CHEMICAL BIOLOGY
Terpenoids in Plants
Christopher I. Keeling, Michael Smith Laboratories, University of British Columbia, British Columbia, Canada
JOrg Bohlmann, Michael Smith Laboratories and Departments of Botany and Forest Sciences, University of British Columbia, British Columbia, Canada
doi: 10.1002/9780470048672.wecb596
Terpenoids are the largest class of all known natural products. Plants produce a variety of terpenoid compounds that number in the thousands. Some terpenoids are involved in plant growth and development directly (i.e., in primary metabolism), but most plant terpenoids are thought to function in interactions of plants with their biotic and abiotic environment and have traditionally been referred to as secondary metabolites. In addition to the isolation and identification of plant terpenoids, research has concentrated on the biosynthesis, the biological function, and the exploitation of plant terpenoids for human use as biomaterials and pharmaceuticals. Plant terpenoids are biosynthesized from C5 precursors by the action of prenyl transferases and terpenoid synthases. Often, terpenes are acted on by cytochromes P450 and other enzymes to increase their functionalization. Terpenoid biosynthesis in plants involves several subcellular compartments. The accumulation of terpenoids requires efficient transport systems and specialized anatomical structures. Using isoprene (a hemiterpene), menthol (a monoterpene), artemisinin (a sesquiterpene), and paclitaxel [better known under the registered trademark Taxol (Bristol Myers Squibb, New York)] and diterpene resin acids (diterpenes) as examples, we highlight some strategies, techniques, and results of plant terpenoid research with a strict focus on the low-molecular-weight (C5-C20) terpenoids of specialized plant metabolism.
All plant terpenoids are derived from C5 precursors, and most plant terpenoids can be grouped according to their number of C5 building blocks as hemiterpenoids (C5), monoterpenoids (C10), sesquiterpenoids (C15), diterpenoids (C20), or polyterpenoids (C5xn). In addition, condensation of C15- and C20-intermediates give rise to triterpenoids (C30) and tetraterpenoids (C40), respectively. Many irregular terpenoids or terpenoid derivates are also found in plants. Terpenoids are involved in all forms of plant interactions with other organisms including plant reproduction, defense, and signaling. They are the most diverse group of plant chemicals and have been used by humans for centuries in both traditional and modern industrial applications. As such, plant terpenoids are used widely as pharmaceuticals, flavor and aroma chemicals, vitamins, pigments, and large-volume biological feedstock for the production of a suite of industrial materials such as, for example, industrial resins and print inks. Plant terpenoids are an important group of natural product chemicals that are actively being explored as alternatives for petroleum-based materials.
Biological Background
Over 20,000 terpenoids have been identified (1), and more are being discovered continuously. Plant terpenoids are important in both primary and secondary (specialized) metabolism. Their importance in primary metabolism includes physiological, metabolic, and structural roles such as plant hormones, chloroplast pigments, roles in electron transport systems, and roles in the posttranslational modification of proteins. In secondary metabolism, the roles of plant terpenoids are incredibly diverse but are associated most often with defense and communication of sessile plants interacting with other organisms. Examples include terpenoid chemicals that form physical and chemical barriers, antibiotics, phytoalexins, repellents and antifeedants against insects and other herbivores, toxins, attractants for pollinators or fruit-dispersing animals, host/nonhost selection cues for herbivores, and mediators of plant-plant and mycorrhiza interactions (2, 3).
Some plants produce terpenoids in specialized cells or tissues such as the glandular trichomes on the surface of peppermint leaves, scent-releasing epidermal cells of certain flowers, or the epithelial cells that surround the resin ducts of conifers. These structures place high concentrations of terpenoids in areas most likely to be encountered by the interacting organism.
The biosynthetic pathways of plant terpenoids are highly regulated and highly spatially organized in subcelluar compartments and sometimes in specialized cells. Terpenoid biosynthesis can be regulated by plant hormones, developmental programs, diurnal cycles, herbivory, or pathogen infection. Identifying what role specific terpenoids play in plants, how and where they are biosynthesized, and how their biosyntheses are regulated allows us to better understand their importance to the survival of the plant and thus make use of this knowledge in crop improvement or in the production of terpenoids for medicinal or industrial uses.
Chemistry
Much chemistry research in plant terpenoids has been to elucidate the structure, define the biosynthetic pathways, characterize the enzymes involved, and develop systems for the large-scale production of medicinally or industrially important terpenoids (4). Progress on the identification and the study of plant terpenoids is reviewed regularly in the journal Natural Product Reports, and the biosynthesis of terpenoids, including plant terpenoids, has been reviewed comprehensively (5). Plant terpenoids can be volatile or nonvolatile, lipophilic or hydrophilic, cyclic or acyclic, chiral or achiral, and they often have double-bond stereochemistry. The chemical diversity of terpenoid structures originates largely from the terpenoid synthase enzymes that stabilize different carbocation intermediates, allow rearrangements or water termination, and direct stereochemistry; the diversity also originates from the many different terpenoid-modifying enzymes.
