CHEMICAL BIOLOGY
Lipids, Chemical Diversity of
Irina A. Guschina and John L. Harwood, School of Biosciences, Cardiff University, Wales, United Kingdom
doi: 10.1002/9780470048672.wecb292
Lipids represent some of the most complex biologic molecules, and their diversity is crucial for their cellular functions. The wide range of chemical and physical properties of different lipids determines a variety of roles for these compounds in biologic processes. According to their structures, lipids can be divided into two main groups: the nonpolar lipids (acylglycerols, sterols, free fatty acids, hydrocarbons, alcohols, wax, and steryl esters) and the polar lipids (phosphoglycerides, glycosylglycerides, and sphingolipids). Triacylglycerols act as energy stores and metabolic fuels. Polar lipids and sterols are important structural components of cell membranes where they may have many diverse functions. Arranged as bilayers, they establish permeability barriers for cells and organelles and provide a microenvironment for membrane-associated proteins as well as participate directly in metabolism and a multitude of membrane fusion events (1). Waxes as surface coverings are integral to water balance and protect organisms from noxious environmental conditions. In addition to a structural role, lipids serve as important intermediates in cell signaling pathways (e.g., sphingolipids, inositol lipids, phosphatidic acid, lysophospholipids, oxidative products) and play a role in mediating cellular responses to the environment (2). In this article, we will summarize our knowledge on the structural diversity of lipids that provide information on the various lipid classes and the fatty acids that occur naturally.
Glycerolipids
Acylglycerols (glycerides)
The basic structure of glycerolipids is a glycerol (propane-1,2,3-triol) backbone to which the hydrophobic acyl groups (fatty acids) are esterified. Monoacylglycerols and diacylglycerols are fatty acid monoesters and diesters of glycerol, respectively. These partial glycerides are important intermediates in metabolism. Moreover, sn-1,2-diacylglycerols are important in animal tissues where they function as signaling molecules in many cellular processes. Triacylglycerols are fatty acid triesters of glycerol. They are accumulated as storage products and are the major components of most natural fats and oils.
Glycerol-derived ethers
Alkyl ethers and alk-1-enyl ethers (or plasmalogens) are two general types of glycerol ethers. Mono- and dialkyl ethers have been reported to occur naturally. Examples include batyl alcohol (sn-1-O-octadecylglycerol) and chimyl alcohol (sn-1-O-hexa-decyl alcohol) as monoalkyl ethers and diphytanylglycerol (sn-2,3-di-O-tetramethylhexadecylglycerol) as a dialkyl ether. Although trialkyl ethers have not yet been found in nature, they have been synthesized with various combinations of saturated and unsaturated chains. In addition, monocyl and diacyl derivatives of alkyl ethers are reported for some natural sources.
Similar to alkyl forms, the mono- and dialk-1-enyl ethers of glycerol have been identified in natural samples, whereas no trialk-1-enyl ethers have been isolated so far. Acylated (mono- and diacyl derivatives) alk-1-enyl ethers (or neutral plasmalogens) have been also reported (2).
Glycerophospholipids
The structure of glycerophospholipids is characterized by a 1,2-diacyl-3-phospho-sn-glycerol, or phosphatidyl moiety, and a variable headgroup linked to the phosphate. Major phos- phoglycerides found in nature are phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylglycerol (PG), phosphatidylserine, phosphatidylinositol (PI), and diphosphatidylglycerol (or cardiolipin). The simplest phosphoglyceride, phosphatidic acid is a minor compound in most tissues.
PC, the trivial name “lecithin,” is a neutral or zwitterionic phospholipid (Fig. 1). It is the most abundant phospholipid in animal tissues and the main component of nonchloroplast membranes in plants, where it amounts to about 50% of the total lipids. In bacterial membranes, PC is found in small quantities. PC is a useful bilayer component, which has a cylindrical shape and a net neutral charge (1).
PE (trivial name “cephalin”) also has a net neutral charge (Fig. 1). PE is widespread and usually the second most abundant phospholipid in animal and plant membranes. It is also the main lipid component of microbial membranes. In animal tissues, phosphatidylethanolamine may exist in diacyl, alkylacyl and alkenylacyl forms. Moreover, animal phosphatidylethanolamine usually contains higher levels of arachidonic and docosahexaenoic acids in comparison with the other zwitterionic phospholipid, PC. The partly methylated derivatives of PE (phosphatidyl-N-methyl-ethanolamine, phosphatidyl-N-dimethyl-ethanolamine) are found in small amounts in many organisms. These derivatives are metabolic intermediates in the conversion of PE into PC. In some tissues, N-acylated derivatives of PE may be present in significant amounts. These compounds are involved into the endocannabinoid signalling system through release of N-acylethanolamines after phospholipase D action (3).
PG is negatively charged (Fig. 1). It is the major phospho- glyceride in photosynthetic tissues and many bacteria. In plants, PG is an only phospholipid present in significant quantities in the thylakoid membranes where it can comprise 10% of the total lipids with a high proportion (up to 70%) in the outer monolayer. An unusual fatty acid, ∆3-trans-hexadecenoic acid [16:1(3t)], is found in all eukaryotic photosynthetic organisms, especially esterifying the sn-2 position of PG (4). It is interesting to note that both the trans-configuration of a double bond and its A3 position are very unusual for natural fatty acids. PG-16:1(3t) is thought to play an essential role in photosynthetic membranes.
Although PG is only a minor phopholipid in animal tissues (1-2% of total phospholipids), it can be a second abundant phospholipid in some lung surfactants at up to 11% of the total (5).
For bacteria, PG is present in almost all types. In Escherichia coli, for example, PG accounts for 20% of its membrane lipids. The diacyl form of PG is more common in many bacteria, but some may contain the alkylacyl- and alkenylacyl derivatives as predominant forms. Several prokaryotic species, including Salmonella typhimurium and E. coli, and parasitic protozoa, such as Trichomonas vaginalis and T. foetus, have been found to contain acylphosphatidylglycerol (1,2-diacyl-sn-glycero-3-phospho-(3'-acyl)-F-sn-glycerol) in appreciable amounts (6). For example, Corynebacterium amycolatum contained about 30% of this lipid with oleate as a dominant fatty acid on the head group of glycerol (7). A fully acylated phosphatidylglycerol (bis-phosphatidic acid or phosphatidyldiacylglycerol) and its plasmalogen analogs have been also identified in a marine bacterium MB 45. In some bacteria, an amino acid (lysine, ornithine, arginine, or alanine) may be attached to the 3'-hydroxyl of the base glycerol to form an O-aminoacylphosphatidylglycerol. Two other unusual derivatives of phosphatidylglycerol have been isolated from the primitive organisms, the Haloarchaea. They were identified as phosphatidylglycerol sulfate and phosphatidylglycerol phosphate methyl ester.
Diphosphatidylglycerol (trivial name cardiolipin) has a unique dimeric structure with four acyl groups and two negative charges (Fig. 1). It is common in bacteria, and it can be found in the inner mitochondrial membranes of eukaryotes (i.e., those membranes that generate an electrochemical potential for substrate transport and ATP synthesis).
Phosphatidylserine also has a net negative charge. It is a widespread but minor lipid in eukaryotes, accounting usually for less than 10% of the total phospholipids. Its greatest concentration has been noted for myelin from brain tissue. Phosphatidylserine is concentrated in the inner monolayer of the plasma membrane and the other cellular membranes (Fig. 1).
