CHEMICAL BIOLOGY
Extracellular Lipid Signals
Jerold Chun, Helen L. Dorris Institute for Neurological and Psychiatric Disorder, Department of Molecular Biology, The Scripps Research Institute, La Jolla, California
doi: 10.1002/9780470048672.wecb289
Lipid molecules can act as extracellular signals. They form a large and expanding class of influences on cellular and organismal homeostasis, which affect all organ systems and participate in many disease processes. Prominent members of this class include eicosanoids (e.g., leukotrienes or prostanoids like prostaglandins, protacyclins, and thromboxane), lysophospholipids (e.g., lysophosphatidic acid and sphingosine 1-phosphate), endocannabinoids (e.g., anandamide and 2-arachidonoylglycerol), ether lipids (e.g., platelet activating factor), and free fatty acids. These signals are in a dynamic steady state with structural phospholipids that make up the lipid membrane bilayer of all cells. Both distinct and shared enzymologies are involved in lipid signal biosynthesis and degradation. A dominant mechanistic theme for all bona fide extracellular lipid signals is their use of cognate 7-transmembrane domain G protein-coupled receptors. Activation of these receptors causes myriad physiologic and pathophysiologic effects that encompass most aspects of cell biology and physiology. Studies on extracellular lipid signals have led to both mechanistic understanding and successful creation of useful medicines.
Extracellular lipid signals are small fat molecules that produce myriad cell signaling effects upon exposure to the extracellular surface of cells. The dominant mechanism for these effects is the activation of specific cognate cell surface receptors, called G protein-coupled receptors (GPCRs), which are characterized by a predicted 7-transmembrane-spanning structure and which activate most known, intracellular heterotrimeric G proteins. These G proteins in turn activate a wide range of downstream signaling pathways, with the actual physiologic responses dependent on receptor expression patterns and involved cell types. All of these lipid mediators are linked, directly or indirectly, to membrane phospholipids that can be thought of as a dynamic repository of signaling lipid precursors. A diverse, yet often overlapping, enzymatic machinery exists for both the biosynthesis and the degradation of these signals. Compared with peptidergic factors, lipid mediators are often one tenth or less in mass, and they have brief half-lives, although binding to carrier proteins can substantially increase their stability. Historically, the understanding of these factors emerged in two phases. The first phase, which began about 70 years ago and continues today, was the important biochemical identification of lipids that showed bioactive properties in animals, and could be chemically isolated and structurally analyzed. This phase was marked by many mechanistic hypotheses to explain the observed bioactivities, including nonreceptor hypotheses. Tools for the identification of lipid components include classic thin layer chromatography, liquid chromatography, mass spectroscopy, as well as the use of bioassays and isotope labeling.
The second phase began with the advent of molecular cloning of cell-surface lipid receptors that commenced in the late 1980s/early 1990s and that allowed rigorous assessment of the existence, pharmacology, and functional roles for extracellular lipid signaling. It included the use of genetics to create mouse mutants that allowed additional analyses of both receptor identity and essential physiologic or pathophysiologic roles. Most receptor-ligand interactions occur at nanomolar affinities, with the exception of free fatty acids that interact at micromolar concentrations. A mainstay of these studies is the use of modern cell and molecular biology leading to the creation of mutant cell lines and animals. A standard in all fields is the use of receptor overexpression or heterologous expression, particularly combined with the use of receptor-null cells and tissues created by targeted deletion of one or more of the receptors in question. Interestingly, deleting individual lipid receptors produces a variety of phenotypes, which demonstrates both the possible redundancy of some receptors as well as the unique functions that cannot be rescued by the large number of lipid GPCRs. These reagents have led to the development of specific chemical agonists and antagonists, including some that have become therapeutic drugs. Intracellular roles for some of these mediators have been documented; however, this aspect of lipid signaling will not be considered further in this article. Major extracellular lipid signals are considered next, in alphabetical order. Representing literally tens of thousands of primary references over the past 70 years, the reference list is necessarily incomplete, and the reader is referred to the recent primary literature for more in-depth data and discussion in this rapidly evolving and expanding field.
