Chemistry Essentials for Dummies
Chapter 8. Electrochemistry: Using Electrons
In This Chapter
· Finding out about redox reactions
· Balancing redox equations
· Taking a look at electrochemical cells
Combustion is a redox reaction. So are respiration, photosynthesis, and many other biochemical processes people depend on for life. In this chapter, I explain redox reactions, go through the balancing of this type of equation, and then show you some applications of redox reactions in an area of chemistry called electrochemistry. Electrochemistry is an area of chemistry in which we use chemical reactions to produce electrons (electricity) or use electrons (electricity) to cause a desired chemical reaction to take place.
Transferring Electrons with Redox Reactions
Redox reactions — reactions in which there’s a simultaneous transfer of electrons from one chemical species (chemical entity such as an atom or molecule) to another — are really composed of two different reactions:
✓ Oxidation: A loss of electrons
✓ Reduction: A gain of electrons
These reactions are coupled, because the electrons that are lost in the oxidation reaction are the same electrons that are gained in the reduction reaction. In fact, these two reactions (reduction and oxidation) are commonly called half-reactions, because you need these two halves to make a whole reaction, and the overall reaction is called a redox (reduction/oxidation) reaction. In Chapter 7, I describe a redox reaction that occurs between zinc metal and the cupric (copper II, Cu2+) ion. The zinc metal loses electrons and the copper II ion gains them.
Oxidation
You can use three definitions for oxidation:
✓ The loss of electrons
✓ The gain of oxygen
✓ The loss of hydrogen
Because I typically deal with electrochemical cells, I normally use the definition that describes the loss of the electrons. The other definitions are useful in processes such as combustion and photosynthesis.
Loss of electrons
One way to define oxidation is with the reaction in which a chemical substance loses electrons in going from reactant to product. For example, when sodium metal reacts with chlorine gas to form sodium chloride (NaCl), the sodium metal loses an electron, which chlorine then gains. The following equation shows sodium losing the electron:
![]()
When it loses the electron, chemists say that the sodium metal has been oxidized to the sodium cation. (A cation is an ion with a positive charge due to the loss of electrons — see Chapter 5.)
Reactions of this type are quite common in electrochemical reactions, reactions that produce or use electricity.
Gain of oxygen
In certain oxidation reactions, it’s obvious that oxygen has been gained in going from reactant to product. Reactions where the gain of oxygen is more obvious than the gain of electrons include combustion reactions (burning) and the rusting of iron. Here are two examples:
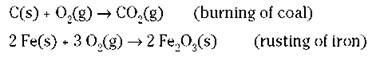
In these cases, chemists say that the carbon and the iron metal have been oxidized to carbon dioxide and rust, respectively.
Loss of hydrogen
In other reactions, you can best see oxidation as the loss of hydrogen. Methyl alcohol (wood alcohol) can be oxidized to formaldehyde:
![]()
In going from methanol to formaldehyde, the compound goes from having four hydrogen atoms to having two hydrogen atoms.
Reduction
You can use three definitions to describe reduction:
✓ The gain of electrons
✓ The loss of oxygen
✓ The gain of hydrogen
Gain of electrons
Chemists often see reduction as the gain of electrons. In the process of electroplating silver onto a teapot, for example, the silver cation is reduced to silver metal by the gain of an electron. The following equation shows the silver cation’s gaining the electron:
![]()
When it gains the electron, chemists say that the silver cation has been reduced to silver metal.
Loss of oxygen
In some reactions, seeing reduction as the loss of oxygen in going from reactant to product is easy. For example, a reaction with carbon monoxide in a blast furnace reduces iron ore (primarily rust, Fe2O3) is to iron metal:
![]()
The iron has lost oxygen, so chemists say that the iron ion has been reduced to iron metal.
Gain of hydrogen
In certain cases, you can describe a reduction as the gain of hydrogen atoms in going from reactant to product. For example, carbon monoxide and hydrogen gas can be reduced to methyl alcohol:
![]()
In this reduction process, the CO has gained the hydrogen atoms.
