Chemistry for Dummies
Part IV. Chemistry in Everyday Life: Benefits and Problems
In this part...
Chemistry isn’t just something that’s done in an academic or industrial lab. Professional chemists aren’t the only individuals who do chemistry. You do chemistry, too. Chemistry touches your life each and every day.
Chemistry gives us great benefits, but it can also give us great problems. Our modern society is complex. Chemistry holds the promise of solving many of the problems facing society, making our lives easier and more meaningful.
In the chapters of this part, I show you some applications of chemistry. I cover the chemistry of carbon and show you how it applies to petroleum and the process of making gasoline. I show you how that very same petroleum can be used to make plastics and synthetic fibers. I zip you home to look at the chemistry behind cleaners and detergents, medicines, and cosmetics of all kinds. And I show you some problems that society, technology, and science have created — air and water pollution.
Chapter 14. The Chemistry of Carbon: Organic Chemistry
In This Chapter
· Taking a look at hydrocarbons
· Seeing how to name some simple hydrocarbons
· Checking out the different functional groups
· Discovering organic chemistry’s place in society
The largest and most systematic area of chemistry is organic chemistry, the m chemistry of carbon. Of the 11 to 12 million chemical compounds known, about 90 percent are organic compounds. We burn organic compounds as fuel. We eat organic compounds. We wear organic compounds. We’re made of organic compounds. Our whole world is built of organic compounds.
In this chapter, I give you a brief introduction to organic chemistry. I spend some time showing you the hydrocarbons, compounds of carbon and hydrogen, as well as some other classes of organic compounds and their uses in everyday life. As you read this chapter, you’ll find that a lot of chemistry can be found in carbon.
Organic synthesis: Where it all began
In the years of chemistry, it was thought that organic compounds could only be produced from living organisms. People thought that there had to be a “vital force” involved. But in 1828, the German scientist Friedrich Wohler changed the field of chemistry forever by developing an organic compound, urea, by accident while trying to make an inorganic compound. This was the beginning of out modern field of organic synthesis.
Hydrocarbons: From Simple to Complex
A natural question chemistry students ask is “Why are there so many compounds of carbon?” The answer: Carbon contains four valence electrons and so can form four covalent bonds to other carbons or elements. (A common mistake organic chemistry students make when drawing structures is not ensuring that every carbon has four bonds attached to it.) The bonds that carbon forms are strong covalent bonds (Chapter 7 covers covalent bonds), and carbon has the ability to bond to itself in long chains and rings. It can form double and triple bonds to another carbon or to another element. No other element, with the possible exception of silicon, has this ability. (And the bonds silicon makes aren’t nearly as strong as carbon’s.) These properties allow carbon to form the vast multitude of compounds needed to make an amoeba or a butterfly or a baby.
The simplest organic compounds are called the hydrocarbons, compounds composed of carbon and hydrogen. Economically, the hydrocarbons are extremely important to us — primarily as fuels. Gasoline is a mixture of hydrocarbons. We use methane (natural gas) and propane and butane, all hydrocarbons, for their ability to burn and release a large amount of energy. Hydrocarbons may contain only single bonds (the alkanes) or double bonds (the alkenes) or triple bonds (the alkynes). And they may form rings containing single or double bonds (cycloalkanes, cycloalkenes, and aromatics).
Even compounds containing only carbon and hydrogen have a great deal of diversity; imagine what can happen when a few more elements are mixed in!
From gas grills to gasoline: Alkanes
The simplest of the hydrocarbons are the alkanes. Alkanes are called saturated hydrocarbons — that is, each carbon is bonded to four other atoms. Carbon can form a maximum of four covalent bonds. If those four covalent bonds are to different atoms, then chemists say that the carbon is saturated. There are no double or triple bonds in the alkanes.
Alkanes have the general formula of CnH2n+2, where n is a whole number. If n = 1, then there are four hydrogen atoms, and the result is CH4, methane.
Table 14-1 lists the names of the first ten normal, or straight-chained, alkanes. They really aren’t straight; that’s just what they’re called. When I draw the structures, though, I often show them in a straight line. (Technically, they’re carbon bonds in a tetrahedral fashion with bond angles of 109.5 degrees. See Chapter 7 for a discussion of this stuff, called molecular geometry.) Every carbon, except the end ones, is bonded to two other carbons. Figure 14-1 shows models of the first four listed in the table.
