Chemistry Essentials for Dummies
Chapter 7. Chemical Reactions
Collision Theory: How Reactions Occur
For a chemical reaction to take place, the reactants must collide. This collision transfers kinetic energy (energy of motion) from one substance to the other. The collision between the molecules provides the energy needed to break the necessary bonds so that new bonds can form. In this section, I discuss two criteria for breaking bonds: The reactants have to collide in the right place, and they have to hit with enough energy to break the bonds.
Hitting the right spot
The molecules must collide in the right orientation, or hit at the right spot, in order for the reaction to occur. The place on the molecule where the collision must take place is called the reactive site. For example, suppose you have an equation showing molecule A-B reacting with C to form C-A and B, like this:
![]()
The way this equation is written, the reaction requires that reactant C collide with A-B on the A end of the molecule. (You know this because the product side shows C hooked up with A — C-A.) If it hits the B end, nothing will happen. The A end of this hypothetical molecule is the reactive site. If C collides at the A end of the molecule, then there’s a chance that enough energy can be transferred to break the A-B bond. After the A-B bond is broken, the C-A bond can form. You can show the equation for this reaction process in this way (I show the breaking of the A-B bond and the forming of the C-A bond as “squiggly” bonds):
![]()
So for this reaction to occur, there must be a collision between C and A-B at the reactive site.
Note that this example is a simple one. I’ve assumed that only one collision is needed, making this a one-step reaction. Many reactions are one-step, but many others require several steps in going from reactants to final products. In the process, several compounds may be formed that react with each other to give the final products. These compounds are called intermediates. You show them in the reaction mechanism, the series of steps that the reaction goes through in going from reactants to products. But in this chapter, I keep it simple and pretty much limit my discussion to one-step reactions.
Adding, releasing, and absorbing energy
REMEMBER. Energy is required to break a bond between atoms. For instance, look at the sample equation ![]() . The collision between C and A-B has to transfer enough energy to break the A-B bond, allowing the C-A bond to form.
. The collision between C and A-B has to transfer enough energy to break the A-B bond, allowing the C-A bond to form.
Sometimes, even if there is a collision, not enough kinetic energy is available to be transferred — the molecules aren’t moving fast enough. You can help the situation somewhat by heating the mixture of reactants. The temperature is a measure of the average kinetic energy of the molecules; raising the temperature increases the kinetic energy available to break bonds during collisions.
The energy you have to supply to get a reaction going is called the activation energy (Ea) of the reaction. Note that even though you’ve added energy, the energy of the products isn’t always higher than the energy of the reactants — heat may be released during the reaction. This section describes two types of reactions, exothermic and endothermic, in which heat is either released or absorbed.
Exothermic reactions: Releasing heat
In an exothermic reaction, heat is given off (released) when you go from reactants to products. The reaction between oxygen and methane as you light a gas stove (from the earlier section “Reactants and Products: Reading Chemical Equations”) is a good example of an exothermic reaction.
Even though the reaction gives off heat, you do have to put in a little energy — the activation energy — to get the reaction going. You have to ignite the methane coming out of the burners with a match, lighter, pilot light, or built-in electric igniter.
Imagine that the hypothetical reaction ![]() is exothermic. The reactants start off at a higher energy state than the products, so energy is released in going from reactants to products. Figure 7-1 shows an energy diagram of this reaction.
is exothermic. The reactants start off at a higher energy state than the products, so energy is released in going from reactants to products. Figure 7-1 shows an energy diagram of this reaction.
In the figure, Ea is the activation energy for the reaction. I show the collision of C and A-B with the breaking of the A-B bond and the forming of the C-A bond at the top of an activation-energy hill. This grouping of reactants at the top of the activation-energy hill is sometimes called the transition state of the reaction. This transition state shows what bonds are being broken and what bonds are being made. As I show in Figure 7-1, the difference in the energy level of the reactants and the energy level of the products is the amount of energy (heat) that is released in the reaction.
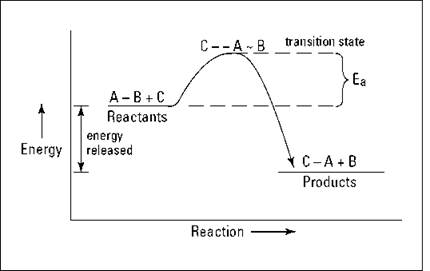
Figure 7-1: Exothermic reaction of ![]() .
.
Endothermic reactions: Absorbing heat
Some reactions absorb energy rather than release it. These reactions are called endothermic reactions. Cooking involves a lot of endothermic reactions — frying those eggs, for example. You can’t just break the shells and let the eggs lie on the pan and then expect the myriad chemical reactions to take place without heating the pan (except when you’re outside in Texas during August; there, the sun will heat the pan just fine).
Suppose that the hypothetical reaction ![]() is endothermic — so the reactants are at a lower energy state than the products. Figure 7-2 shows an energy diagram of this reaction.
is endothermic — so the reactants are at a lower energy state than the products. Figure 7-2 shows an energy diagram of this reaction.
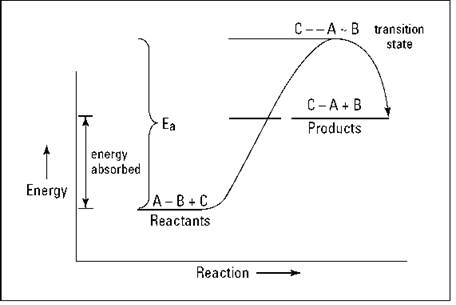
Figure 7-2: Endothermic reaction of ![]() .
.