Phosphatidylinositol is another widespread and minor lipid with a negative charge. It is an important lipid, both as a membrane constituent and in various phosphorylated forms as key molecules in signaling processes in eukaryotes (Fig. 1). The phosphatidylinositol phosphates are found with phosphates attached at any position of the inositol ring. However, PI-4-phosphate, PI-4,5-bis phosphate, and their 3-phosphates are the most important. PI also forms part of the glycerophosphatidylinositol (GPI) anchor for certain surface proteins.

Figure 1. Phosphoglycerolipids.
The acyl structure diversity
Monoacyl derivatives of phosphoglycerides, lyso-derivatives, are present in small amounts in most tissues, but their occurrence in large levels usually indicates lipid degradation before or during lipid extraction. Only cereal grains contain lysophos- phatidylcholine and lysophosphatidylethanolamine as the major phospholipids (as starch inclusion compounds). Moreover, lysophospholipids, especially lysophosphatidic acid, are important signaling molecules (8).
Plasmalogens are monoacyl monoalk-1-enyl ether forms of phospholipids. The most common forms of plasmalogens are the choline, ethanolamine, and serine derivatives. They are present in most animal tissues, especially in the mammalian brain. Plasmalogens are structural components of membranes. It has been proposed that plasmalogens protect membranes against oxidative stress (9).
Platelet-activating factor or 1-alkyl-2-acetyl-sn-glycero-3-phosphocholine is an ether analog of phosphatidylcholine. This biologically active lipid is of great current interest. Initially, it was found to affect aggregation of platelets and to induce a hypertensive response at very low concentrations. Recent research has revealed its role as a mediator of inflammation and of other physiologic effects on many different types of cells.
Archaebacteria contain large amounts of unique lipids, which are based on 2,3-dialkyl-sn-glycerol backbones, of the opposite stereochemical configuration to that found in the two other primary kingdoms: eubacteria and eukaryotes (10). As another diversity, they may have several polar head groups, existing as both phospholipids and glycolipids as well as their sulfated forms, and the alkyl groups are usually phytanyl (10).
Glycosylglycerides
Glycosylglycerides are characterized by a 1,2-diacyl-sn-glycerol moiety with a monosaccharide or oligosaccharide attached at the sn-3 position of the glycerol backbone. In photosynthetic bacteria, cyanobacteria, algae, and higher plants, glycosylglycerides (glycolipids) are located predominantly in photosynthetic membranes. The major plastid lipids, galactosylglycerides, are neutral lipids. They contain one or two galactose molecules linked to the sn-3 position of the glycerol that corresponds to 1,2-diacyl-3-O-(β-D-galactopyranosyl)-sn-glycerol (or mono-galactosyldiacylglycerol, MGDG) and 1,2-diacyl-3-O-(α-D-galactopyranosyl-(1→6)-O-P-D-galactopyranosyl-sn-glycerol (or digalactosyldiacylglycerol, DGDG) (Fig. 2). MGDG and DGDG are present at 40-55% and 15-35% of the total lipids in thylakoid membranes, respectively. A characteristic feature of these lipids is a very high content of polyunsaturated fatty acids. In addition to these main galactolipids, trigalactosyldi- acylglycerols and tetragalactosyldiacylglycerols as well as other homologs with many different sugar combinations (e.g., mannose, glucose, rhamnose) have been reported for bacteria, algae, and higher plants (11, 12). For example, in eubacteria, glycosyldiacylglycerols with one to three glycosyl units linked to sn-1,2-diacylglycerol were most common, but others with up to five glycosyl units have been also found. Monoglucosyldi-acylglycerols and diglucosyldiacylglycerols with the diglucoside moiety that have an α-(1→2)linkage (as in kojibiose) and a galactosyldiacylglycerol have been isolated from some Streptococcus species. Monomannosyldiacylglycerols and dimannosyldiacylglycerols have been reported for Micrococcus luteus. In Bacillus megaterium, N-acetylgalactosamine has been found linked to a diacylglycerol. In glycophospholipids present in some bacteria, a glycerophosphate group is linked to a carbohydrate moiety (11).
Various glucoglycerolipids and galactoglycerolipids have been also isolated in small amounts from animal tissues (13). The galactoglycerolipids usually contain a single galactose residue that is linked through a β-glycosidic link between the C-1 of galactose and the C-3 of glycerol. The glucogly-cerolipids that have been reported may contain up to eight glucose residues. Moreover, alkylacyl and diacyl lipids as well as sulfated forms have been identified (13).
Another class of glycosylglycerides, which is present in appreciable amounts in both photosynthetic and in nonphotosynthetic species, is the plant sulfolipid sulfoquinovosyldiacylglycerol, or 1,2-diacyl-3-O-(6-deoxy-6-sulfo-α-D-glucopyranosyl)-sn-glycerol (SQDG) (Fig. 2). This lipid is unusual because of its sulfonic acid linkage. It consists of monoglycosyldiacylglycerol with a sulfonic acid in position 6 of the monosaccharide moiety. The sulfonoglucosidic moiety (6-deoxy-6-sulfono-glucoside) is described as sulfoquinovosyl. The sulfonic residue carries a full negative charge at physiologic pH. In the unicellular alga Chlamydomonas reinhardtii, an acylated derivative of this sulfolipid, 2-O-acyl-sulfoquinovosyldiacylglycerol has been found. Sulfoquinovosylmonogalactosylglycerol has been isolated from the marine red alga, Gracilaria verrucosa. For a review of algal lipids, see Reference 12.
In addition to sulfoquinovosyldiacylglycerol, the marine diatom Nitzschia alba has been found to contain a sulfonium analog of phosphatidylcholine, phosphatidylsulfocholine (14). This lipid has two methyl groups attached to the sulphur atom, and it substitutes completely for phosphatidylcholine in Nitzschia alba. In other marine diatoms and algae, both lipids have been found (15).
Several lipids conjugated to taurine (2-amino ethylsulfonic acid, H2NCH2CH2SO3H) have been reported for many organisms. So, membrane lipids of certain bacteria and algae contain taurine linked to diacylglucuronylglycerol. In a seawater bacterium Hyphomonas jannaschiana, which is unique because of an absence of phospholipids, a tauroglycolipid, 1,2-diacylglycerol-3-glucuronopyranosyl-sn-glycerol taurineamide was identified. An unusual glycolipid sulfate ester has been isolated from extremely halophilic bacteria (11), and a glycolipid sulfate, 2,3,6,6’-tetraacetyl-a,a-tetrahalose-2'-sulfate has been isolated from Mycobacterium tuberculosis (16). For a review on the isolation, characterization, and biochemistry of taurolipids, see Reference 17.

Figure 2. Glycoglycerolipids.
Betaine lipids
Betaine lipids contain a betaine moiety as a polar group linked to the sn-3 position of glycerol by an ether bond. No phosphorus or carbohydrate group exists in betaine lipids. Three types of betaine lipids have been identified: 1,2-diacylglyceryl-3-O-4'-(N,N,N-trimethyl)-homoserine (DGTS), 1,2-diacylglyceryl-3-O-2'-(hydroxymethyl)-(N,N,N-trimethyl)-β-alanine (DGTA), and 1,2-diacylglyceryl-3-O-carboxy-(hydroxymethyl)-choline (DGCC) (Fig. 3) (18). These betaine lipids are all zwitterionic at pH 7 because they have a positively charged trimethylammonium group and a negatively charged carboxyl group (Fig. 3). Betaine lipids are not found in higher plants, either gymnosperms or angiosperms, but they are distributed widely in photosynthetic bacteria, algae, ferns, bryophytes, lichens, some fungi, and protozoans. Based on an obvious structural similarity between betaine lipids and phosphatidylcholine and on their taxonomical distribution (namely, their reciprocal relationship in many species), it has been suggested that betaine lipids, especially DGTS, are more primitive lipids evolutionarily that, in the lower plants, play the same functions in membranes that PC does in higher plants and animals (19).