Table 14-1. The First Ten Normal Alkanes (CnH2n+2,)
|
n |
Formula |
Name |
|
1 |
CH4 |
Methane |
|
2 |
C2H6 |
Ethane |
|
3 |
C3H8 |
Propane |
|
4 |
C4H10 |
Butane |
|
5 |
C5H12 |
Pentane |
|
6 |
C6H14 |
Hexane |
|
7 |
C7H16 |
Heptane |
|
8 |
C8H18 |
Octane |
|
9 |
C9H20 |
Nonane |
|
10 |
C10H22 |
Decane |
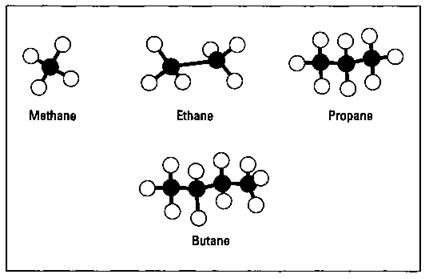
Figure 14-1: The first four alkanes.
Molecular and structural formulas
Table 14-1 shows the molecular formula of some of the alkanes. The molecular formula shows what atoms are present in the compound and the actual number of each. They’re all normal, or straight-chained, hydrocarbons, but the bonding pattern can be illustrated better by using a structural formula. The structural formula shows the atoms present, the actual number of each, and the bonding pattern, or what is bonded to what.
The structural formula can be shown in a number of different ways. One is called the expanded structural formula, which basically shows each covalent bond as a line. With organic compounds such as the hydrocarbons, if you’re really just interested in showing the way the carbons are bonded, you can omit the hydrogen atoms on the expanded form and just indicate them by the covalent-bond line. You can also use a condensed form, which groups parts of the molecule and still indicates the bonding pattern. The condensed form can be done several ways. Figure 14-2 shows a couple of expanded and three condensed forms of the structural formula of butane, C4H10.
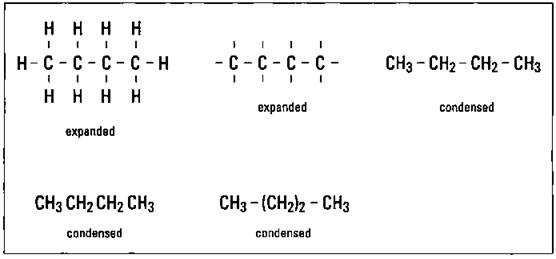
Figure 14-2: Structural formulas of butane.
Naming problems
Sometimes two entirely different compounds — with two entirely different sets of properties — have the same molecular structure. The difference is in the way the atoms are bonded — what’s bonded to what. These types of compounds are called isomers, compounds that have the same molecular formula but different structural formulas. Simply knowing the molecular formula isn’t enough to distinguish between them.
An isomer of butane, for example, has the same molecular formula as the straight-chained compound shown in Figure 14-2, C4H10, but a different bonding pattern. This isomer is mostly referred to by the common name isobutane and is what I call a branched hydrocarbon. Check out Figure 14-3 to see it shown in a variety of ways.
So how do you differentiate which butane you’re talking about when faced with the formula C4H10? Use a unique name that stands only for that one compound. For the straight-chained compound, you can say butane or normal-butane or, better yet, n-butane. The n- makes it perfectly clear to a chemist that you’re talking about the straight-chained isomer.
But what about the other isomer, isobutane? You can use the common name, but it isn’t really accepted everywhere. Chemists all over the world need to agree on the name to help communication among scientists of all nations.
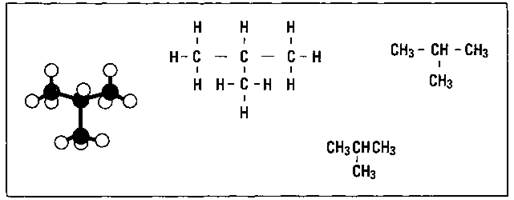
Figure 14-3: Isobutane.