Eicosanoids
Eicosanoids, also referred to as icosanoids, are so named because of the 20-carbon constituency that identifies this class of oxygenated lipid molecules. A primary synthetic pathway for these molecules involves the phospholipase-mediated cleavage of a membrane phospholipid to produce arachidonic acid [(all-Z)-ikosa-5,8,11,14-tetraenoic acid]. From this biologically essential intermediate fatty acid, two major subclasses of eicosanoids can be produced: 1) leukotrienes, via the action of lipooxygenases, and 2) prostanoids, via the action of cyclooxygenases (COX-1 and COX-2). Examples of chemical structures for a leukotriene (Fig. 1a) and three types of prostanoids (Fig. 1b-d) underscore their shared arachidonate origin.
Leukotrienes
Leukotrienes were identified in the 1930s as a bioactive substance released by smooth muscle in the lung after an antigenic challenge. Their current name reflects their well-recognized role in leukocyte (white blood cell) activities and the three prominent adjacent double-bonds in their chemical structure (Fig. 1a). There are multiple leukotriene species, including leukotriene (LT) A4, B4, C4, D4, E4, and F4. The C-, D-, and E4 species are named cysteinyl leukotrienes because of the presence of the amino acid cysteine in their structure or slow-reacting substance of anaphylaxis (SRS-As) recognizing their shared biologic activity. Relatively low levels of leukotrienes are found in resting cells; cell activation through calcium signaling stimulates the activity of lipoxygenases to produce leukotrienes, primarily as part of an immune response (1). Four cognate GPCRs bind leukotrienes with high affinity: LTB4/BLT1, BLT2, CysLTi, and CysLT2, all of which couple to Gq/11, which can increase intracellular calcium levels. The BLT receptors also couple to Gi/0, which can trigger a decrease in cAMP levels. Low level expression of the receptors in many tissues has been reported; however, their primary physiologic function is attributed to high expression on granulocytes, activated lymphocytes, macrophages, endothelium, and smooth muscle (2). Leukotrienes are important lipid inflammatory mediators, stimulating the release of anti-inflammatory or proinflammatory molecules, promoting neutrophil migration and adherence, and causing the contraction of bronchial smooth muscle (3). This latter role has been linked to bronchial disorders (4), and many treatments for asthma and allergic rhinitis are leukotriene receptor antagonists, including montelukast (Singulair; Merck, Whitehouse Station, NJ), pranlukast (Onon; Ono Pharmaceuticals, Osaka, Japan, and Schering-Plough, Kenilworth, NJ), and zafirlukast (Accolate; AstraZeneca Pharmaceuticals, Wilmington, DE) (5).
Prostanoids
These lipid factors were first identified in the 1930s as bioactive components of semen. Their name is derived from the original belief that they were secreted from the prostate gland. Their unique chemical structures are characterized by a trans double bond at C13-14, a hydroxyl group at C15, and a cyclic carbon ring (Fig. 1b-d). Three subclasses of prostanoids are defined by their cyclic ring configuration: 1) the well-known prostaglandins which have a cyclopentane ring (Fig. 1b); 2) prostacyclins that are cyclized (Fig. 1c); and 3) thromboxanes that contain a six-residue ring (Fig. 1d). Naturally occurring prostaglandins include PGD2, PGE2, PGF2a, and PGH2, of which the latter is a common precursor in the synthetic pathway of many protanoids. PGI2 and TXA2 are the most common naturally occurring prostacyclin and thromboxane forms, respectively. Like leukotrienes, prostanoids are important lipid mediators in the inflammatory response and are not synthesized unless their source cells are activated (3). Because of their short half-life, these lipids are primarily paracrine and autocrine extracellular effectors, which signal through at least nine cognate GPCRs (6). Alternatively spliced isoforms of these receptors also exist, with varying affinities for individual prostanoids. Signaling through prostanoid GPCRs encompass at least three classes of heterotrimeric G proteins: Gs(receptors DP, IP, EP2, EP4), Gq/11 (receptors FP, TP, EP1), and Gi/0 (receptor EP3) (7).