Figure 3. Betaine lipids.
Diol Lipids
Diol lipids have been reported only recently when techniques for their structure elucidation were developed. Small quantities of diol lipids, such as diacylpropane-1,3-diol, diesters of butane-1,3-diol, and butane-1,4-diol are found in mammalian and fish liver, mammalian adipose tissues, egg yolk, corn seed, and yeast (20). From Actynomyces olivaceus, mixed acyl- and alk-1-enyl derivatives of simple diols have been isolated (21). An acylated diol phospholipid has been produced by the yeast Lypomyces starkeyi when grown on propane-1,2-diol (22).
Sphingolipids
Sphingolipids are lipids that contain sphingosine (trans-D-erythro-1,3-dihydroxy-2-amino-4-octadecene) or a related amino alcohol. Sphingolipids are a group of very complex lipids with a range of diverse functions. It has recently become apparent that sphingolipids are involved in many human diseases including diabetes, cancers, neurologic syndromes, infections, and diseases of the cardiovascular and respiratory systems (23). Moreover, an increased interest in these molecules results from the involvement of sphingolipids in the segregation/compartmentalization of membranes by self-organizing functional lipid microdomains, known as lipid rafts. Sphingolipids are located exclusively in the outer (exoplasmic) leaflet of the plasma membrane bilayer. In contrast, raft glycerophospholipids largely prefer the inner (cytoplasmic) leaflet, and cholesterol is most likely abundant in both leaflets. The lipid raft model is based on the assumption that sphingomyelin and other sphingolipids together with cholesterol are not distributed evenly in the plasma membrane, but rather they assemble to specific microdomains or raft. They act to compartmentalize and to separate membrane proteins with different biochemical functions. It is believed that up to 50% of the plasma membrane may consist of such rafts, and rafts formation has been shown to be crucial in modulation of signaling events in cells (24).
Sphingophospholipids and glycosphingophospholipids
Sphingomyelin (or ceramide phosphorylcholine) is the most common lipid in this class, and is the phosphorylcholine ester of an N-acylsphingosine (or ceramide) (Fig. 4). It is a major lipid in certain membranes in animal tissues. Although it is usually lower in concentration than phosphatidylcholine, it can comprise about 50% of the lipids in certain tissues. Sphingomyelin replaces phosphatidylcholine in erythrocytes of most ruminant animals entirely. Sphingomyelin is probably absent from microorganisms and plants.
In the plasma of the newborn pig and infant, 3-O-acyl-D-erythro-sphingomyelin has been detected. In this lipid, position 3 of the sphingosine residue was esterified by an additional fatty acid.
In additional, lyso-sphingomyelin and sphingosine phospho- rylcholine have been reported to be present in tissues in small amounts. They are involved in some signaling processes in the cell in a manner similar to sphingosine-1-phosphate (25, 26). Sphingosine-1-phosphate has been found in insects, yeasts, and plants; its function as an important cellular messenger has been shown in many studies. It regulates calcium mobilization inside the cell as well as cell growth and proliferation in response to several stress factors.
Ceramide phosphorylinositol, or myo-inositol-(1-O)-phosphoryl-(O-1)-ceramide, the sphingolipid analog of phosphati-dylinositol, is a major component of sphingolipids in yeasts (e.g., Saccharomyces cerivisiae). In these organisms, two other related inositol-containing sphingophospholipids, mannosyl-inositolphosphorylceramide and mannosyldiinositolphosphoryl-ceramide, have been also found.
Ceramide phosphorylethanolamine, the sphingolipid analogue of phosphatidylethanolamine, has been reported for the first time in the housefly, Musca domestica. Shortly after, it was identified in the bacteria Bacteroides ruminicola and B. melanogenicus as well as in some protozoa, snails, marine bivalves, insects, chicken liver, and rat liver (27). In recent studies, this lipid has been isolated from three species of oomycete plant pathogens (28) and from several species of Sphingobacterium (29).
Ceramide phosphorylglycerols are known as membrane components in anaerobic bacteria of the genus Bacteroides and in the oral pathogen Porphyromonas gingivalis (30).
A sphingoid analog of phosphatidic acid, ceramide-1-phosphate, has recently emerged as potent bioactive agent. Recent studies defined new biologic functions for this lipid related to control of numerous aspects of cell physiology, which include cell survival and mammalian inflammatory responses (26).
The first glycolipids identified in some plants were glycolipids with a phytosphingosine and/or dehydrosphingosine backbone that contains both inositol and phosphate. They have been named “phytoglycosphingolipids” and more than 20 molecular forms have been identified with glucuronic acid, glucosamine, and many other carbohydrate moieties. The analysis of such compounds has been discussed thoroughly in (31).
From the bacterium Sphingobacterium spiritivorum, ceramide phosphorylmannose has been recently reported for the first time. Archaebacteria have been shown to contain various phosphoglycolipids (11).
Another type of glycosphingophospholipids are glycosphin-golipids that are phosphorylated even more (i.e., where the ceramide is linked directly to sugar moieties not via phosphate group). The earthworm, Pheretima hilgendorfi, has been shown to contain such lipids, namely cholinephosphoryl-6Galβ1-1Cer and cholinephosphoryl-6Galβ1-6Galβ1-1Cer. In addition, triglycosylsphingophospholipids with either a terminal mannose or a galactose linked to phosphorylcholine were also present in this species. Some filamentous fungi contain glycosphin- gophospholipids (e.g., phosphocholine-containing glycosyl inositol phosphorylceramides) (30).
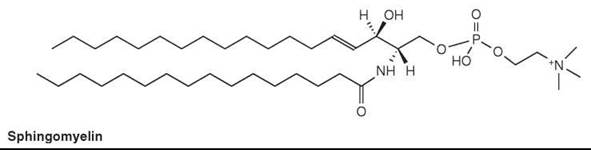
Figure 4. Sphingomyelin.
Ceramides
Ceramide is formed by attachment of a fatty acid (linked by an amide bond) to the amino group of sphingosine or other related amino alcohol. The most commonly found sphingosyl alcohols are D-erythro-sphinganine (dihydrosphingosine, sphinganine), D-erythro-sphingosine (sphingosine, 4-sphingenine), C20-dihydrosphingosine (icosasphinganine), and 4-hydroxy-D-erythro-sphinganine (phytosphingosine, 4-hydroxysphinganine).
Although ceramides are found usually at trace levels in tissues, they play important biologic roles (1). Moreover, they are synthesized as the key intermediates in the biosynthesis of complex sphingolipids, in which glucosylceramide is a key precursor of complex oligoglycolipids (Table 1).
Table 1. Classification of oligoglycolipids
|
Root name |
Abbreviation |
Partial structure* IV III II I |
|
Ganglio |
Gg |
Galβ1-3GalNAcβ1-4Galβ1-4Glc-Cer |
|
Lacto |
Lc |
Galβ1-3GlcNAcβ1-4Galβ1-4Glc-Cer |
|
Neolacto |
nLc |
Galβ1-4GlcNAcβ1-4Galβ1-4Glc-Cer |
|
Globo |
Gb |
GalNAcβ1-3Galα1-4Galβ1-4Glc-Cer |
|
Isoglobo |
iGb |
GalNAcβ1-3Galα1-3Galβ1-4Glc-Cer |
|
Mollu |
Mu |
GalNAcβ1-2Manα1-3Manα1-4Glc-Cer |
|
Arthro |
At |
GalNAcβ1-4GlcNAcβ1-3Manβ1-4Glc-Cer |
*Roman numerals show sugar positions in the root structure.