Two major complementary approaches to studying plant terpenoids have been established. One approach involves the isolation and the structural identification of terpenoid chemicals of interest from plant tissues based on traditional natural products research followed by targeted search for the relevant enzymes and genes that control biosynthesis. The second approach explores the emerging plant genome sequences to discover complete sets of genes that encode terpenoid biosynthetic enzymes. The combination of these two approaches is the most powerful approach to a comprehensive understanding of plant terpenoid chemistry and its biosynthetic origins.
The diversity of plant terpenoids reflects the complexity and the diversity of the pathways that biosynthesize them. The recent sequencing of the genomes of four different plant species and large collections of expressed sequence tags (ESTs) from many other plants may indicate the diversity of pathways and chemicals we might expect in any one species. For example, the genes that encode putatively active terpenoid synthases (TPS) comprise at least 32 in the Arabidopsis (Arabidopsis thaliana) genome (6), at least 15 in the rice (Oryza sativa) genome (7), at least 47 in the poplar (Populus trichocarpa) genome (8), and at least 89 in the genome of a highly inbred grapevine (Vitis vinifera) Pinot Noir variety (9). The large gene family of TPS, which is important to generating structural diversity of terpenoid chemicals in plants, apparently results from repeated gene duplication and subsequent neofunctionalization or subfunctionalization (10, 11). Most TPS produce more than one product from a single substrate, and these products are often modified by the action of additional enzymes such as cytochromes P450 and reductases. Thus, the number of distinct terpenoids found in any one plant species is predicted to be manifold higher than the number of TPS genes present in that species. Genomics approaches, which can identify the candidate genes for terpenoid production, together with functional characterization of heterologously expressed enzymes and the identification of the resulting plant terpenoids, can enhance the discovery of the biochemical pathways substantially in planta as has been demonstrated in recent years with research in Arabidopsis (12), rice (13), and grapevine (14). Ideally, the functional genomics approach is combined with classical and modern approaches of isolation, identification, and metabolite profiling of terpenoids from plant tissue.
The combined genomics and chemical approaches to plant terpenoid research are not restricted to the few plant species for which more or less complete genome sequences are now available. The discovery of many of the genes and enzymes for the formation of terpenoids such as menthol and related monoterpenes in peppermint (Mentha x piperita) (15), artemisinin in Artemisia annua (16), Taxol in the yew tree (Taxus) (17), or conifer diterpene resin acids in species of spruce (Picea) and pine (Pinus) (18) have been possible on the foundation of highly specialized efforts of EST and full-length cDNA sequencing combined with characterization of recombinant enzymes and analysis of the terpenoid metabolome of the target plant species.
Common steps in plant terpenoid biosynthesis
The universal precursors to terpenoids, the C5-compounds dimethylallyl pyrophosphate (DMAPP) and isopentenyl pyrophosphate (IPP), originate from two pathways in plants (Fig. 1). The mevalonate (MEV) pathway is well described in many eukaryotic organisms. This pathway is present in the cytosol/endoplasmic reticulum of plants. More recently, another pathway has been described, the 2C-methyl-D-erythritol-4-phosphate (MEP) pathway, which is found in the plastids of plants (19). The localization of the different pathways and the plastid-directing transit peptides found in hemi-TPS, mono-TPS, and di-TPS, but not in sesqui-TPS, result in the production of terpenoids from at least two different precursors pools.
Hemiterpenoids are produced from the isoprenyl diphosphate DMAPP. All other terpenoids are produced from DMAPP and IPP via longer-chain prenyl diphosphate intermediates formed by prenyl transferases. Prenyl transferases (20) catalyze the formation of geranyl diphosphate (GPP), farnesyl diphosphate (FPP), and geranylgeranyl diphosphate (GGPP) from one molecule of DMAPP and one, two, or three molecules of IPP, respectively (Fig. 1). Isoprenyl diphosphates are the substrates for all TPS, which lead to the hemiterpenoids, monoterpenoids, sesquiterpenoids, and diterpenoids, which will be highlighted with selected examples in the following sections.

Figure 1. The two pathways to the universal precursors of plant terpenoids.