Prostanoids are most prominently recognized as proinflammatory factors that are vasoactive, cause blood coagulation, and produce associated symptoms of fever and pain (8). With the development of mouse receptor-null mutants, an increasingly wide range of biologic effects mediated by prostanoids is beginning to be appreciated (9). Complex physiologies associated with the numerous receptors are still being determined and point to kidney, cardiovascular, gastrointestinal, and reproductive system functions (6). Inhibitors of the rate-limiting enzymes in prostanoid synthesis, COX1 and COX2, have been the basis for nonsteroidal anti-inflammatory drugs, most prominently the nonselective COX inhibitors such as aspirin, ibuprofen, and naproxen, as well as the selective COX-2 inhibitors celecoxib (Celebrex; Pfizer, New York, NY) and the recently withdrawn drug, roficoxib (Vioxx; Merck).

Figure 1. Chemical structures of representative eicosanoid molecules: (a) leukotriene, (b) prostaglandin, (c) prostacyclin, and (d) thromboxane. Common names, chemical formulas, and molecular weights (g/mol) are shown.
Lipoxins
A third class of eicosanoids, the lipoxins (LX), has recently been defined based on its unique anti-inflammatory signaling. LXs derive their name from their generation via lipoxygenase interactions, requiring at least two lipoxygenases in their biosynthesis. Unlike the LTs and prostanoids, which are proinflammatory, LXs reduce inflammation through multiple pathways, including binding LX-specific GPCRs (e.g., LXA4), inhibiting LT-mediated inflammation through competitive inhibition of cysLT1 and cysLT2 receptors, and directly interacting with intracellular targets, e.g., transcription factors (10). Recently, the carbon-15 R epimers of LXs, termed aspirin-triggered lipoxins (ATLs), have been identified as a novel component in the anti-inflammatory mechanism of action for aspirin (10). Production of LXs has been observed in many human tissues, and reduced production is implicated in several inflammation-related diseases, such as arthritis, atherosclerosis, liver disease, cystic fibrosis, and periodontitis (11).
Endocannabinoids
The psychotropic properties of the plant-derived chemical compounds in marijuana, most prominently A9-THC (delta-9-tetrahydrocannabinol), have been known since the mid-1960s. The mechanism through which these exogenous compounds act was a subject of controversy until the early 1990s when the first cannabinoid receptors, referred to as CB1 and CB2, were cloned (discussed below). This quickly led to the identification of endogenous cannabinoid receptor ligands, “endocannabinoids,” which fall into two major classes: the N-acyl ethanolamines (NAEs) and monoacylglycerols (MAGs). The NAE, “anan- damide” or N-arachidonoylethanolamide (Fig. 2a), is a high affinity agonist for both CB1 and CB2 and can produce all of the known neurobehavioral effects of marijuana. Anandamide is synthesized through a two-step process from the membrane phospholipid, phosphoethanolamine, and is created in a one-to-one ratio with phosphatidic acid, which is another important lipid signal (12). The second major endocannabinoid is 2-arachidonoylglycerol (2-AG), a MAG with lower affinity for both cannabinoid receptors and many distinct physiologic effects. It has an esterfied arachidonic acid acyl chain in the sn-2 position of the glycerol backbone (Fig. 2b); thus, 2-AG can also be a precursor for some prostanoids (13). Many enzymes are involved in the biosynthesis and catabolism of endocannabinoids, regulating their diverse physiologic functions (14). One important catablolic enzyme is the fatty acid amide hydroxylase (FAAH) and inhibitors of FAAH are currently being investigated for their analgesic properties (15).
The cannabinoid receptors are GPCRs that are known to couple to Gi/0, although other coupling may exist in different cell types (16, 17). CB1 and CB2 have distinct expression profiles, with the former primarily expressed in the nervous system and the latter expressed in immune cells and in intestine (18). Within the central nervous system (CNS), CB1 receptors are widely expressed, particularly in the basal ganglia, the hypothalamus, and limbic areas, which suggests roles in mood, endocrine regulation, and memory (19). Indeed, genetic deletion of CB1 in mice is associated with enhanced short-term memory and weight loss (20). CB1 is also expressed in the male and female reproductive systems, although the role of cannabinoid signaling in reproduction is still unknown.
Cannabinoid receptors have been of great therapeutic interest in a variety of conditions, in particular obesity and neuropathic pain (21). Several medicines targeting these receptors are now on the market, including nabilone (Cesamet; Valeant Pharmaceuticals International, Costa Mesa, CA), dranabinol (Marinol; Unimed Pharmaceuticals, Deerfield, IL), and the recently released rimonabant (Acomplia; Sanofi-Aventis, Bridgewater, NJ), a selective CB1 antagonist for obesity management.