One's loss is the other's gain
Neither oxidation nor reduction can take place without the other. When those electrons are lost, something has to gain them. Consider, for example, the net-ionic equation (the equation showing just the chemical substances that are changed during a reaction — see Chapter 7) for a reaction with zinc metal and an aqueous copper(II) sulfate solution:
![]()
This overall reaction is really composed of two half-reactions:
![]() (oxidation half-reaction — the loss of electrons)
(oxidation half-reaction — the loss of electrons)
![]() (reduction half-reaction — the gain of electrons)
(reduction half-reaction — the gain of electrons)
TIP. To help yourself remember which reaction is oxidation and which is reduction in terms of electrons, memorize the phrase “LEO goes GER” (Lose Electrons Oxidation; Gain Electrons Reduction).
Zinc loses two electrons; the copper(II) cation gains those same two electrons. Zn is being oxidized. But without Cu2+ present, nothing will happen. That copper cation is the oxidizing agent. It’s a necessary agent for the oxidation process to proceed. The oxidizing agent accepts the electrons from the chemical species that’s being oxidized.
Cu2+ is reduced as it gains electrons. The species that furnishes the electrons is the reducing agent. In this case, the reducing agent is zinc metal.
REMEMBER. The oxidizing agent is the species that’s being reduced, and the reducing agent is the species that’s being oxidized. Both the oxidizing and reducing agents are on the left (reactant) side of the redox equation.
Oxidation numbers
Oxidation numbers are bookkeeping numbers. They allow chemists to do things such as balance redox equations. Oxidation numbers are positive or negative numbers, but don’t confuse them with charges on ions or valences. Chemists assign oxidation numbers to elements using these rules:
✓ For free elements: The oxidation number of an element in its free (uncombined) state is zero (for example, Al(s) or Zn(s)). This is also true for elements found in nature as diatomic (two-atom) elements (H2 O2, N2, F2, Cl2, Br2, or I2) and for sulfur, found as S8.
✓ For single-atom ions: The oxidation number of a monatomic (one-atom) ion is the same as the charge on the ion (for example, Na+ = +1, S2- = -2).
✓ For compounds: The sum of all oxidation numbers in a neutral compound is zero. The sum of all oxidation numbers in a polyatomic (many-atom) ion is equal to the charge on the ion. This rule often allows chemists to calculate the oxidation number of an atom that may have multiple oxidation states, if the other atoms in the ion have known oxidation numbers. (See Chapter 6 for examples of atoms with multiple oxidation states.)
✓ For alkali metals and alkaline earth metals in compounds: The oxidation number of an alkali metal (IA family) in a compound is +1; the oxidation number of an alkaline earth metal (IIA family) in a compound is +2.
✓ For oxygen in compounds: The oxidation number of oxygen in a compound is usually -2. If, however, the oxygen is in a class of compounds called peroxides (for example, hydrogen peroxide, or H2O2), then the oxygen has an oxidation number of -1. If the oxygen is bonded to fluorine, the number is +1.
✓ For hydrogen in compounds: The oxidation state of hydrogen in a compound is usually +1. If the hydrogen is part of a binary metal hydride (compound of hydrogen and some metal), then the oxidation state of hydrogen is -1.
✓ For halogens: The oxidation number of fluorine is always -1. Chlorine, bromine, and iodine usually have an oxidation number of -1, unless they’re in combination with an oxygen or fluorine. (For example, in ClO-, the oxidation number of oxygen is -2 and the oxidation number of chlorine is +1; remember that the sum of all the oxidation numbers in ClO- has to equal -1.)
These rules give you another way to define oxidation and reduction — in terms of oxidation numbers. For example, consider this reaction, which shows oxidation by the loss of electrons:
![]()
Notice that the zinc metal (the reactant) has an oxidation number of zero (the first rule), and the zinc cation (the product) has an oxidation number of +2 (the second rule). In general, you can say that a substance is oxidized when there’s an increase in its oxidation number.
Reduction works the same way. Consider this reaction:
![]()
The copper is going from an oxidation number of +2 to zero. A substance is reduced if there’s a decrease in its oxidation number.