Isoprene (C5, hemiterpene)
Isoprene (2-methyl 1,3-butadiene, Fig. 2) is the simplest terpenoid produced by many plants, and is produced abundantly by some tree species such as poplars. The mechanism of isoprene biosynthesis is a straightforward diphosphate ionization of the DMAPP precursor followed by deprotonation of the allylic cation (Fig. 2). Only a few isoprene synthases have been identified and characterized in plants, from Populus spp. (poplar) (21, 22) and Pueraria montana (kudzu vine) (23). These TPS contain a transit peptide that targets the plastids, and thus isoprene biosynthesis is derived from products of the MEP pathway.
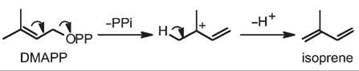
Figure 2. Pathway of isoprene biosynthesis.
Isoprene is a major biogenic volatile organic compound (VOC; rivalling methane in global production) with emission from plants estimated in the order of more than 1012 kg per year and therefore has been well studied for its role in atmospheric chemistry (24). Isoprene is also important in the context of global cycles of carbon fixation versus carbon emission from natural sources. Although isoprene is emitted in large amounts from poplars, which are actively being promoted as plantation species for biofuel (ethanol) production, its physiological function in plants is somewhat unclear. The protection from thermal and oxidative stress as well as release of excess carbon flux and photosynthetic energy are thought to be the main functions of isoprene in plants (21, 22).
Very recently, new molecular approaches have established a function of isoprene in thermotolerance through the use of under- and over-producing transgenic lines of poplar (25) or Arabidopsis (which normally produces no isoprene) using the poplar isoprene synthase (26). Other possible functions of isoprene remain to be tested using similar molecular approaches. The effect of down-regulation of isoprene emission in poplars remains to be tested in the field to explore whether it is viable to reduce emission of this biogenic VOC in plantation forests for biofuels production to maximize carbon fixation and minimize carbon emission. A closely related hemiterpene of plant origin is 2-methyl-3-buten-2-ol. It is produced abundantly in needles of conifers and is emitted into the atmosphere (27).
(-)-Menthol (C10, monoterpenoid)
(-)-Menthol is a well-known terpenoid from the essential oil of mint (Mentha spp.) (15), and is described here as a representative of the different acyclic and cyclic plant monoterpenoids. Because of its pleasant odor, taste, and anesthetic and antimicrobial effects, (-)-menthol is an industrially important terpenoid and is produced commercially in large scale both from the essential oils of Mentha spp. and by asymmetric synthesis. The essential oil is produced in glandular trichomes, which are secretory cells that number in the thousands on Mentha leaves. The presence of these specialized cells, which easily can be separated physically from other cell types, has greatly facilitated studying (-)-menthol biosynthesis.
(-)-Menthol biosynthesis involves a series of enzymes (Fig. 3) that first generate a cyclic monoterpene and then functionalize it. The biochemistry of this pathway has been elucidated by substrate feeding, cell-free enzyme assays with plant extracts, and characterization of cloned and recombinantly expressed enzymes (28). The biosynthesis of menthol, which has been studied for more than two decades by Croteau et al. (reviewed in Reference 15), is arguably the best-characterized pathway of a functionalized monoterpenoid and can serve as a paradigm for many monoterpenoids in plants in general. First, using geranyl diphosphate as substrate, (-)-limonene synthase generates (-)-limonene and minor amounts of myrcene, (-)-alpha-pinene, and (-)-beta-pinene. (-)-Limonene then undergoes a series of transformations ultimately to yield (-)-menthol. These modifications involve first the allylic hydroxylation to (-)-trans -isopiperitenol by the cytochrome P450 limonene-3-hydroxylase (CYP71D13). Allylic oxidation of this alcohol to (-)-isopiperitenone is then catalyzed by the NAD-dependent isopiperitenol dehydrogenase. Subsequently, NADPH-dependent (-)-isopiperitenone reductase catalyzes the formation of (+)-cis-isopulegone. (+)-cis -Isopulegone is enzymatically isomerized to the more stable alpha,beta-unsaturated ketone—(+)-pulegone—by (+)-cis-isopulegone isomerase. (-)-Menthone and (+)-isomenthone (in a 2:1 to 10:1 ratio) are then formed by the action of the NADPH-dependent (+)-pulegone reductase. Finally, (-)-menthone reductase reduces (-)-menthone to (-)-menthol. All of these enzymes and their corresponding genes in Mentha have been isolated, functionally characterized, and their enzymology studied (15). For some enzymes, substrate specificities or product outcomes after directed mutations as well as enzyme localization have been investigated. The structure of (-)-limonene synthase has been determined recently (29). Based on the detailed knowledge of (-)-menthol and its biosynthetic pathway, it has become possible to improve the composition of the monoterpene-rich essential oil of Mentha through metabolic engineering (30, 31).