Cerebrosides
Cerebrosides are glycosides of N-acyl long-chain bases (ceramides). Galactose and glucose are the monosaccharides usually found. Galactosylceramide is the principal glyco- sphingolipid in brain tissue (2). In general, galactosylceramides are present in all nervous tissues, and account for up to 2% of the dry weight of gray matter and 12% of white matter. Oligodendrocytes contain large amounts of these lipids.
Glucosylceramides are considered as characteristic compounds of plants but also found in low amounts in animal tissues, such as spleen, nervous tissues, and erythrocytes. In plants, they are abundant in photosynthetic tissues (31). From nonphotosynthetic tissues, monoglycosylceramides that contain a β-D-mannopyranosyl residue have been isolated.
In addition, fucosylceramide has been reported in a colon carcinoma, a xylose-containing cerebroside was identified in an avian salt gland, and glycosylceramides with mannose and glucuronic or galacturonic acids as sugar moieties have been isolated from some microorganisms. O-Acylated (with fatty acids in various positions of the sugar moiety) glucosylceramides and galactosylceramides have been also found in brain tissues in small amounts. In the equine brain, a galactosyl-ceramide with a long-chain cyclic acetal at the carbohydrate moiety, plasmalo-galactosylceramide, has been identified (32).
Galactosylceramides usually contain large amounts of 2-hydroxy fatty acids, which are linked to the sphingosine base through the amino group. In addition, odd chain fatty acids may also present. Typical fatty acids reported are behenic (C22:0), lignoceric (C24:0), nervonic (C24:1), cerebronic (α-OH C24:0), and 2-hydroxynervonic (α-OH C24:1) (2).
Oligoglycosylceramides
Di- and oligoglycosphingolipids (with two or more sugar residues linked to a ceramide base) are important components of cellular membranes in most eukaryotic organisms and bacteria. Lactosylceramide is the most abundant diglycosphingolipids (or diosylceramide), and is found in many animal tissues at low levels. In kidney and pancreas, galabiosylceramide has been also found. Diosylceramide with mannose units have been reported for some primitive animal species and in plants (e.g., diosyl-ceramide with 1-O-[β-mannosyl (1→4)-O-β-glycosyl]- residue was isolated from bran and endosperm of rice grains).
Neutral oligoglycosylceramides that contain 3 and up to over 20 sugar units in the chain are characteristic of some animal tissues. Seven main types of these compounds exist based on their structural and biosynthetic relationships. In each type, glucose is the primary sugar linked to ceramide with galactose, N-acetylglucosamine, N-acetylgalactosamine or mannose as additional monosaccharides (2).
In general, hundreds of different neutral oligoglycosphingolipids have been identified, and each mammalian organ has a characteristic pattern of neutral ceramides with kidney, lung, spleen, and blood that contain large amounts. In erythrocytes, for example, megaloglycolipids with up to 50 sugar groups are present, but triglycosylceramides to pentaglycosylceramides are usually more abundant.
Most compounds are located on the plasma membrane facing into the extracellular space. The structure of carbohydrate units may determine the function of oligoglycosphingolipids, and one of most important of these is in the immune defense system (33).
In plants, two series of oligoglycosylceramides with either mannosyl or galactosyl residues are synthesized by elongation of glucosylceramide. They are located in the endoplasmic reticulum, Golgi, tonoplast, and plasma membranes. Their functions in plants are poorly understood (34).
Gangliosides
Gangliosides are glycosphingolipids that contain a oligoglycosylceramide to which a sialic acid (P-D-N-acetylneuraminic acid) residue is attached via glycosidic linkages to one or more of the monosaccharide units (i.e., via the hydroxyl group on position 2) or to another sialic acid residue. The level of these lipids is around 6% of total lipids in the brain tissues. Neuronal membranes contain 10-12% gangliosides of the total lipid content (20-25% of the outer layer). They are also present at low levels in all animal tissues but not found outside of the animal kingdom.
Gangliosides are involved in control of cell growth and differentiation as well as cell interactions (33). They also play a key role in the immune defense systems. Many reactions are mediated through the location of these molecules in membrane rafts.
Glycosphingolipid sulfates
Glycosphingolipids with a sulfate ester group attached to the sugar moiety are termed glycosphingolipid sulfates (sulfatides or sulfoglycosphingolipids). The distribution of these compounds in mammalian tissues as well as their fatty acid composition and base composition are usually similar to those of cerebrosides. 3’-Sulfo-galactosylceramide is an example of one of the more abundant glycolipid compounds of brain myelin. It has been also found in many other organs, especially in the kidney. In addition to these “classic” sulfatides, a whole series of sulfatides in organisms that range from mycobacteria to mammals have now been characterized. These include sulfolactosylceramide and other sulfate esters derived from oligoglycosylceramides of the globo series and ganglio series isolated from human kidney, and the ganglioside sulphatides isolated from from echinoderms (35).
Phosphonolipids
Phosphono analogs of various phospholipids, which consist of aminoethylphosphonic acid residues, have been found in nature. The aminoethylphosphonic acid moiety may be linked through a phosphorus-carbon bond to either a ceramide or diacylglycerol backbone in phosphonolipids. Ceramide aminoethyl phosphonate (or ceramide ciliatine) was the first phosphono analog of sphingosylphosphatides, and has been identified in sea anemones (Fig. 5). Lately, it has been also detected in several molluscs, protozoa, bacteria, and bovine brain tissue together with its N-methyl derivative. In some bacterial species, phosphonolipids with a 1-hydroxy-2-aminoethane residue linked to the phosphorus moiety have been also detected. Phosphonogly-cosphingolipids such as 6-O-(aminoethylphosphono)galactosyl ceramide (Fig. 5) and its N-methyl analog, have been isolated from some marine invertebrate. The role and biochemical functions of these lipids are still unknown.
The phosphono form of phosphatidylethanolamine, 1,2-diacyl-sn-glycerol-3-(2'-aminoethyl)phosphonate (Fig. 5), is present in several species of protozoa as well as in bovine tissues and in human aorta. Its alkylacyl and alkenylacyl forms have also been found. From the freshwater cyanobacteria Aphanizomenon flos-aquae, 2-acyloxyethylphosphonate (with unusual biosurfactant properties) has been isolated. Phosphonolipids have been reviewed in References 36 and 37.

Figure 5. Phosphonolipids.
Lipid-Soluble Arsenic Compounds (Arsenolipids)
Lipid-soluble arsenic compounds were first identified from marine organisms over 30 years ago. By now, more than 100 naturally occurring arsenolipids have been detected in a wide range of organisms, including lichens, fungi, freshwater and marine algae, invertebrates, fishes, plants, and animals (38). The level of accumulation of these lipids may vary from 1.5 to 33.8 μg/g dry weight. Some plants and hyper-accumulators contain more than 1 mmol As g-1 dry weight. Many species of bacteria as well as most algal species examined seem to convert arsenic compounds into their methylated derivatives, such as trimethylarsine (39). Structures, distribution, the possible metabolism of arsenolipids, as well as analytical techniques used for their separation, identification, and quantification are thoroughly reviewed in Reference 38.
Other Esters
A group termed “other esters” includes a large variety of functionally diverse lipids. Wax esters are a typical example. The term “wax” is used commonly for esters of long-chain fatty acids with long-chain primary alcohols, but sometimes it is used for the entire mixture of lipids that contain waxes. Ester waxes are present in both plants and animals where they form the water-repellent surface coating (e.g., skin surface of animals and the leaf cuticle). Esters of normal alcohols with monobranched or multibranched fatty acids have been found in the preen glands of birds (2). Complex waxes (in which either the fatty acid or the alcohol component or both has a complex structure) have been isolated from some bacteria. Diesters of phthiocerols (C33-C35 branched-chain diols) with mycocerosic acids (C29-C32 branched-chain acids) are examples of these compounds that have been reported for Mycobacterium spp. (40).