An international group of chemists sets rules for things such as the naming of organic compounds. This group is called IUPAC, the International Union of Pure and Applied Chemistry. These chemists have developed an extremely systematic set of rules for the naming of compounds, and they meet regularly to decide how to name new types of compounds discovered in nature or made in the laboratory.
Systematically naming all the various types of organic compounds would probably take another book, IUPAC Nomenclature For Dummies, Here, I just show you the rules for naming simple alkanes (even the naming of alkanes can get complicated, so it’s important to use the KISS Rule — Keep It Simple, Silly):
ü Rule 1: Locate the longest continuous carbon chain in the alkane (longest means the greatest number of carbon atoms, and continuous means starting at one end of the chain and connecting the carbons with your pencil without picking it up or backtracking). The straight-chained hydrocarbon that has the greatest number of carbons is the parent, or base, name of the alkane. The parent name ends in the suffix –ane.
ü Rule 2: The parent name is modified by adding the names of substituent groups that are attached as branches to the parent compound. Substituent groups are those groups that have been substituted for a hydrogen atom in the alkane parent. For alkane hydrocarbons, these substituent groups are alkane branches that attach to the parent. They are named by taking the alkane name, dropping the -ane, and substituting -yl. So, for example, methane becomes methyl, ethane becomes ethyl, and so on.
ü Rule 3: The position of a particular substituent group on the parent carbon chain is indicated by location numbers. They’re assigned by consecutively numbering the carbons of the parent carbon chain from one end to the other so that the sum of the location numbers of all substituent groups will be as small as possible. (If this doesn’t make any sense — and I feel your pain here — see “Naming examples” for, well, some examples. They make this stuff pretty clear.) The location number of the carbon to which the group is attached is placed in front of the substituent group name and separated from the name by a hyphen.
ü Rule 4: The names of the substituent groups are placed in front of the parent name in alphabetical order. If there are a number of identical substituent groups, then the numbers of all the carbons to which these groups are attached — separated by commas — are used, and the common Greek prefixes — such as di-, tri-, tetra-, penta- — are used. These prefix names are not used to determine alphabetical order.
ü Rule 5: The last substituent alkyl group is used as a prefix to the parent alkane name.
Naming examples
Okay. You’re ready to throw down this book in disgust, aren’t you? I know the alkane naming system sounds ridiculous, but it’s really much easier than it looks. In fact, most of my students find that naming organic compounds is one of the most fun things to do in organic chemistry.
To show you how easy it can be, I’m going to walk you through the process of naming the compound shown in Figure 144 (cross-referencing the rule numbers as I go along).
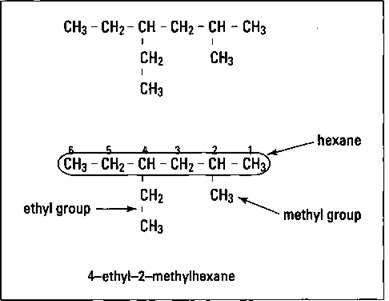
Figure 144: Naming of an alkane.
Using the condensed structural formula, the longest continuous carbon chain is composed of six carbons. Three different 6-carbon chains can actually be used (with the same name resulting eventually), but start with the horizontal one. The chain has six carbons, so the parent name is hexane (Rule 1). You have two substituent groups, one composed of two carbons (ethyl) and another with one carbon (methyl) (Rule 2). Number the parent chain from right to left, giving you alkyl groups at carbons 2 and 4 (sum of 6). Now do the same thing numbering from left to right on the parent chain, giving you groups at carbons 3 and 5 (sum of 8). Compare the right-to-left sum with the left-to-right sum and go with the smallest sum's numbers. So you have a 4-ethyl and a 2-methyl (Rule 3). Placing them alphabetically and with the last substituent alkyl group used as a prefix to the parent alkane name, the result is 4-ethyl-2-methylhexane (Rules 4 and 5).
Got it?
Now you try one: Name the alkane shown in Figure 14-5.