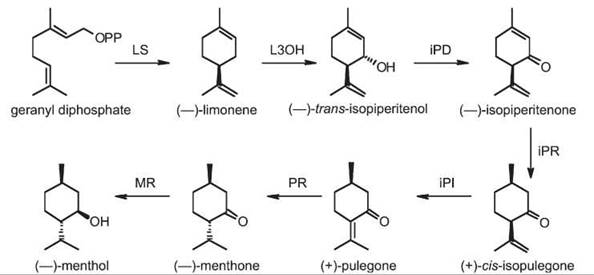
Figure 3. Pathway of (-)-menthol biosynthesis in Mentha. LS, (-)-limonene synthase; L3OH, (-)-limonene-3-hydroxylase; iPD, (-)-trans-isopiperitenol dehydrogenase; iPR, (-)-isopiperitenone reductase; iPI, (+)-cis-isopulegone isomerase; PR, (+)-pulegone reductase; MR, (-)-menthone reductase.
Artemisinin (C15, sesquiterpenoid)
Artemisinin is used here as an example of a plant sesquiterpenoid with both traditional value as well as with medicinal and social value in the twenty-first century. Research on artemisinin has also established new benchmarks for biochemical engineering and functional genomics of plant terpenoids. Artemisinin is a functionalized sesquiterpene with a unique peroxide linkage from the sweet wormwood (Artemisia annua). Chinese herbalists have used it since ancient times, and it is now used for its unique efficacy to treat multidrug-resistant strains of the malaria parasite Plasmodium falciparum. Its medicinal importance has prompted studies into its biosynthesis and its biochemical engineering so that cost-effective methods for producing it in large scale and in consistent quality may be realized.
Biosynthesis of artemisinin in planta begins with the formation of the sesquiterpene amorpha-4,11-diene in glandular tricomes of A. annua leaves (Fig. 4) by amorpha-4,11-diene synthase (32, 33). Amorphadiene is oxidized to artemisinic acid in three steps by a multifunctional cytochrome P450 (CYP71AV1) (16). The remaining steps have not yet been established but are predicted to include nonenzymatically catalyzed photooxidation reactions (34). The application of a semi-synthetic route from artemisinic acid to artemisinin, along with the availability of the characterized plant enzymes to produce artemisinic acid described above from A. annua, have permitted the complete synthesis of artemisinin via microbial host cells (16). Introducing these enzymes into Escherichia coli or Saccharomyces cerevisiae and engineering an unnatural, or fine-tuning the natural, mevalonate pathway in these microorganisms have resulted in significant production of artemisinic acid in fermentations (16, 35, 36).
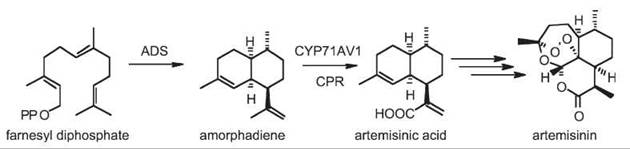
Figure 4. Pathway of artemisinin biosynthesis in Artemisia annua. ADS, amorphadiene synthase; CPR, cytochrome P450 reductase.
Taxol (C20, diterpenoid)
Taxol is another example of a medicinally important functionalized plant terpene, in this case a diterpenoid, with large pharmaceutical and economic value (Fig. 5). Taxol is a potent anticancer drug that was isolated and identified from the bark of the Pacific yew (Taxus brevifolia) more than 35 years ago (37). The name Taxol is now a registered trademark, but the literature commonly uses this name rather than the generic name paclitaxel. The total synthesis of Taxol is possible (38), but it is not economically feasible currently because of the challenges of stereochemistry, low yield, and high cost. The study of Taxol by Croteau et al. (17) is an exceptional example of how a terpenoid biosynthetic pathway was rationalized, and the synthesis and testing of various hypothetical precursors with cell-free extracts have yielded the discovery of many enzymes and genes in this complex pathway. Because Taxol is only found at very low levels in slow growing trees, and is one of hundreds of Taxol-like compounds produced in a metabolic grid, the use of an inducible Taxus cell culture system has accelerated this research.
Taxol is biosynthesized in 19 steps from GGPP that originates from precursors of the MEP pathway (Fig. 5). The biosynthesis of Taxol begins with the formation of the tricyclic diterpene skeleton of taxa-4(5),11(12)-diene (17, 39). All genes for the enzymes in this early pathway have been identified in Taxus cuspidata and a taxa-4(5),11(12)-diene synthase has been identified in several Taxus species. The mechanism of this di-TPS has been explored in detail (39). Taxa-4(5),11(12)-diene is then hydroxylated by several cytochrome P450 taxoid oxygenases to yield a putative intermediate decorated with seven alcohol or ester groups. Many cytochrome P450 enzymes that catalyze these transformations have been identified and characterized, but two enzymes remain uncharacterized (17). The biosyntheses of the ester functionalities have been studied, and several acyl and aroyl transferases have been identified and characterized. Finally, studies on the steps in the aromatic side chain assembly and attachment has yielded several enzymes that include a phenylalanine aminomutase, a C13-phenylpropanoyl-CoA transferase, and an N-benzoyl transferase. Although the pathway of biosynthesis has not been resolved fully, what is known can be applied to improve Taxol production, which continues to rely on Taxus plants or cell cultures.

