DA, Sudek S, Eisen JA, Haygood MG, Ravel J. Patellamide A and C biosynthesis by a microcin-like pathway in Prochloron didemni, the cyanobacterial symbionts of Lissoclinum patella. Proc. Natl. Acad. Sci. U.S.A. 2005; 102:7315-7320.
115. Long PF, Dunlap WC, Battershill CN, Jaspars M. Shotgun cloning and heterologous expression of the patellamide gene cluster as a strategy to achieving sustained metabolite production. Chembiochem. 2005; 6:1760-1765.
116. Donia MS, Hathaway BJ, Sudek S, Haygood MG, Rosovitz MJ, Ravel J, Schmidt EW. Natural combinatorial peptide libraries in cyanobacterial symbionts of marine ascidians. Nat. Chem. Biol. 2006; 2:729-735.
117. Rodriguez II, Shi YP, Garcia OJ, Rodriguez AD, Mayer AMS, Sanchez JA, Ortega-Barria E, Gonzalez J. New pseudopterosin and seco-pseudopterosin diterpene glycosides from two Colombian isolates of Pseudopterogorgia elisabethae and their diverse biological activities. J. Nat. Prod. 2004; 67:1672-1680.
118. Mayer AMS, Jacobson PB, Fenical W, Jacobs RS, Glaser KB. Pharmacological characterization of the pseudopterosins: novel anti-inflammatory natural products isolated from the Caribbean soft coral, Pseudopterogorgia elisabethae. Life Sci. 1998; 62:PL401-407.
119. Puyana M, Narvaez G, Paz A, Osorno O, Duque C. Pseudopterosin content variability of the purple sea whip Pseu- dopterogorgia elisabethae at the islands of San Andres and Providencia (SW Caribbean). J. Chem. Ecol. 2004; 30:1183-1201.
120. Mydlarz LD, Jacobs RS, Boehnlein J, Kerr RG. Pseudopterosin biosynthesis in Symbiodinium sp., the dinoflagellate symbiont of Pseudopterogorgia elisabethae. Chem. Biol. 2003; 10:1051-1056.
121. Kohl AC, Kerr RG. Identification and characterization of the pseudopterosin diterpene cyclase, elisabethatriene synthase, from the marine gorgonian, Pseudopterogorgia elisabethae. Arch. Biochem. Biophys. 2004; 424:97-104.
122. Mydlarz LD, Jacobs RS. Comparison of an inducible oxidative burst in free-living and symbiotic dinoflagellates reveals properties of the pseudopterosins. Phytochem. 2004; 65:3231-3241.
123. Tchernov D, Gorbunov MY, deVargas C, Yadav SN, Milligan AJ, Haggblom M, Falkowski PG. Membrane lipids of symbiotic algae are diagnostic of sensitivity to thermal bleaching in corals. Proc. Natl. Acad. Sci. U.S.A. 2004; 101:13531-13535.
124. Currie CR. A community of ants, fungi, and bacteria: a multilateral approach to studying symbiosis. Annu. Rev. Microbiol. 2001; 55:357-380.
125. Mueller UG, Schultz TR, Currie CR, Adams RMM, Malloch D. The origin of the attine ant-fungus mutualism. Q. Rev. Biol. 2001; 76:169-197.
126. Seifert KA, Samson RA, Chapela IH. Escovopsis aspergilloides, a rediscovered hyphomycete from leaf-cutting ant nests. Mycologia 1995; 87:407-413.
127. Currie CR, Scott JA, Summerbell RC, Malloch D. Fungus-growing ants use antibiotic-producing bacteria to control garden parasites. Nature 1999; 398:701-704. Erratum in: Nature 2003; 423:461.
128. Kaltenpoth M, Gottler W, Herzner G, Strohm E. Symbiotic bacteria protect wasp larvae from fungal infestation. Curr. Biol. 2005; 15:475-479.
129. Kaltenpoth M, Goettler W, Dale C, Stubblefield JW, Herzner G, Roeser-Mueller K, Strohm E. 'Candidatus Streptomyces philanthi’, an endosymbiotic streptomycete in the antennae of Philanthus digger wasps. Int. J. Syst. Evol. Microbiol. 2006; 56:1403-1411.
130. Kellner RLL, Dettner K. Allocation of pederin during lifetime of Paederus rove beetles (Coleoptera: Staphylinidae): evidence for polymorphism of hemolymph toxin. J. Chem. Ecol. 1995; 21:1719-1733.
131. Piel J. A polyketide synthase-peptide synthetase gene cluster from an uncultured bacterial symbiont of Paederus beetles. Proc. Natl. Acad. Sci. U.S.A. 2002; 99:14002-14007.
132. Kellner RLL. Molecular identification of an endosymbiotic bacterium associated with pederin biosynthesis in Paederus sabaeus (Coleoptera: Staphylinidae). Insect Biochem. Mol. Biol. 2002; 32:389-395.
133. Kellner RLL. What is the basis of pederin polymorphism in Paederus riparius rove beetles? The endosymbiotic hypothesis. Entomol. Exp. Appl. 1999; 93:41-49.
134. Piel J, Hoefer I, Hui D. Evidence for a symbiosis island involved in horizontal acquisition of pederin biosynthetic capabilities by the bacterial symbiont of Paederus fuscipes beetles. J. Bacteriol. 2004; 186:1280-1286.
135. Forst S, Dowds B, Boemare N, Stackebrandt E. Xenorhabdus and Photorhabdus spp.: bugs that kill bugs. Annu. Rev. Microbiol. 1997; 51:47-72.
136. Forst S, Nealson K. Molecular biology of the symbiotic-pathogenic bacteria Xenorhabdus spp. and Photorhabdus spp. Microbiol. Rev. 1996; 60:21-43.