Two types of diester waxes are characteristic of animal skin-surface lipids. In the first type, acyl derivatives of hydroxy acids formed by reaction between the carboxyl group of a fatty acid with the hydroxyl group of a hydroxy acid. In the second type, waxes consist of an alkane α,β-diol in which both hydroxyls are esterified with fatty acids (41).
Ester forms are found in the commonly occurring sterols and vitamin alcohols, such as vitamin A, the D vitamins, and vitamin E, as well as in carotenoids (flowers and green algae) and terpenoids (rose petals). Various other esters have been reported in different bacteria and yeasts where they may occur as glycoside and amino acid derivatives. For more details, see Reference 2.
Fatty Acids
Fatty acids are aliphatic monocarboxylic acids. Over 1000 fatty acids are known with different chain length, positions, configurations, and types of unsaturation. Addition of various functional groups along the aliphatic chain brings more structural and functional diversity to this group.
Saturated fatty acids
In most natural lipids, straight- or normal-chain saturated components (even-numbered) account for up to 10-40% of the total fatty acids. Straight-chain fatty acids with 16 and 18 carbon atoms are the most common naturally-occurring compounds in animals and plants although odd-numbered and even-numbered homologs from 2C to 36C carbon atoms have been reported for natural samples (2). In general, fatty acids do not exist as free carboxylic acids because their high affinity for many proteins. Moreover, elevated levels of free (nonesterified) fatty acids are usually artifacts of cell damage that allow lipases to breakdown the endogenous acyl lipids. Table 2 shows examples of the most important straight chain saturated fatty acids together with the information on their distribution.
Table 2. Some naturally occurring straight chain saturated fatty acids
|
Number of C atoms |
Systematic name |
Common name |
Melting point (C°) |
Occurrence |
|
2 |
Ethanoic |
Acetic |
16.7 |
Esterified to glycerol in ruminant milk fats, some plant triacylglycerols and as alcohol acetates in many plants. As a component of platelet-activating factor. |
|
3 |
Propanoic |
Propionic |
-22.0 |
In the rumen. |
|
4 |
Butanoic |
Butyric |
-7.9 |
In milk fat of ruminants, in the rumen. |
|
6 |
Hexanoic |
Caproic |
-8.0 |
In milk fat. |
|
8 |
Octanoic |
Caprylic |
12.7 |
Minor compound of most animal and plant fats, major component of milk and some seed triacylglycerols. |
|
10 |
Decanoic |
Capric |
29.6 |
Minor compound in many tissues. Major acid in milk and some seed triacylglycerols. |
|
12 |
Dodecanoic |
Lauric |
42.2 |
Widely occurring, major acid in some seed fats, e.g., coconut oil and palm kernel oil. |
|
14 |
Tetradecanoic |
Myristic |
52.1 |
Widespread, occasionally as a major acid. |
|
16 |
Hexadecanoic |
Palmitic |
60.7 |
The most common saturated fatty acid in animals, plants and microorganisms. |
|
18 |
Octadecanoic |
Stearic |
69.6 |
Major fatty acid in animal tissues and some fungi, usually minor in plants, major only in a few plant species (e.g., cocoa butter). |
|
20 |
Eicosanoic |
Arachidic |
75.4 |
Usually minor compounds and only occasionally significant. |
|
22 |
Docosanoic |
Behenic |
80.0 |
Minor component in some seed triacylglycerols and plant waxes. |
|
24 |
Tetracosanoic |
Lignoceric |
84.2 |
Widespread as a constituent of sphingolipids, minor acid in some seed triacylglycerols and plant waxes. |
|
26 |
Hexacosanoic |
Cerotic |
87.7 |
Usual as component of plant and insect Waxes. |
|
28 |
Octacosanoic |
Montanic |
90.9 |
Major acid in some plant waxes. |
Branch-chain fatty acids
Branch-chain fatty acids are usually saturated and have a methyl-group as the branch. In marine animals, unsaturated branched-chain fatty acids have been found, and branches other than methyl are present in microbial lipids. Two distinct types, which are often found in bacteria, are the iso-methyl branched fatty acids with the branch point on the penultimate carbon (one from the end) and anteiso-methyl branched fatty acids with the branch point on the ante-penultimate carbon atom (two from the end). Fatty acids with these structures and with 10 to more than 30 carbon atoms have been identified in some bacteria and in other microorganisms. However, branch points are also found in other positions. For example, 10-R-methyloctadecanoic acid (or tuberculostearic acid) (Fig. 6) is a characteristic of the tubercle bacillus and related species. A wide range of mono-methyl branched fatty acids occurs in some mammalian tissues with the uropygial (preen) gland of birds as a major source. In membranes, they have a function (as an alternative to double bonds) in increasing the fluidity of the lipid bilayer (1).
From Mycobacteria and related species, fatty acids with β-hydroxy-α-alkyl branched structure and high molecular weight (60 to 88 carbons or more) have been isolated. These mycolic acids can contain a variety of functional groups, which include double bonds, cyclopropane rings, methoxy groups, epoxy groups, and keto groups depending on bacterium species.
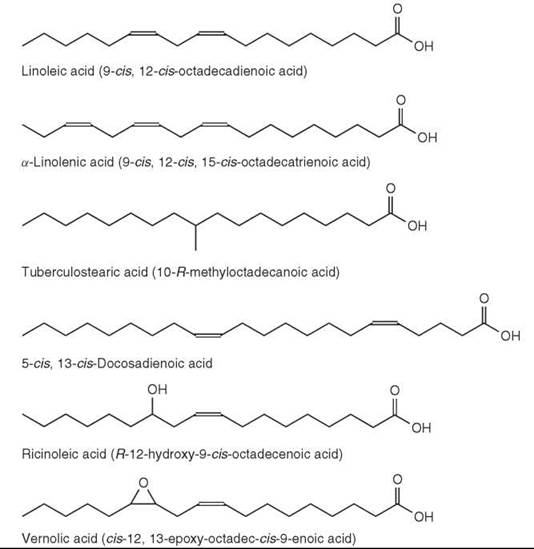
Figure 6. Fatty acid structures.
Monoenoic (Monounsaturated)
Fatty Acids
Straight- or normal-chain (even-numbered) monoenoic fatty acids (with one double bond) amount to a significant proportion of the total fatty acids in most natural lipids. The double bond is usually of the cis- or Z-configuration, but some fatty acids with trans- or E-double bonds are found. For example, a major fatty acid esterified to phosphatidylglycerol in the photosynthetic membranes of higher plants and algae is trans-3-hexadecenoic acid. Often the cis bond is present at the ∆9 position.
The most common monoenes are straight-chain compounds with 16 or 18 carbon atoms. cis-9-Octadecanoic acid (the trivial name oleic acid) is one of the most abundant fatty acids in nature. Table 3 shows examples and the occurrence of the most abundant monoenoic fatty acids.
A cis-bond in a fatty acid introduces a kink (a 60° bend) in the acyl chain with a restriction in motion at that point. Very long chain (C22:1 and longer) cis-monoenoic acids have relatively high melting points, whereas the more common C18 monoenes are usually liquid at room temperature (1, 2).
In addition to the normal ethylenic double bonds, some fatty acids possess acetylenic bonds. They have been found in rare seed oils and some moss species.