Figure 5. Pathway of Taxol biosynthesis in Taxus spp.
Diterpene resin acids (C20, diterpenoids)
Diterpene resin acids are abundantly produced in conifers of the pine family (Pinaceae) and in other plant species (Fig. 6). They are produced in the epithelial cells that surround the resin ducts that are found constitutively, or they are induced in the xylem upon wounding and are important for the physical and chemical plant defenses against herbivores and pathogens (18, 40). Industrially, diterpene resin acids are important chemicals for the naval stores industry, in printing inks, as potential antimicrobials and pharmaceuticals, and are byproducts of wood pulping processes.
Two major steps exist in the biosynthesis of diterpene resin acids: the formation of the diterpene and the stepwise oxidation of the diterpene to the corresponding acid. Most conifer diterpenes are tricyclic; they are biosynthesized by bifunctional di-TPS that first cyclize geranylgeranyl diphosphate to (+)-copalyl diphosphate and then cyclize this intermediate even more to form the various diterpenes (11, 18) (Fig. 6). Some di-TPS produce multiple products, whereas others produce only single products. These diterpenes are then oxidized stepwise to the resin acids by multifunctional, multisubstrate cytochromes P450 (41).

Figure 6. Pathway of conifer diterpene resin acid biosynthesis. Bifunctional di-TPS convert geranylgeranyl diphosphate to various diterpenes, which are oxidized stepwise by multisubstrate and multifunctional cytochromes P450 to the corresponding diterpene acid. The oxidation of (-)-abietadiene to (-)-abietic acid is shown as an example.
Chemical Tools and Techniques
The study of plant terpenoids shares many of the same tools for their isolation, identification, characterization, and synthesis that are required in other natural product research. Advances in separation science, analytical chemistry, spectroscopic tools, and synthetic organic chemistry all affect the study of terpenoids in plants.
Extraction and separation
Isolation of the plant terpenoids usually begins with some form of extraction from the plant source. Certain tissue types such as roots, leaves, or flowers are often extracted. Often, the terpenoids of interest are produced in very specialized tissues on plant surfaces or within the plant such as the glandular trichomes of Mentha spp. or the resin ducts of conifer species. If known, the increased concentration and reduced complexity gained by selectively extracting these specific cell types may outweigh the increased difficulty of isolating them. Homogenization of the tissue in an appropriate solvent usually is sufficient to extract the terpenoids. If the terpenoids of interest are relatively volatile, then vapor-phase collection can reduce sample complexity; this can be done without the use of solvent in some instances. Recently, Tholl et al. (42) have reviewed the analytical methods of headspace sampling, solid phase micro-extraction (SPME), and the capture of plant volatiles onto absorbent media for subsequent analysis by gas chromatography.
Structural determination
Hanson (43) has published an excellent review of the methods and the strategies for rigorous structural determination of terpenoids. Routine survey-style analyses of plant terpenoids often are not so rigorous in structural assignment. Databases of mass spectra [such as Wiley (New York) and NIST (Gaithersburg, MD) MS databases] or databases that combine mass spectra and retention indices (44, 45) facilitate assignment of some commonly encountered plant terpenoids. Of course, these databases are only helpful if they include the specific terpenoids that are being analyzed and the likely alternatives. Sometimes, in addition to having similar mass-spectral fragmentation patterns, two terpenoids may share very similar retention indices on the 5% phenyl methylpolysiloxane GC column used for these databases, which makes structural assignment without additional information impossible. The availability of retention index information on at least two columns of different polarity increases the confidence in structural assignment. Such information is available for the more volatile terpenoids involved in flavors and fragrances (46), and insect semiochemicals (47). For nonvolatile terpenoids, LC-MS is often used, but there are no comparable databases of terpenoids for LC-MS as there are for GC-MS.
Stereochemistry often is an integral component to both the chemical structure and the biological function of plant terpenoids. For volatile terpenoids, chiral GC stationary phases (48) provide the enantiomeric separation for quantitative analysis, and, provided an authentic standard of known absolute configuration is available, elution comparison can establish absolute configuration of the unknown sample (49). Chiral phases for liquid chromatography can also be used to resolve enantiomers, both analytically and preparatively (50). Often, chiral synthesis is necessary to clearly establish the absolute configuration of an unknown terpenoid.