Figure 2. Endocannabinoid chemical structures: (a) anandamide and (b) 2-arachidonoylglycerol. Chemical formulas and molecular weights (g/mol) are given.
Ether Lipids
Platelet-activating factor (PAF, 1-O-alkyl-2-acetyl-sn-glycero-3-phosphocholine) was the first extracellular lipid signal for which a cloned receptor was identified (22). A hallmark of the molecule is an alkyl ether at the sn-1 position of the glycerolphosphate backbone (Fig. 3), in contrast with ester-linked molecules such as the lysophospholipids. In addition, the inactivation for PAF by platelet activating factor acetylhydrolase (PAFAH, a specialized phospholipase A2) is highly specific for the short-chain acyl group off the sn-2 carbon (23). PAF is the best characterized of the ether lipid signaling molecules, with potent activity on platelets and other immune cells, as its name implies (3). However, since its initial characterization in the immune system, a broad range of additional physiologic or pathophysiologic processes have been linked to this lipid molecule, including reproduction, neural migration, and blood circulation (24). Its receptor is a GPCR linked to numerous intracellular signaling pathways. Although the PAF receptor has widespread expression in many tissues, the subtle phenotype of the null-mutant mouse suggests the involvement of compensatory signaling mechanisms that involve other as yet unidentified receptors or redundancy of signaling mechanisms. In-depth studies using both transgenic and receptor-null models have demonstrated influences on allergic response, inflammation, infection, as well as osteoporosis (25), reproduction, wound healing, and subtle CNS functions (26).
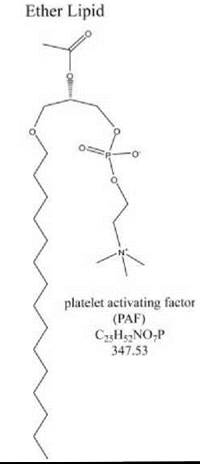
Figure 3. The chemical structure of a well-known ether lipid, platelet activating factor, showing stereochemistry. Chemical formula and molecular weight (g/mol) are included.
Free Fatty Acids (FFAs)
The most recent class of molecules to be characterized as extracellular lipid signals are free fatty acids (Fig. 4). Fatty acids are essential residues of biologic phospholipids, and they can exist in numerous permutations of length and saturation. FFAs are “free” in that they are not linked to other molecules such as the glycerophosphate backbone found in cell membranes and have important roles in both energy production and cell signaling. They can be produced through several metabolic processes such as cleavage of membrane phospholipids by several different phospholipases (e.g., phospholipase A). Both short-chain and long-chain FFAs of varying carbon lengths and saturation have demonstrated biologic activity consistent with being lipid signals. Supporting this interpretation, recent receptor de-orphaning research has identified a set of four GPCRs—GPR40, 41, 43, and 120—that mediate a response to a range of FFA forms (27-29). These GPCRs couple to Gq/11 resulting in an increase in intracellular calcium (coupling has not been reported for GPR120, although it can alter calcium levels consistent with a Gq/11 coupling). An important distinction to be made between FFAs and other extracellular lipid signals is that, unlike the other receptor-ligand interactions considered in this entry, these FFA receptors respond only to microM concentrations, and do so with a relative lack of selectivity for specific FFA forms. This raises the possibility that other endogenous ligands with higher affinities might exist, although this remains to be determined.
The FFA receptors serve as sensors for levels of FFAs that allow homeostatic metabolic mechanisms to be activated. Expression is primarily in the pancreas and gut (GPR40, -41, -120), which is consistent with a role in metabolic functions. For example, GPR120 increases circulating blood insulin levels indirectly through the release of glucagon-like peptide 1 (GLP1) (27). Other receptor expression patterns in adipose tissue (GPR 41), brain (GPR40), and lymphocytes (GPR43) have also been reported (28, 29). However, their functional significance is not yet known. Data from receptor-null mutants have not yet been reported.

Figure 4. Examples of short-chain and long-chain free fatty acids are shown: (a) 4-carbon butyric acid and (b) 16-carbon palmitic acid. Chemical names, formulas, and molecular weights (g/mol) are given.