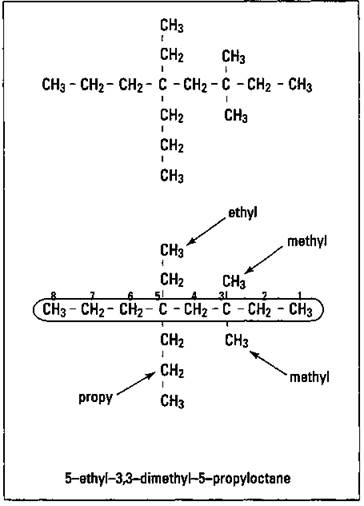
Figure 14-5: Naming another alkane.
The longest carbon chain has eight carbon atoms, so you have an octane parent name. You have two methyl groups (dimethyl), an ethyl group, and a propyl group. Again, number right to left (3+3+5+5=16) instead of left to right (4+4+6+6=20) so that you have 3,3-dimethyl (remember that if you have identical substituent groups, then you include the numbers of all the carbons to which these groups are attached, separated by commas), 5-ethyl, and 5-propyl groups. Then arrange them alphabetically, remembering that the di- of dimethyl doesn’t count: 5-ethyl-3,3-dimethyl-5-propyloctane.
Now, that wasn’t as hard as you thought it’d be, was it?
As you may figure, the more carbons you have, the more isomers are possible. For an alkane with the formula C20H42, there are over 300,000 possible isomers, and for C40H82, there are about 62 trillion possible isomers!
Ring in the cycloalkanes
Alkanes may also form ring systems to make compounds called cycloalkanes. The naming of these compounds is very similar to the branched alkanes, except the cyc/o- prefix is used on the parent name. In the condensed structured formula, the ring is often drawn as lines where the intersection of two straight lines indicates a carbon atom, and the hydrogen atoms aren’t shown at all. Figure 14-6 shows both the expanded and condensed form of 1,3-dimethyl cyclohexane.
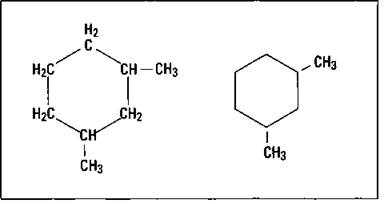
Figure 14-6: 1,3-dimethyl cyclohexane.
Straight-chained alkanes and some cycloalkanes are primarily used as fuels. Methane is the primary component in natural gas and, like most hydrocarbons, is odorless. The gas companies add a stinky organic compound containing sulfur, called a mercaptan, to the natural gas to help alert you to gas leaks. Butane is used in lighters, and propane is used in gas grills. Some of the heavier hydrocarbons are found in petroleum. Combustion is the primary reaction of alkanes.
Say hello to halogenated hydrocarbons
The halogenated hydrocarbons are a related class of compounds. These are hydrocarbons, including alkanes, in which one or more of the hydrogen atoms have been replaced by some halogen — normally chlorine or bromine. Halogen substituents are named as ch/oro-, bro/no-, and so on. Members of this class of compound include chloroform, once used as an anesthetic; carbon tetrachloride, used at one time in dry cleaning solvent; and freons (chlorofluorocarbons, CFCs), elements that have played a major role in the depletion of the ozone layer. See Chapter 18 for a discussion of CFCs and ozone.
Unsaturated hydrocarbons: Alkenes
Alkenes are hydrocarbons that have at least one carbon-to-carbon double bond (C=C). Alkenes that have only one double bond have the general formula CnH2n. For every additional double bond, subtract two hydrogen atoms.
These compounds are called unsaturated hydrocarbons because they don’t have the maximum possible number of hydrogen atoms attached to the carbons. (I’m sure that you’ve heard the terms saturated and unsaturated used in regard to fats and oils in nutrition discussions. They mean exactly the same thing there — saturated fats and oils contain no carbon-to-carbon double bonds, unsaturated fats and oils do, and polyunsaturated fats and oils have more than one C=C per molecule.)
Naming alkenes
Alkenes have a parent name ending with the -ene suffix. You find the longest carbon chain containing the double bond and number it so that the carbon atoms involved in the double bond have the lowest location numbers.