Molecular biology
The recent availability of plant genome sequences and the methods to clone homologous genes from different plant species using molecular biology approaches has provided the ability to identify the capacity of plants to produce additional terpenoids that may not have been detected in that specific plant species before. Rather than isolate, fractionate, and chemically identify a particular plant terpenoid profile, plant TPS or cytochromes P450 are cloned into a heterologous expression system, and the enzyme assay products of these recombinant enzymes with isoprenoid substrates are analyzed directly. This approach has proven useful in situations in which the terpenoids themselves may not be detectable in the plant under normal growing conditions, and the inducer or environmental conditions required for their production are not yet known (12).
Synthesis
Independent synthesis of the identified terpenoid is often required to confirm structural assignment (51) and to test the biological and/or pharmacological functions. In addition, biosynthetic studies often require the synthesis of putative precursors for functional characterization of enzymes. Terpenoid structures challenge chemists in many of the same ways that other natural products do. Their structural diversity (complicated by stereochemistry, carbocyclic skeletons, and often multiple functionalization) provides opportunities for synthetic organic chemists to develop new methodologies for synthesis. Recently, Maimone and Baran (52) have reviewed some synthetic challenges terpenoids present and the solutions employed.
Biochemical and metabolic engineering
Often, the original natural plant sources of medicinally or industrially important terpenoids cannot supply sufficient material for their demand. In some instances, such as Taxus for the production of Taxol (53), the original plant species is amenable to growth in plant cell culture for commercial production. For several plant terpenoids, an understanding of their biosynthetic pathway has advanced such that many, if not all, enzymes and genes involved have been identified and characterized. These developments provide the opportunity to engineer bacteria or yeast to produce these terpenoids de novo or from more readily available precursors in large-scale fermentations (54). In addition, the increased, reduced, or altered biosynthesis of a terpenoid in a plant can be engineered, which results in plant products with greater benefit for human use (55). The enhanced production of terpenoids both in planta and in microbial fermentations may provide a renewable alternative feedstock for petroleum-based industrial materials and fuels.
An active area of research is the directed evolution of TPS by site-directed mutagenesis of amino acids that are important in influencing product outcomes. These studies not only allow us to understand how the diversity of terpenoids evolved from gene duplication and neofunctionalization, but also allow us to modify multi-product TPS to favor one of the wild type products or to force the enzyme into producing unnatural products. Both techniques have use in the large-scale production of medicinally or industrially important terpenoids.
References
1. Conolly JD, Hill RA. Dictionary of Terpenoids. 1991. Chapman & Hall, London.
2. Gershenzon J, Dudareva N. The function of terpene natural products in the natural world. Nat. Chem. Biol. 2007; 3:408-414.
3. Keeling CI, Bohlmann J. Genes, enzymes and chemicals of terpenoid diversity in the constitutive and induced defence of conifers against insects and pathogens. New Phytol. 2006; 170:657-675.
4. Dudareva N, Pichersky E, Gershenzon J. Biochemistry of plant volatiles. Plant Physiol. 2004; 135:1893-1902.
5. Cane DE, ed. Isoprenoids, Including Carotenoids and Steroids. 1999. Elsevier, London.
6. Aubourg S, Lecharny A, Bohlmann J. Genomic analysis of the terpenoid synthase (AtTPS) gene family of Arabidopsis thaliana. Mol. Genet. Genomics 2002; 267:730-745.
7. Goff SA, Ricke D, Lan T-H, Presting G, Wang R, Dunn M, Glazebrook J, Sessions A, Oeller P, Varma H,. et al. A draft sequence of the rice genome (Oryza sativa L.ssp. japonica). Science 2002; 296:92-100.
8. Tuskan GA, Difazio S, Jansson S, Bohlmann J, Grigoriev I, Hellsten U, Putnam N, Ralph S, Rombauts S, Salamov A. et al. The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 2006; 313:1596-1604.
9. Jaillon O, Aury J-M, Noel B, Policriti A, Clepet C, Casagrande A, Choisne N, Aubourg S, Vitulo N, Jubin C. et al. The grapevine genome sequence suggests ancestral hexaploidization ini major angiosperm phyla. Nature 2007; 449:463-467.
10. Bohlmann J, Meyer-Gauen G, Croteau R. Plant terpenoid synthases: Molecular biology and phylogenetic analysis. Proc. Natl. Acad. Sci. U.S.A. 1998; 95:4126-4133.