Lysophospholipids
Representing one the simplest forms of phospholipids, these molecules are characterized by a phosphate headgroup attached to a glycerol or sphingoid 3 carbon backbone with acyl chains that can vary with respect to length and saturation. The two best-studied forms of lysophospholipids are lysophosphatidic acid (LPA) and sphingosine 1-phosphate (S1P) (Fig. 5). Bioactive forms of LPA are numerous and include 1-oleyl LPA (18 carbons, 1 double bond acyl chain at the sn-1 position), as well as forms with acyl chains at the sn-2 position. LPA can be produced by the action of PLA2 on phosphatidic acid, as well as through the action of autotaxin, a lysophopholipase D enzyme that produces LPA from LPC (30). By contrast, S1P is produced by the action of sphingosine kinase 1 or 2 on sphingosine resulting in the phosphorylated bioactive lipid (31). Lysophospholipids can be degraded by multiple lipid phospholipases, lipid phosphate phosphatases, and/or lyases. Other lysophospholipids, such as lysophosphatidylcholine (LPC), lysophosphatidylserine (LPS), and sphingosylphosphorylcholine (SPC), may have biologic activity as extracellular signaling lipids; however, bona fide signaling via defined receptors remains unclear. Many cell types can produce most forms of lysophospholipids, but it is often not possible to distinguish the signaling pools versus structural or other nonsignaling pools of these lipids within a cell.
To date, five GPCRs have been identified for LPA and five for S1P: LPA1-5 and S1P1-5. Although most of these receptors share a high degree of homology within and even between ligand classes, lower homology receptors for both classes have been identified, particularly two recently reported LPA receptors, LPA4 and LPA5 (32-34). A wide range of G proteins are activated by these receptors, including Gi/0, Gq/11, G12/13, and Gs. As a group, these receptors show widespread expression during development and postnatal life, and they have been documented across phylogeny, from humans through fish. Clear cellular effects include cell survival, actin cytoskeletal alteration and cell shape changes, proliferation, electrophysiologic changes, and transcriptional activation, among others (35). These effects in turn contribute to a rich and complex physiology and pathophysiology that has been prominently revealed by receptor-null animals, as well as other approaches (36). Loss of LPA receptors can affect normal development, craniofacial formation, bleeding, CNS development and function, initiation of neuropathic pain, and normal fertility via uterine implantation. S1P receptor deletion can affect embryonic survival (S1P1), vascular formation, immunologic lymphocyte egress, cardiovascular function, and fertility (37).
Lysophospholipid receptors provide an attractive therapeutic target for multiple human disorders, including neuropathic pain, infertility, and multiple sclerosis, as well as graft rejection after transplantation (38). Underscoring this point, the experimental drug FTY720 is currently in phase III clinical trials for multiple sclerosis and kidney transplantation (39). This compound, when phosphorylated, acts as a nonselective agonist at four of five S1P receptors inducing lymphocyte sequestration and immunosuppression. Additional examination of lipid receptor mutants, as well as the identification of new receptors, will expand the biologic and therapeutic importance of these extracellular lipid signals.

Figure 5. Two representative lysophospholipid chemical structures showing stereochemistry. Molecules can exist with variable acyl chain saturation. Common names, chemical formulas, and molecular weights (g/mol) are given.
Acknowledgements
Thank you to Christine Paczkowski and Danielle Letourneau for critical reading of this manuscript. The author gratefully acknowledges NIH support from NIMH, NINDS, NIDA, and NICHD.
References
1. Shimizu T, Wolfe LS. Arachidonic acid cascade and signal transduction. J. Neurochem. 1990; 55:1-15.
2. Yokomizo T, Izumi T, Chang K, Takuwa Y, Shimizu T. A G protein-coupled receptor for leukotriene B4 that mediates chemo- taxis. Nature 1997; 387:620-624.
3. Christie PE, Henderson WR Jr. Lipid inflammatory mediators: leukotrienes, prostaglandins, platelet-activating factor. Clin. Allergy Immunol. 2002; 16:233-254.
4. Peters-Golden M, Gleason MM, Togias A. Cysteinyl leukotrienes: multi-functional mediators in allergic rhinitis. Clin. Exp. Allergy 2006; 36:689-703.
5. Riccioni G, Di Ilio C, Conti P, Theoharides TC, D’Orazio N. Advances in therapy with antileukotriene drugs. Ann. Clin. Lab. Sci. 2004; 34:379-387.