Ethene, written as H2C=CH2 or CH2=CH2, and propene, CH3CH=CH2, are the first two members of the alkene family. These two alkenes are often called by their common names, ethylene and propylene, respectively. They are two of the most important chemicals produced by the chemical industry in the United States. Ethylene is used in the production of polyethylene, one of the most useful plastics produced, and in the production of ethylene glycol, the principal ingredient in most antifreeze. Propylene is used in the production of isopropyl alcohol (rubbing alcohol) and some plastics. Figure 14-7 shows a couple of ways to represent the structural formula of ethene (ethylene).
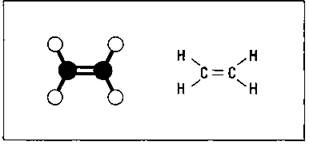
Figure 14-7: Ethene.
Alkene reactions
Although the alkenes will readily undergo combustion, their primary reaction is addition reactions. A double bond is very reactive. One of the bonds can easily be broken, and the two carbons can then form new single bonds to other atoms. One of the most economically important addition reactions is the process of hydrogenation, in which hydrogen is added across the double bond. Here’s the hydrogenation of propene: CH3CH=CH2 + H2 → CH3CH2CH3
This hydrogenation reaction is used in the food industry to convert unsaturated vegetable oils to solid fats (vegetable oil to margarine, for example) and requires the use of a nickel metal catalyst.
Another important addition reaction of alkenes is hydration, the addition of a water molecule across the double bonding, yielding an alcohol. Here’s the hydration of ethylene that gives ethyl alcohol (notice that I show the water molecule in a slightly different way so you can tell where the -OH ends up):
![]()
The ethyl alcohol produced in this way is identical to the ethyl alcohol produced by the fermentation process, but, by federal law, it can’t be sold for human consumption in alcoholic beverages.
Undoubtedly, the most important reaction of the alkenes is polymerization, in which the double bond reacts to produce long chains of the once-alkenes bonded together. This is the process used to produce plastics (see Chapter 16).
It takes alkanes to make the world
Alkynes are hydrocarbons that have at least one carbon-to-carbon triple bond. These compounds have the IUPAC suffix -yne. Hydrocarbons with only a single triple bond have the general formula of CnH2n-2. The simplest alkyne is ethyne, commonly called acetylene. Figure 14-8 shows the structure of acetylene.
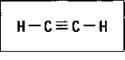
Figure 14-8: Ethyne (Acetylene).
Acetylene is produced in a variety of ways. One way is to react coal with calcium oxide to produce calcium carbide, CaC2. Calcium carbide is then reacted with water to produce acetylene. Miners’ lamps used to be powered by this reaction. Water was dripped on calcium carbide, and the acetylene burned to produce light. Today, most of the acetylene produced is either used in oxyacetylene torches in cutting and welding or to make a variety of polymers (plastics).
Aromatic compounds: Benzene and other smelly compounds
Aromatic hydrocarbons are hydrocarbons that contain a cyclohexene type of ring system that has alternating single and double bonds. The simplest aromatic compound is benzene, C6H6. Benzene is far less reactive than you’d imagine, having those three sets of double bonds. In the current model for benzene, six electrons, two from each of the three double bonds, are donated to an electron cloud associated with the entire benzene molecule. These electrons are delocalized over the entire ring instead of simply located between two carbon atoms. This electron cloud is above and below the planar ring system. Figure 14-9 shows a couple of traditional ways to represent the benzene molecule and a couple of ways to represent the delocalized structure.
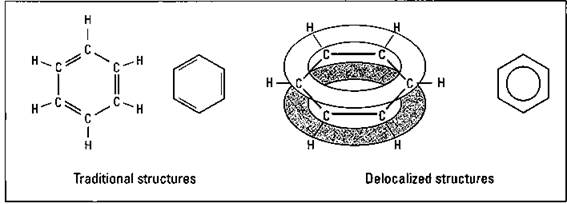
Figure 14-9: Benzene.
Many groups may be attached to this benzene ring, making many new aromatic compounds. For example, an -OH may replace a hydrogen atom. The resulting compound is called phenol. Phenol is used as a disinfectant and in the manufacture of plastics, drugs, and dyes. Two benzene rings fused together make naphthalene, which is commonly called mothballs.