137. Akhurst R, Dunphy GB. Tripartite interactions between symbiotically associated entomopathogenic bacteria, nematodes, and their insect hosts. In Parasites and Pathogens of Insects 2. Beckage N, Thompson S, Federici B, eds. 1993. Academic Press, New York. pp. 1-23.
138. Poinar GO Jr. Biology and taxonomy of Steinermatidae and Het- erorhabditidae. In Entomopathogenic Nematodes in Biological Control. Gaugler R, Kaya HK, eds. 1990. CRC Press, Boca Raton, FL. pp. 23-62.
139. ffrench-Constant RH, Bowen DJ. Novel insecticidal toxins from nematode-symbiotic bacteria. Cell. Mol. Life Sci. 2000; 57:828-833.
140. Hu K, Li J, Webster JM. Nematicidal metabolites produced by Photorhabdus luminescens (Enterobacteriaceae), bacterial symbiont of entomopathogenic nematodes. Nematology 1999; 1:457- 469.
141. Hu K, Li J, Wang W, Wu H, Lin H, Webster JM. Comparison of metabolites produced in vitro and in vivo by Photorhabdus luminescens, a bacterial symbiont of the entomopathogenic nematode Heterorhabditis megidis Can. J. Microbiol. 1998; 44:1072-1077.
142. Ciche TA, Blackburn M, Carney JR, Ensign JC. Photobactin: a catechol siderophore produced by Photorhabdus luminescens, an entomopathogen mutually associated with Heterorhabdi- tis bacteriophora NC1 nematodes. Appl. Environ. Microbiol. 2003; 69:4706-4713.
143. Li J, Chen G, Webster JM, Czyzewska E. Antimicrobial metabolites from abacterial symbiont. J. Nat. Prod. 1995; 58:1081-1086.
144. Mclnerney BV, Taylor WC, Lacey MJ, Akhurst RJ, Gregson RP. Biologically active metabolites from Xenorhabdus spp., Part 2. Benzopyran-1-one derivatives with gastroprotective activity. J. Nat. Prod. 1991; 54:785-795.
145. Paik S, Park YH, Suh SI, Kim HS, Lee IS, Park MK, Lee CS, Park SH. Unusual cytotoxic phenethylamides from Xenorhabdus nematophilus. Bull. Korean Chem. Soc. 2001; 22:372-374.
146. Paul VJ, Frautschy S, Fenical W, Nealson KH. Antibiotics in microbial ecology. J. Chem. Ecol. 1981; 7:589-597.
147. Poinar GO Jr, Thomas G, Haygood M, Nealson KH. Growth and luminescence of the symbiotic bacteria associated with the terrestrial nematode Heterorhabditis bacteriophora. Soil Biol. Biochem. 1980; 12:5-10.
148. Farmer JJ III. Other genera of the family of Enterbacteriaceae. In Bergey’s Manual of Systematic Bacteriology 1. Krieg, NR, Holt, JG, eds. 1984. Williams & Wilkins, Baltimore, MD. pp. 506-516.
149. Iwasaki S, Kobayashi H, Furukawa J, Namikoshi M, Okuda S, Sato Z, Matsuda I, Noda T. Studies on macrocyclic lactone antibiotics. VII. Structure of a phytotoxin “rhizoxin” produced by Rhizopus chinensis. J. Antibiot. 1984; 37:354-362.
150. Noda T, Hashiba T, Sato Z. The structural changes in young swollen roots of rice seedlings infected with Rhizopus chinensis Saito. Ann. Phytopathol. Soc. Jpn. 1980; 46:40-45.
151. Tsuruo T, Oh-hara T, Iida H, Tsukagoshi S, Sato Z, Matsuda I, Iwasaki S, Okuda S, Shimizu F, Sasagawa K, Fukami M, Fukuda K, Arakawa M. Rhizoxin, a macrocyclic lactone antibiotic, as a new antitumor agent against human and murine tumor cells and their vincristine-resistant sublines. Cancer Res. 1986; 46:381-385.
152. Cassidy J, Kaye SB. New drugs in clinical development in Europe. Hematol. Oncol. Clin. North Am. 1994; 8:289-303.
153. Partida-Martinez LP, Hertweck C. Pathogenic fungus harbours endosymbiotic bacteria for toxin production. Nature 2005; 437:884-888.
154. Scherlach K, Partida-Martinez LP, Dahse HM, Hertweck C. Antimitotic rhizoxin derivatives from a cultured bacterial endosymbiont of the rice pathogenic fungus Rhizopus microsporus. J. Am. Chem. Soc. 2006; 128:11529-11536.
155. Gunatilaka AA. Natural products from plant-associated microorganisms: distribution, structural diversity, bioactivity, and implications of their occurrence. J. Nat. Prod. 2006; 69:509-526.
156. Porter JK. Ergot alkaloids and alkaloids from other endophytes, responsible for causing toxic syndrome in cattle after eating contaminated grass. Prikl. Biokhim. Mikrobiol. 1993; 29:51-55.
157. Porter JK. Analysis of endophyte toxins: fescue and other grasses toxic to livestock. J. Anim. Sci. 1995; 73:871-880.
158. Steiner U, Ahimsa-Muller MA, Markert A, Kucht S, Gross J, Kauf N, Kuzma M, Zych M, Lamshoft M, Furmanowa M, Knoop V, Drewke C, Leistner E. Molecular characterization of a seed transmitted clavicipitaceous fungus occurring on dicotyledoneous plants (Convolvulaceae). Planta. 2006; 224:533-544.
159. Tan RX, Zou WX. Endophytes: a rich source of functional metabolites. Nat. Prod. Rep. 2001; 18:448-459.