6. Narumiya S, FitzGerald GA. Genetic and pharmacological analysis of prostanoid receptor function. J. Clin. Invest. 2001; 108:25-30.
7. Bos CL, Richel DJ, Ritsema T, Peppelenbosch MP, Versteeg HH. Prostanoids and prostanoid receptors in signal transduction. Int. J. Biochem. Cell. Biol. 2004; 36:1187-1205.
8. Tilley SL, Coffman TM, Koller BH. Mixed messages: modulation of inflammation and immune responses by prostaglandins and thromboxanes. J. Clin. Invest. 2001; 108:15-23.
9. Kobayashi T, Narumiya S. Function of prostanoid receptors: studies on knockout mice. Prostaglandins Other Lipid Mediat. 2002; 68-69:557-573.
10. Serhan CN, Levy BD, Clish CB, Gronert K, Chiang N. Lipoxins, aspirin-triggered 15-epi-lipoxin stable analogs and their receptors in anti-inflammation: a window for therapeutic opportunity. Ernst Schering Res. Found Workshop 2000: 143-185.
11. Serhan CN. Lipoxins and aspirin-triggered 15-epi-lipoxins are the first lipid mediators of endogenous anti-inflammation and resolution. Prostaglandins Leukot. Essent. Fatty Acids 2005; 73:141-162.
12. Bisogno T, Delton-Vandenbroucke I, Milone A, Lagarde M, Di Marzo V. Biosynthesis and inactivation of N-arachidonoylethanolamine anandamide. and N-docosahexaenoylethanolamine in bovine retina. Arch Biochem. Biophys. 1999; 370:300-307.
13. Di Marzo V, Bisogno T, De Petrocellis L, Melck D, Orlando P, Wagner JA, Kunos G. Biosynthesis and inactivation of the endocannabinoid 2-arachidonoylglycerol in circulating and tumoral macrophages. Eur. J. Biochem. 1999; 264:258-267.
14. Kogan NM, Mechoulam R. The chemistry of endocannabinoids. J. Endocrinol. Invest. 2006; 29:3-14.
15. Cravatt BF, Lichtman AH. The endogenous cannabinoid system and its role in nociceptive behavior. J. Neurobiol. 2004; 61:149-160.
16. Howlett AC. The cannabinoid receptors. Prostaglandins Other Lipid Mediat. 2002; 68-69:619-631.
17. Ishii I, Chun J. Anandamide-induced neuroblastoma cell rounding viathe CB1 cannabinoid receptors. Neuroreport. 2002; 13:593-596.
18. Izzo AA. Cannabinoids and intestinal motility: welcome to CB2 receptors. Br. J. Pharmacol. 2004; 142:1201-1202.
19. Grotenhermen F. Pharmacology of cannabinoids. Neuro. Endocrinol. Lett. 2004; 25:14-23.
20. Valverde O, Karsak M, Zimmer A. Analysis of the endocannabi- noid system by using CB1 cannabinoid receptor knockout mice. Handb. Exp. Pharmacol. 2005: 117-145.
21. Makriyannis A, Mechoulam R, Piomelli D. Therapeutic opportunities through modulation of the endocannabinoid system. Neuropharmacology 2005; 48:1068-1071.
22. Honda Z, Nakamura M, Miki I, Minami M, Watanabe T, Seyama Y, Okado H, Toh H, Ito K, Miyamoto T, et al. Cloning by functional expression of platelet-activating factor receptor from guinea-pig lung. Nature 1991; 349:342-346.
23. Prescott SM, Zimmerman GA, Stafforini DM, McIntyre TM. Platelet-activating factor and related lipid mediators. Annu. Rev. Biochem. 2000; 69:419-445.
24. Stafforini DM, McIntyre TM, Zimmerman GA, Prescott SM. Platelet-activating factor, a pleiotrophic mediator of physiological and pathological processes. Crit. Rev. Clin. Lab. Sci. 2003; 40: 643-672.
25. Hikiji H, Ishii S, Shindou H, Takato T, Shimizu T. Absence of platelet-activating factor receptor protects mice from osteoporosis following ovariectomy. J. Clin. Invest. 2004; 114:85-93.