Benzene and its related compounds burn, but they burn with a sooty flame. It’s also been shown that benzene and some of its related compounds are either known or suspected carcinogens.
Functional Groups: That Special Spot
The preceding section covers hydrocarbons, or compounds of just carbon and hydrogen. Can you imagine how many new organic compounds can be generated if a nitrogen atom, halogen atom, sulfur atom, or some other element is thrown in?
Consider some alcohols. Ethyl alcohol (drinking alcohol), methyl alcohol (wood alcohol), and isopropyl alcohol (rubbing alcohol) are quite different and yet remarkably the same in terms of the kinds of chemical reactions they undergo. The reactions all involve the -OH group on the molecule, the part of the molecule that really defines the identity of an alcohol, just as the double bond really defines the identity of an alkene. In many cases, it doesn’t really matter what the rest of the molecule turns out to be. In reactions, one alcohol is pretty much the same as another.
The atom or group of atoms that defines the reactivity of the molecule is called the functional group. For alcohols, it’s the -OH; for alkenes, it’s the C=C; and so on. This makes it much easier to study and classify the properties of compounds. You can learn the general properties of all alcohols instead of the properties of every individual one, for example. The use of functional groups makes the study of organic chemistry much easier.
This section places a spotlight on a few functional groups. What can really make things complex in the lab is that a molecule may have two, three, or more functional groups present, which leads to a wide array of reactions. But this is one of the things that makes organic chemistry challenging — and fun.
Alcohols (rubbing to drinking): R-OH
Alcohols are a group of organic compounds that contain the -OH functional group. In fact, alcohols are often generalized as R-OH, where the R stands for the Rest of the molecule (like that radio newscaster’s “rest of the story”). Alcohols are named using the -o/ suffix replacing the -ane of the corresponding alkane.
Methanol, methyl alcohol, is sometimes called wood alcohol because years ago, its primary synthesis involved heating wood in the absence of air. The more current method of synthesis of methanol involves reacting carbon monoxide and hydrogen with a special catalyst at elevated temperatures:
![]()
About half of the methanol produced in the United States is used in the production of formaldehyde, which is used as embalming fluid and in the plastics industry. It’s also sometimes added to ethanol to make it unfit for human consumption, a process called denaturing. Methanol is also being considered as a replacement to gasoline, but some major problems still need to be overcome. A process that uses methanol in the production of gasoline does exist. New Zealand currently has such a plant that produces about a third of its gasoline.
Ethanol, ethyl alcohol or grain alcohol, is produced primarily in one of two ways. If the ethanol is to be used in alcoholic beverages, it’s produced by the fermentation of carbohydrates and sugars by the enzymes in yeast:
![]()
As a brewer of beer and mead, I can attest that the yeast beasties certainly know how to make good alcohol!
If the ethanol is to be used for industrial purposes, such as a solvent in perfumes and medicines or as an additive to gasoline (making it gasohol), it’s produced by the hydration of ethylene using an acid catalyst:
![]()
Carboxylic acids (smelly things): R-COOH
Figure 14-10 shows the structure of the carboxylic acid functional group.
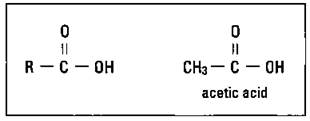
Figure 14-10: The carboxylic acid functional group and acetic acid.
Chemists often use -COOH or -CO2H to indicate this functional group. These compounds are named with an -oic acid suffix. Acetic acid, shown in Figure 14-10, is also called ethanoic acid.
Carboxylic acids can be prepared by the oxidation of an alcohol. For example, leave a bottle of wine in contact with the air or some other oxidizing agent, and the ethanol oxidizes to acetic acid:
![]()
This is something that really breaks my heart, especially if I paid a lot for that particular bottle.
Formic acid, or methanoic acid, can be isolated by the distillation of ants.
Yes, as in the critters that make mountainous sand piles in the cracks of the sidewalk in front of your house. The sting resulting from the bite of an ant is due to formic acid. That’s why applying some base, such as baking soda, helps to neutralize the acid and relieve the pain. (Chapter 12 is a lively read about acids and bases, if you’re interested.)