11. Martin DM, Faldt J, Bohlmann J. Functional characterization of nine Norway spruce TPS genes and evolution of gymnosperm terpene synthases of the TPS-d subfamily. Plant Physiol. 2004; 135:1908-1927.
12. Ro D-K, Ehlting J, Keeling CI, Lin R, Mattheus N, Bohlmann J. Microarray expression profiling and functional characterization of AtTPS genes: Duplicated Arabidopsis thaliana sesquiterpene synthase genes At4g13280 and At4g13300 encode root-specific and wound-inducible (Z)-y-bisabolene synthases. Arch. Biochem. Biophys. 2006; 448:104-116.
13. Peters RJ. Uncovering the complex metabolic network underlying diterpenoid phytoalexin biosynthesis in rice and other cereal crop plants. Phytochemistry 2006; 67:2307-2317.
14. Martin DM, Bohlmann J. Identification of Vitis vinifera (-)-alpha-terpineol synthase by in silico screening of full-length cDNA ESTs and functional characterization of recombinant terpene synthase. Phytochemistry 2004; 65:1223-1229.
15. Croteau RB, Davis EM, Ringer KL, Wildung MR. (-)-Menthol biosynthesis and molecular genetics. Naturwissenschaften 2005; 92:562-577.
16. Ro D-K, Paradise EM, Ouellet M, Fisher KJ, Newman KL, Ndungu JM, Ho KA, Eachus RA, Ham TS, Kirby J. et al. Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 2006; 440:940-943.
17. Croteau R, Ketchum REB, Long RM, Kaspera R, Wildung MR. Taxol biosynthesis and molecular genetics. Phytochem. Rev. 2006; 5:75-97.
18. Keeling CI, Bohlmann J. Diterpene resin acids in conifers. Phytochemistry 2006; 67:2415-2423.
19. Lichtenthaler HK. The 1-deoxy-D-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plants. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1999; 50:47-65.
20. Takahashi S, Koyama T. Structure and function of cis-prenyl chain elongating enzymes. Chem. Rec. 2006; 6:194-205.
21. Miller B, Oschinski C, Zimmer W. First isolation of an isoprene synthase gene from poplar and successful expression of the gene in Escherichia coli. Planta. 2001; 213:483-487.
22. Sasaki K, Ohara K, Yazaki K. Gene expression and characterization of isoprene synthase from Populus alba. FEBS Letters 2005; 579:2514-2518.
23. Sharkey TD, Yeh S, Wiberley AE, Falbel TG, Gong D, Fernandez DE. Evolution of the isoprene biosynthetic pathway in kudzu. Plant Physiol. 2005; 137:700-712.
24. Guenther A, Karl T, Harley P, Wiedinmyer C, Palmer PI, Geron C. Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature). Atmos. Chem. Phys. Dis. 2006; 6:3181-3210.
25. Behnke K, Ehlting B, Teuber M, Bauerfeind M, Louis S, Hansch R, Polle A, Bohlmann J, Schnitzler JP, Transgenic, non-isoprene emitting poplars don’t like it hot. Plant J. 2007; 51:485-499.
26. Loivamaki M, Gilmer F, Fischbach RJ, Sorgel C, Bachl A, Walter A, Schnitzler JP. Arabidopsis, a model to study biological functions of isoprene emission? Plant Physiol. 2007; 144:1066-1078.
27. Fisher AJ, Baker BM, Greenberg JP, Fall R. Enzymatic synthesis of methylbutenol from dimethylallyl diphosphate in needles of Pinus sabiniana. Arch. Biochem. Biophys. 2000; 383:128-134.
28. Wise ML, Croteau R. Monoterpene biosynthesis. In: Isoprenoids, Including Carotenoids and Steroids. Cane DE, ed. 1999. Elsevier, London. pp. 97-153.
29. Hyatt DC, Youn B, Zhao Y, Santhamma B, Coates RM, Croteau RB, Kang C. Structure of limonene synthase, a simple model for terpenoid cyclase catalysis. Proc. Natl. Acad. Sci. U.S.A. 2007; 104:5360-5365.
30. Mahmoud SS, Croteau RB. Metabolic engineering of essential oil yield and composition in mint by altering expression of deoxyxylulose phosphate reductoisomerase and menthofuran synthase. Proc. Natl. Acad. Sci. U.S.A. 2001; 98:8915-8920.
31. Mahmoud SS, Croteau RB. Menthofuran regulates essential oil biosynthesis in peppermint by controlling a downstream monoterpene reductase. Proc. Natl. Acad. Sci. U.S.A. 2003; 100:14481-14486.