Table 3. Some naturally occurring monoenoic fatty acids
|
Number of C atoms |
Systematic name |
Common name |
Melting point (C°) |
Occurrence |
|
16 16 16 |
trans-3-hexadecenoic cis-5-hexadecenoic cis-7-hexadecenoic |
|
53 |
Plant leaf tissues, eukaryotic algae, exclusively as an acyl chain of phosphatidylglycerol in chloroplasts. Bacilli. Bacteria, algae, higher plants. |
|
16 |
cis-9-hexadecenoic |
Palmitoleic |
1 |
Widely distributed in microorganisms, plants and animals. Major components in some seed oils. |
|
18 |
cis-6-octadecenoic |
Petroselenic |
33 |
Major acid in seed oil of the Umbelliferae family. |
|
18 |
cis-9-octadecenoic |
Oleic |
16 |
Most abundant in plants and animals in both structural and storage lipids. Also found in microorganisms. |
|
18 |
trans -9-octadecenoic |
Elaidic |
44 |
In the rumen and in hydrogenated margarines. |
|
18 |
trans -11-octadecenoic |
Vaccenic |
44 |
In rumen fats as a result of hydrogenation of polyunsaturated fatty acids. |
|
18 |
cis -11-octadecenoic |
Cis-Vaccenic |
15 |
Common in bacteria, minor acid in most animal and plant lipids. |
|
20 |
cis -11-eicosenoic |
Gondoic |
24 |
In fish oils and rape seed oil. |
|
22 |
cis-13-docosenoic |
Erucic |
34 |
Major component of rapeseed oil and oils of Cruciferae family, small amounts in animal tissues and fish oil. |
Methylene-interrupted polyunsaturated acids
Significant amounts of polyunsaturated fatty acids (PUFA) with methylene-interrupted (two or more double bonds of the cis-configuration separated by a single methylene group) are found in all higher organisms. In higher plants, the number of double bonds are usually two or three, but in algae and other lower plants as well as in animals there can be up to six (Table 4).
The n-6 and n-3 families are two principal families of polyunsaturated fatty acids occurring in nature and derived biosynthetically from linoleic (9-cis, 12-cis-octadecadienoic or C18:2n-6) and a-linolenic (9-cis, 12-cis, 15-cis-octadecatrienoic or C18:3n-3) acids, respectively (Fig. 6). Both fatty acids are synthesized in plants that can insert double bonds at the ∆9, ∆12, and ∆15 positions in a C18 chain but not in animals (they can insert double bonds at the ∆9, but not at ∆12 and ∆15). Therefore, these two acids are essential dietary components. In animals, additional double bonds are inserted between the carboxyl group and the ∆9 position by ∆5 and ∆6 desaturase enzymes, and the chain can also be extended in two carbon units at the carboxyl end of the molecules by elongase enzymes.
In animal biology, long chain metabolites of linoleate and linolenate may be even more important. The functions of arachidonic (C20:4n-6), eicosapentaenoic (C20:5n-3), and docosahexaenoic (C22:6n-3) acids make them essential conditionally (42). They are signaling molecules and are involved in the regulation of gene expression. These C20 fatty acids are also precursors of eicosanoids, which include prostaglandins (e.g., PGE2 and PGE3 series), thromboxanes, leukotrienes, and lipoxins, which have a variety of important biologic properties.
Polyunsaturated fatty acids are found in most lipid classes, but they are especially important as acyl chains of the phospholipids where they contribute to the particular physical and biologic functional properties of the membranes (1, 2).
Table 4. Some naturally occurring polyunsaturated fatty acids
|
Number of |
Systematic |
Trivial |
Melting |
Occurrence |
|
C atoms |
name |
name |
point (C°) |
|
|
Dienoic acids |
|
|
|
|
|
18 |
cis,cis-6,9- |
Petroslinoleic |
-11 |
Minor acid in animal tissues. |
|
|
octadecadienoic |
|
|
|
|
18 |
cis,cis-9,12- |
Linoleic |
-5 |
Major acid in algae and plant tissues. Essential component for mammals. |
|
|
octadecadienoic |
|
|
|
|
Trienoic acids (methylene interrupted) |
|
|
|
|
|
16 |
All-cis -7,10,13- |
|
|
Algae and higher plants. |
|
|
hexadecatrienoic |
|
|
|
|
18 |
All-cis -6,9,12- |
γ-Linolenic |
|
Minor component of animal tissues, found in a few seed oils (e.g., evening primrose oil). |
|
|
octadecatrienoic |
|
|
|
|
18 |
All-cis-,9,12,15- |
α-Linolenic |
-11 |
Higher plants and algae, component of galatosyldiacylglycerols. |
|
|
octadecatrienoic |
|
|
|
|
Trienoic acids (conjugated) |
|
|
|
|
|
18 |
cis-9, trans-11, trans-13- |
Eleostearic |
49 |
Occasional in some seed oils, especially tung oil. |
|
|
octadecatrienoic |
|
|
|
|
Tetraenoic acids |
|
|
|
|
|
16 |
All-cis-4,7,10,13- |
|
|
In some algae, e.g., Chlorella spp. |
|
|
hexadecatetraenoic |
|
|
|
|
20 |
All-cis-5,8,11,14- |
Arachidonic |
-49.5 |
Most abundant PUFA of animal phospholipids. Common is some algae and bryophytes. |
|
|
eicosatetraenoic |
|
|
|
|
Pentaenoic acids |
|
|
|
|
|
20 |
All-cis -5,8,11,14,17- |
Timnodonic |
|
In marine algae and fish oils as a major component, ubiquitous component of animal phospholipids. |
|
|
eicosapentaenoic |
|
|
|
|
22 |
All-cis -7,10,13,16,19- |
|
|
In marine organisms. |
|
|
docosapentaenoic |
|
|
|
|
Hexaenoic acids |
|
|
|
|
|
22 |
All-cis-4,7,10,13,16,19- |
Clupanodonic |
|
In animals as phospholipid component and in fish. |
|
|
docosahexaenoic |
|
|
|
Bis- and polymethylene-interrupted acids
Fatty acids with bis- or polymethylene-interrupted double bonds, or a mixture of methylene- and polymethylene-separated unsaturation, are found in some plants and marine organisms. From sponges and other marine invertebrates, a wide range of such fatty acids, the demospongic acids, have been isolated. They have bis-methylene-interrupted cis-double bonds and chain lengths (both odd and even) that range from C16 to C34. Double bonds in positions 5 and 9 are usual, although additional bonds may be present in position 7 and/or 9 together with methyl branching.
Bis-methylene-interrupted acids with a A5 cis-double bond only are common in gymnosperms (conifers) with a typical example being pinolenic acid (5-cis, 9-cis, 12-cis-octade-catrienoic acid). This fatty acid has been found in several pine and larch species at the levels of 25-30% of total fatty acids. In angiosperms, 5-cis, 13-cis-docosadienoic acid (Fig. 6) was reported in the seed oil of meadowfoam (Limnanthes alba) (16% of the total fatty acids). An analog of pinolenic acid with a trans double bond in position 5 (5t,9c,12c-18:3) was found to be the main fatty acid of the seed oil of Aquilegia vulgaris (columbine) (1, 2).
Conjugated fatty acids
Fatty acids with two or more conjugated double bonds are found in some plants and animals. In tissues of ruminant animals (and, hence, in meat and dairy products), fatty acids with conjugated diene system were detected as intermediates or by-products in the biohydrogenation of linoleic acid by microorganisms in the rumen. The main isomer, 9-cis, 11-trans-octadecadienoic acid, may account for up to 1% of the total fatty acids of milk fat. 9-cis, 11-trans-15-cis-octadecatrienoic acid, derived from α-linolenic acid, is present in ruminant tissues only in trace levels. This fatty acid has been shown to have several medical properties, especially anti-cancer and anti-atherosclerosis effects.