Many of these organic acids have a distinct odor associated with them. I’m sure you’re familiar with the odor of vinegar, or acetic acid, but other acids have distinct odors, such as those mentioned in Table 14-2.
Table 14-2. Nasty Smells and What They Are
|
CH3(CH2)2COOH |
Butyric acid |
Odor of rancid butter |
|
CH3(CH2)3COOH |
Pentanoic acid |
Odor of manure |
|
CH3(CH2)4COOH |
Hexanoic acid |
Odor of goats |
Esters (more smelly things, but mostly good odors): R-COOR’
The ester functional group is very similar to the carboxylic acid functional group except that another -R group has replaced the hydrogen atom. Esters are made by reacting a carboxylic acid with an alcohol, producing an ester and water. Figure 14-11 shows the synthesis of an ester.
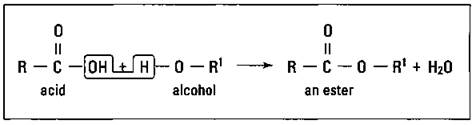
Figure 14-11: Synthesis of an ester.
Although many of the carboxylic acids that esters are made from have foul odors, many esters have pleasant odors. Oil of wintergreen is an ester. Other esters have the odor of bananas, apples, rum, roses, and pineapples. Esters are often used in the flavoring and perfume industry.
Aldehydes and ketones: Related to alcohols
Both aldehydes and ketones are produced by the oxidation of alcohols. These functional groups are shown in Figure 14-12.
Eewww— what's that smell?
I liked organic chemistry when I was in college and enjoyed the lab experiments a bunch — especially the synthesis labs where I got to build complex molecules from simpler ones. I wasn't really wild about the odors, though. Organic chemistry is one of the main reasons chemistry has the reputation for being stinky.
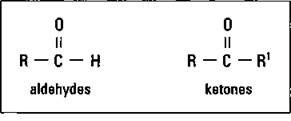
Figure 14-12: Aldehyde and ketone functional groups.
Formaldehyde, HCHO, is an economically important aldehyde. It’s used as a solvent and for preservation of biological specimens. Formaldehyde is also used in the synthesis of certain polymers, such as Bakelite and Melmac (used in melamine dishes). Other aldehydes, especially those with a benzene ring in their structure, have pleasing odors and, like esters, are used in the perfume and flavoring industry.
Acetone, CH3-CO-CH3, is the simplest ketone and has many uses as a solvent, especially for paint. Many of us are familiar with acetone-based fingernail polish remover. And methyl ethyl ketone is the solvent in model airplane glue.
Ethers (sleepy time): R-O-R
Ethers contain an oxygen atom bonded to two hydrocarbon groups, R-O-R. This reminds me of a corny joke that chemists are wont to tell: Did you hear about the ether that was found in the Playboy Mansion? She was an ether bunny!
Diethyl ether was once used as an anesthetic, but its high flammability has caused it to be largely replaced in operating rooms. Because ethers are fairly unreactive (except for combustion), they’re commonly used as solvents in organic reactions. They will, however, slowly react with the oxygen in the atmosphere to form explosive compounds called peroxides.
You can synthesize ethers by the reaction of alcohols with the loss of water (a dehydration reaction). Diethyl ether can be made by reacting ethyl alcohol in the presence of sulfuric acid:
![]()
If you use two different alcohols, you get what’s called a mixed ether, where the two R groups are not the same.
Amines and amides: Organic bases
Amines and amides are derived from ammonia and contain nitrogen in their functional groups. Figure 14-13 shows the amine and amide functional groups.
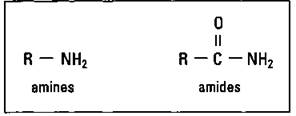
Figure 14-13: Amine and amide functional groups.
Take another look at the figure. Any of the hydrogen atoms attached to the nitrogen on both the amine and amide can be replaced by some other R group.
Amines and amides, like ammonia, tend to be weak bases (see Chapter 12). Amines are used in the synthesis of disinfectants, insecticides, and dyes. They’re in many drugs, both naturally occurring and synthetic. Alkaloids are naturally occurring amines found in plants. Most amphetamines are amines.