32. Bertea CM, Freije JR, van der Woude H, Verstappen FW, Perk L, Marquez V, De Kraker JW, Posthumus MA, Jansen BJ, de Groot A,. et al. Identification of intermediates and enzymes involved in the early steps of artemisinin biosynthesis in Artemisia annua. Planta Med. 2005; 71:40-47.
33. Mercke P, Bengtsson M, Bouwmeester HJ, Posthumus MA, Brodelius PE. Molecular cloning, expression, and characterization of amorpha-4,11-diene synthase, a key enzyme of artemisinin biosynthesis in Artemisia annua L. Arch. Biochem. Biophys. 2000; 381:173-180.
34. Wallaart TE, Bouwmeester HJ, Hille J, Poppinga L, Maijers NC. Amorpha-4,11-diene synthase: cloning and functional expression of a key enzyme in the biosynthetic pathway of the novel antimalarial drug artemisinin. Planta 2001; 212:460-465.
35. Chang MCY, Eachus RA, Trieu W, Ro D-K, Keasling JD. Engineering Escherichia coli for production of functionalized terpenoids using plant P450s Nat. Chem. Biol. 2007; 3:274-277.
36. Shiba Y, Paradisea EM, Kirbya J, Ro D-K Keasling JD. Engineering of the pyruvate dehydrogenase bypass in Saccharomyces cerevisiae for high-level production of isoprenoids. Metabol. Eng. 2007; 9:160-168.
37. Wani MC, Taylor HC, Wall ME, Coggan P, McPhail AT. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J. Am. Chem. Soc. 1971; 93:2325-2327.
38. Xiao Z, Itokawa H, Lee K-H. Total synthesis of taxoids. In: Taxus-The genus Taxus. Itokawa H, Lee K-H, eds. 2003. Taylor & Francis, London. pp. 245-297.
39. Walker K, Croteau R. Taxol biosynthetic genes. Phytochemistry 2001; 58:1-7.
40. Langenheim JH. Plant Resins: Chemistry, Evolution, Ecology and Ethnobotany. 2003. Timber Press, Portland, OR.
41. Ro D-K, Arimura G, Lau SY, Piers E, Bohlmann J. Loblolly pine abietadienol/abietadienal oxidase PtAO (CYP720B1) is a multifunctional, multisubstrate cytochrome P450 monooxygenase. Proc. Natl. Acad. Sci. U.S.A. 2005; 102:8060-8065.
42. Tholl D, Boland W, Hansel A, Loreto F, Rose US, Schnitzler JP. Practical approaches to plant volatile analysis. Plant J. 2006; 45:540-560.
43. Hanson Jr. The development of strategies for terpenoid structure determination. Nat. Prod. Rep. 2001; 18:607-617.
44. Adams RP. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry. 2007. Allured Publishing Co., Carol Stream, IL.
45. Hochmuth DH. MassFinder 3. 2007. Hamburg, Germany.
46. Acree T, Arn H. Flavornet and Human Odor Space. http://www.flavornet.org.
47. El-Sayed AM. The Pherobase: Database of Insect Pheromones and Semiochemicals. http://www.pherobase.com.
48. Shurig V. Separation of enantiomers by gas chromatography. J. Chrom. A 2001; 906:275-299.
49. Konig WA, Hochmuth DH. Enantioselective gas chromatography in flavor and fragrance analysis: strategies for the identification of known and unknown plant volatiles. J. Chrom. Sci. 2004 42:423-439.
50. Keeling CI, Ngo HT, Benusic KD, Slessor KN. Preparative chiral liquid chromatography for enantiomeric separation of pheromones. J. Chem. Ecol. 2001; 27:487-497.
51. Nicolaou KC, Snyder SA. Chasing molecules that were never there: misassigned natural products and the role of chemical synthesis in modern structure elucidation. Angew. Chem. Int. Ed. Engl. 2005; 44:1012-1044.
52. Maimone TJ, Baran PS. Modern synthetic efforts toward biologically active terpenes. Nat. Chem. Biol. 2007; 3(7):396-407.
53. Tabata H. Paclitaxel production by plant-cell-culture technology. Adv. Biochem. Engin. Biotechnol. 2004; 7:1-23.
54. Withers ST, Keasling JD. Biosynthesis and engineering of isoprenoid small molecules. Appl. Microbiol. Biotechnol. 2007; 73:980-990.
55. Townsend BJ, Llewellyn DJ. Reduced terpene levels in cottonseed add food to fiber. TRENDS in Biotech. 2007; 25:239-241.
See Also
Chemical Ecology: an Overview
Chiral Separations of Biomolecules
Design and Engineer Natural Products, Methods to
Isoprenoids
Natural Products: an Overview
Natural Products as Anticancer Agents
Natural Products Discovery, Molecular Biological Approaches to
Terpenoid Biosynthesis