The only conjugated dienoic fatty acid isolated from plant sources (the seed oil of Chilopsis linearis) is reported to be trans-10, trans-12-octadecadienoic acid. In contrast, fatty acids with conjugated triene systems have been detected in various plant species. Tung oil is the commercial source of the most widespread fatty acid from this group, 9-cis, 11-trans, 13-trans- octadecatrienoic (α-eleostearic) acid (1, 2).
As an example of conjugated tetraenes, a naturally occurring cis- (or α-) parinaric acid (C18:4 9c,11t,13t,15c), should be mentioned. This acid was isolated from the seeds of the Makita tree (Parinarium laurinum), which is a tropical rainforest tree indigenous to Fiji. It is also isolated from Impatiens spp. cis-Parinaric acid has been used to measure phospholipase activity, lipase activity, and as an indicator of lipid peroxidation.
Cyclic fatty acids
Cyclic fatty acids with a carbon ring alone or at the end of the alkyl chain occur naturally in plants, especially in certain seed oils and in microorganisms. Cyclopropane fatty acids are reported occasionally from marine animals and may be synthesized by symbiotic bacteria. In addition, a variety of carbocyclic structures are formed from methylene-interrupted polyenes during food processing.
The cyclopropane fatty acid, cis-11,12-methylene-octadecanoic acid, was reported first for Lactobacillus arabinosus and was given the trivial name lactobacillic acid. By now, fatty acids with a mid-chain cyclopropane group have been found in many bacterial species, including aerobic, anaerobic, Gram-negative, and Gram-positive species. They comprise up to 35% of the membrane lipids depending on the culture conditions.
This type of fatty acid is usually found at low levels (around 1%) in plant oils that contain cyclopropene acids. Cyclopropene acids are characteristic for oils from the Malvalaceae, Stercu- liaceae, Bombaceae, Tiliaceae, and Sapicidaceae families with sterculic acid (9,10-methyleneoctadec-9-enoic acid) and malvalic acid (8,9-methyleneheptadec-8-enoic acid) as two major compounds. Sterculic acid is more abundant (about 50% of the total fatty acids in Sterculiafoetida oil). 2-Hydroxysterculic acid may also be present in this oil as a possible intermediate in the biosynthesis of malvalic acid (through a-oxidation of sterculic acid).
A fatty acid with a terminal ring structure, 11-cyclohexylundecanoic acid, was first isolated as a minor compound of butter fat. It is produced by bacteria in the rumen and may be later digested and accumulated by the host animals. Together with homologous fatty acids, it has been also reported for many bacterial species, especially for those isolated from the extreme environments (e.g., hot springs). For example, in strains of the acidophilic and thermophilic Bacillus acidocardarius, 11-cyclohexylundecanoic acid and 13-cyclohexyltridecanoic acid amounted for 70-90% of the total fatty acids. Moreover, certain Bacillus species may also synthesize fatty acids with terminal C4 to C7 rings when supplied with appropriate precursors (1, 2). In plants, fatty acids with a terminal cyclopent-2-enyl ring have been isolated from the seeds of Flacourtiaceae plant family. 11-Cyclopent-2-enyl-undecanoic (hydnocarpic), 13-cyclopent-2-enyl-tridecanoic (chaulmoogric), and 13-cyclopent-2-enyltridec-6-enoic (gorlic) acids are the most common fatty acids of this type.
Fatty acids with oxygen-containing functional groups
Several fatty acids and their metabolites have oxygen-containing functional groups, which are most commonly a hydroxyl, epoxide, or a furan ring.
Ricinoleic acid (R-12-hydroxy-9-cis-octadecenoic acid) (Fig. 6) accounts for 80-90% of fatty acids in castor oil (from Ricinus communis). It is found in other plant species and in the sclerotia of the ergot fungus (Claviceps purpurea). Lesquerolic acid (R-14-hydroxy-11-cis-eicosenoic acid), which is a C20 homolog of ricinoleic acid, occurs in Lesquerella species (up to 70% of total fatty acids). Isoricinoleic acid (R-9-hydroxy-12-cis-octadecenoic acid, or 9-OH 18:2 12c) is a major acid in the Wrightia species. In plants, several C16 and C18 mono, di, and trihydroxy fatty acids are structural components of cutin (a polyester constituent of plant cuticle).
2-Hydroxy or α-hydroxy acids have been shown to occur in sphingolipids, skin lipids, wool wax, bacterial cell wall lipids and in some seed oils. 3-Hydroxy or β-hydroxy acids are present in bacterial lipids.
Vernolic acid (or cis-12,13-epoxy-octadec-cis-9-enoic acid) (Fig. 6) was the first naturally occurring epoxy fatty acid isolated from the seed oil of Vernonia anthelmintica. It is also found in several Compositae, Malvaceae, and Euphorbiaceae species in significant amounts. Other epoxy acids include coronoric acid (9,10-epoxy-12-cis-octadecenoic acid), which is found in some Compositae species, sunflower oil, and other oils. A C20 homolog of vernolic acid is reported for Alchornea cordifolia.
The most abundant member of the family of furanoid fatty acids is a C20 furanoid acid with two methyl groups on the furan ring. Furanoid fatty acids were detected first in fish oils, but their origin from photosynthetic organisms has been proven. They are now isolated from a wide range of organisms that include yeasts, algae, marine bacteria, and plants (seeds, leaves, and fruits). A particular role of these acids as scavengers of hydroxyl and hydroperoxyl radicals has been suggested (1, 2).
References
1. Gurr MI, Harwood JL, Frayn KN. Lipid Biochemistry. An Introduction. 2002. Blackwell, Oxford.
2. Gunstone FD, Harwood JL, Dijkstra AF. eds. The Lipid Handbook, 3rd edition. 2007. Taylor and Francis, Boca Raton FL.
3. Chapman KD. Occurrence, metabolism and prospective functions of N-acetylethanolamines in plants. Prog. Lipid Res. 2004; 43:302-327.
4. Siegenthaler PA, Tremolieres A. Role of Acyl Lipids in the Function of Photosynthetic Membranes in Higher Plants. In: Lipids in Photosynthesis: Structure, Function and Genetics. Siegenthaler PA and Murata N, eds. 1998. Kluwer Academic Publishers, Dordrecht, Holland. pp. 145-173
5. Harwood JL. Lung surfactant. Prog. Lipid Res. 1987; 26:211-256.
6. Costello CE, Beach DH, Singh BN. Acidic glycerol lipids of Trichomonas vaginalis and Tritrichomonas foetus. Biol. Chem. 2001; 382:275-281.
7. Cronan JE. Bacterial membrane lipids: where do we stand? Annu. Rev. Microbiol. 2003; 57:203-224.
8. Pyne S. Lysolipids: Sphingosine-1-phosphate and Lysophosphatidic acid. In: Bioactive Lipids. Nicolaou A, and Kokotos G, eds. 2004. The Oily Press, Bridgewater, UK. pp. 85-106
9. Nagan N, Zoeller RA. Plasmalogens: biosynthesis and functions. Prog. Lipid Res. 2001; 40:199-229.
10. Synder F. Ether-linked lipids and their bioactive species: occurrence, chemistry, metabolism, regulation and function. In: Biochemistry of Lipids and Lipoproteins and Membranes. Vance DE and Vance JE, eds. 1996. Elsevier, Amsterdam. pp. 183-210
11. Kates M. Glyco-, phosphoglyco- and sulfoglycoglycerolipids from bacteria. In: Handbook of Lipid Research. Glycolipids, Phos- phoglycolipids and Sulfoglycolipids. Kates M, ed. 1990. Plenum Press, New York. pp. 1-122
12. Guschina IA, Harwood JL. Lipids and lipid metabolism in eukaryotic algae. Prog. Lipid Res. 2006;45:160-186.
13. Murray RK, Narasimhan R. Glycerolipids in animal tissues. In: Handbook of Lipid Research. Glycolipids, Phosphoglycolipids and Sulfoglycolipids. Kates M, ed. 1990. Plenum Press, New York. pp. 321-361
14. Anderson R, Kates M, Volcani BE. Identification of the sulfolipids in the non-photosynthetic diatom Nitzschia alba. Biochim. Bio- phys. Acta 1978; 528:89-106.