26. Ishii S, Shimizu T. Platelet-activating factor PAF. receptor and genetically engineered PAF receptor mutant mice. Prog. Lipid Res. 2000; 39:41-82.
27. Hirasawa A, Tsumaya K, Awaji T, Katsuma S, Adachi T, Yamada M, Sugimoto Y, Miyazaki S, Tsujimoto G. Free fatty acids regulate gut incretin glucagon-like peptide-1 secretion through GPR120. Nat. Med. 2005; 11:90-94.
28. Brown AJ, Goldsworthy SM, Barnes AA, Eilert MM, Tcheang L, Daniels D, Muir AI, Wigglesworth MJ, Kinghorn I, Fraser NJ, Pike NB, Strum JC, Steplewski KM, Murdock PR, Holder JC, Marshall FH, Szekeres PG, Wilson S, Ignar DM, Foord SM, Wise A, Dowell SJ. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003; 278:11312-11319.
29. Briscoe CP, Tadayyon M, Andrews JL, Benson WG, Chambers JK, Eilert MM, Ellis C, Elshourbagy NA, Goetz AS, Minnick DT, Murdock PR, Sauls HR Jr, Shabon U, Spinage LD, Strum JC, Szekeres PG, Tan KB, Way JM, Ignar DM, Wilson S, Muir AI. The orphan G protein-coupled receptor GPR40 is activated by medium and long chain fatty acids. J. Biol. Chem. 2003; 278:11303-11311.
30. Yokoyama K, Baker DL, Virag T, Liliom K, Byun HS, Tigyi G, Bittman R. Stereochemical properties of lysophosphatidic acid receptor activation and metabolism. Biochim. Biophys. Acta, 2002; 1582:295-308.
31. Le Stunff H, Milstien S, Spiegel S. Generation and metabolism of bioactive sphingosine-1-phosphate. J. Cell. Biochem. 2004; 92:882-899.
32. Hla T. Sphingosine 1-phosphate receptors. Prostaglandins Other Lipid Mediat. 2001; 64:135-142.
33. Noguchi K, Ishii S, Shimizu T. Identification of p2y9/GPR23 as a novel G protein-coupled receptor for lysophosphatidic acid, structurally distant from the Edg family. J. Biol. Chem. 2003; 278: 25600-25606.
34. Lee CW, Rivera R, Gardell S, Dubin AE, Chun J. GPR92 as a new G12/13.- and Gq.-coupled lysophosphatidic acid receptor that increases cAMP, LPA5. J. Biol. Chem. 2006; 281:23589-23597.
35. Ishii I, Fukushima N, Ye X, Chun J. Lysophospholipid receptors: signaling and biology. Annu. Rev. Biochem. 2004; 73:321-354.
36. Anliker B, Chun J. Cell surface receptors in lysophospholipid signaling. Semin. Cell Dev. Biol. 2004; 15:457-465.
37. Kono M, Mi Y, Liu Y, Sasaki T, Allende ML, Wu YP, Yamashita T, Proia RL. The sphingosine-1-phosphate receptors S1P1, S1P2, and S1P3 function coordinately during embryonic angiogenesis. J. Biol. Chem. 2004; 279:29367-29373.
38. Gardell SE, Dubi AE, Chun J. Emerging medicinal roles for lysophospholipid signaling. Trends Mol. Med. 2006; 12:65-75.
39. Chun J, Rosen H. Lysophospholipid receptors as potential drug targets in tissue transplantation and autoimmune diseases. Curr. Pharm. Des. 2006; 12:161-171.
Further Reading
Hanahan D. A Guide to Phospholipid Chemistry. Academic Press, 1998.
Fukushima, N, Ishii, I, Contos, JJ, Weiner, JA, Chun, J. Lysophospholipid receptors. Annu. Rev. Pharmacol. Toxicol. 2001; 41:507-34.
Moolenaar, WH, van Meeteren, LA, Giepmans, BN. The ins and outs of lysophosphatidic acid signaling. Bioessays. 2004; 26:870-81.
Hla, T. Genomic insights into mediator lipidomics.. Prostaglandins Other Lipid Mediat. 2005; 77:197-209.
www.lipidlibrary.co.uk is a reference website with up-to-date information on the structure, function, and analysis of lipids.
www.xpharm.com is a database containing pharmacology reference information.