15. Bisseret P, Ito S, Tremblay PA, Volcani BE, Dessort D, Kates M. Occurrence of phosphatidylsulfocholine, the sulfonium analog of phosphatidylcholine in some diatoms and algae. Biochim. Biophys. Acta 1984; 796:320-327.
16. Goren MB. Sulfolipid I of mycobacterium tuberculosis, strain H37Rv. Biochim. Biophys.Acta 1970; 210:127-138.
17. Kaya K. Chemistry and biochemistry of taurolipids. Prog. Lipid Res. 1992; 31:87-108.
18. Dembitsky VM. Betaine ether-linked glycerolipids: chemistry and biology. Prog. Lipid Res. 1996; 35:1-51.
19. Eichenberger W, Gribi C. Lipids of Pavlova lutheri: cellular site and metabolic role of DGCC. Phytochemistry 1997; 45:1561-1567.
20. Ratledge C, Wilkinson SG. An overview of microbial lipids. In: Microbial Lipids. Ratledge C, Wilkinson SG, eds. 1988. Academic Press, London. pp. 3-22
21. Batrakov SG, Panosyan AG, Konova IV, Bergelson LD. Identification of threo-butane-2,3-diol phospholipid from Actinomyces olivaceus. Biochim. Biophys. Acta 1974; 337:29-40.
22. Suzuki T, Hasegawa K. Diol lipids in the phospholipid fraction of Lipomyces starkeyi grown in the medium containing 1,2-propanediol. Agric. Biol. Chem. 1974; 38:613-620.
23. Zheng W, Kollmeyer J, Symolon H, Momin A, Munter E, Wang E, Kelly S, Allegood J.C, Liu Y, Peng Q, Ramaraju H, Sullards MC, Cabot M, Merrill AH. Ceramides and other bioactive sph- ingolipid backbones in health and disease: lipidomic analysis, metabolism and roles in membrane structure, dynamics, signaling and autophagy. Biochim. Biophys. Acta 2006; 1758:1864-1884.
24. Zeyda M, Stulnig TM. Lipid Rafts & Co.: an integrated model of membrane organization in T cell activation. Prog. Lipid Res. 2006; 45:187-202.
25. Spiegel S, Milstien S. Sphingosine-1-phosphate: an enigmatic signalling lipid. Nature Rev. Molec. Cell Biol. 2003; 4:397-407.
26. Chalfant CE, Spiegel S. Sphingosine-1-phosphate and ceramide-1-phosphate: expanding roles in cell signaling. J. Cell Sci. 2005; 118:4605-4612.
27. Broad TE, Dawson RMC. Formation of ceramide phosphorylethanolamine from phosphatidylethanolamine in the rumen protozoon Entodinium caudatum. Biochem. J. 1973; 134:659-662.
28. Moreau RA, Young DH, Danis PO, Powell MJ, Quinn CJ, Beshah K, Slawecki RA, Dilliplane RL. Identification of ceramide-phosphorylethanolamine in oomycete plant pathogens: pythium ultimum, phytophthora infestans, and phytophthora capsici. Lipids 1998; 33:307-317.
29. Naka T, Fujiwara N, Yana I, Maeda S, Doe M, Minamino M, Ikeda N, Kato Y, Watabe K, Kumazawa Y, Tomiyasu I, Kobayashi K. Structural analysis of sphingophospholipids derived from sphingobacterium spiritivorum, the type species of genus sphingobacterium. Biochim. Biophys. Acta 2003; 1635:83-92.
30. Olsen E, Jantzen E. Sphingolipids in bacterial and fungi. Anaerobe 2001; 7:103-112.
31. Sperling P, Heinz E. Plant sphingolipids: structural diversity, biosynthesis, first genes and functions. Biochim. Biophys. Acta 2003; 1632:1-15.
32. Kolter T. Glycosphingolipids. In: Bioactive Lipids. Nicolaou A, Kokotos G, eds. 2004. The Oily Press, Bridgwater, UK.
33. Merrill AH, Sandhoff K. Sphingolipids: metabolism and cell signalling. In: Biochemistry of Lipids, Lipoproteins and Membranes. Vance DE, Vance J, eds. 2002. Elsevier, Amsterdam. pp. 373-407
34. Heinz E. Plant glycolipids: structure, isolation and analysis. In: Advances in Lipid Methodology - Three. Christie WW, ed. 1996. Oily Press, Dundee. pp. 211-332
35. Ishizuka I. Chemistry and functional distribution of sulfoglycolipids. Prog. Lipid Res. 1997; 36:245-319.
36. Hori T, Nozawa Y. Phospholipids. In: Phospholipids. Hawthorne JN, Ansell GB, eds. 1982. Elsevier Biomedical Press, Amsterdam. pp. 95-128
37. Moschidis MC. Phosphonolipids. Prog. Lipid Res. 1985; 23:223-246.
38. Dembitsky VM, Levitsky DO. Arsenolipids. Prog. Lipid Res. 2004; 43:403-448.
39. Shariatpanahi M, Anderson AC, Abdelghani AA, Engglande AJ, Hughes J, Wilkinson RF. Biotransformation of the pesticide sodium arsenate. J. Environ. Sci. Health 1981; 16:35-47.
40. Barry CE, Lee RE, Mdluli K, Sampson AE, Schroeder BG, Slayden RA, Yuan Y. Mycolic acids: structure, biosynthesis and physiological functions. Prog. Lipid Res. 1998; 37:143-179.
41. Nicolaides F, Fu HC, Ansari MNA. Diester waxes in surface lipids of animal skin. Lipids 1970; 5:299-307.
42. Cunnane SC. Problems with essential fatty acids: time for a new paradigm? Prog. Lipid Res. 2003; 42:544-568.
Further Reading
Christie WW. Lipid Analysis. 2003. Oily Press, Bridgwater.
Fahy E, et al. A comprehensive classification system for lipids. J. Lipid Res. 2005; 46:839-861
Gurr MI. Lipids in Nutrition and Health: A Reappraisal. 1999. Oily Press, Bridgwater.
Hamilton RJ, ed. Waxes: Chemistry, Molecular Biology and Functions. 1995. The Oily Press, Dundee.
Larsson K, Quinn P, Sato K, Tiberg F. Lipids: Structure, Physical Properties and Functionality. 2006. Oily Press, Bridgwater.
Sebedio JL, Christie WW, eds. Trans Fatty Acids in Human Nutrition. 1998. Oily Press, Bridgwater.
Cyberlipid Center: Fats and Oils. http://www.cyberlipid.org.
The Lipid Library. http://www.lipidlibrary.co.uk.
LipidBank: The Official Database of Japanese Conference on the Biochemistry of Lipids (JCBL). http://www.lipidbank.jp.
Lipids Metabolites and Pathways Strategy. www.lipidmaps.org.
See Also
Glycosphingolipids, Chemistry of
Fatty Acid Desaturases, Chemistry of
Lipid Domains, Chemistry of