Industrial Chemistry: For Advanced Students - Mark A. Benvenuto 2015
Vitamins
Vitamins are generally small organic molecules that are essential for healthy life in most animals, including humans, always in small amounts. Although the knowledge that eating certain foods had health effects was known to some ancient cultures, their discovery, and understanding of them, really began only at the beginning of the 20th century — and indeed the complete understanding of the role of each of them is an ongoing field of study today. The earliest discoverers conjectured that these materials were all amines, and in the earliest publications the spelling of the word was often “vitamines,” a combination of vita and amines meaning living amines.
There are 13 recognized vitamins today, and several other materials that are sometimes referred to as “vitamin-like substances.” They are present not only in foods consumed by humans, but also in several different types of animal feeds. Trade organizations promote the safe use and consumption of vitamins (Natural Products Association, 2014; Vitamins.org, 2014).
12.1 Sources and production
All vitamins have numerous natural sources, and indeed were first isolated from some natural source. In many cases, plants are the source for vitamins, and humans and other animals have throughout history always eaten these plants to stay healthy. But some animals produce vitamins that other animals, humans included, must then ingest.
12.1.1 Vitamin A
As with several other vitamins, vitamin A is actually a group of related compounds that include several carotenoids, as well as retinal, retinol, and retinoic acid. The structure of retinol is shown in Figure 12.1.
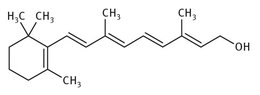
Fig. 12.1: Structure of retinol.
Vitamin A is fat soluble, and is found in many food products, both animal and vegetable. The following are high in vitamin A: cod liver oil; beef, chicken and turkey liver; carrots; dandelion greens; sweet potatoes.
The earliest work on the large scale, multistep synthesis of vitamin A was done by Hoffmann—La Roche, BASF, and Rhône—Poulenc. Hoffmann-La Roche sold its vitamins interests to DSM in 2003, and ultimately Rhône—Poulenc’s medicine and pharmaceutical operations became part of Bayer (Hoffmann-La Roche, 2014; Bayer, 2014; BASF, 2014).
12.1.2 Vitamin B1
Vitamin B1 is also known as thiamine or thiamin, and is classified as a water-soluble vitamin. Natural synthesis of thiamine is through plants, as well as through some bacteria and fungi. The Lewis structure of thiamine is shown in Figure 12.2.
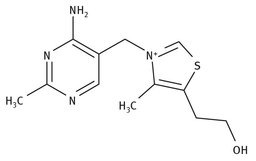
Fig. 12.2: Structure of thiamine.
The large-scale synthesis of thiamine has been pursued for decades, with Hoffmann—La Roche filing an early patent for a biologically based process (US Patent, 2014).
12.1.3 Vitamin B2
Vitamin B2 is also known as riboflavin, another water-soluble vitamin. It is found in a variety of animal and vegetable sources. Cheeses, meats, liver, and kidneys are animal sources that can be high in riboflavin. Almonds, mushrooms, and certain yeasts are other sources that are high in riboflavin. The Lewis structure is shown in Figure 12.3.
Vitamin B2 can be produced on an industrial scale through the use of microbial systems and enzymes. Different companies may use different organisms, for example, BASF appears to utilize Ashbya gossypii exclusively, and produces riboflavin for both human consumption and animal feed (BASF, 2014).
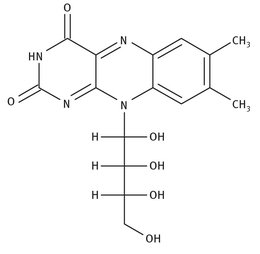
Fig. 12.3: Structure of riboflavin.
12.1.4 Vitamin B3
Vitamin B3 is also known as niacin, and is another water-soluble vitamin. It is found widely in animal and vegetable sources, including beef, chicken, and fish, as well as cereals and peanuts. The Lewis structure of it is shown in Figure 12.4.
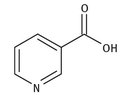
Fig. 12.4: Lewis Structure for niacin.
Niacin can be produced enzymatically from tryptophan, but can also be manufactured from 3-methylpyridine, which itself can be produced from acrolein and ammonia, as shown in Figure 12.5. Thus, the starting materials for this vitamin ultimately come from crude oil, as even the hydrogen in synthetic ammonia comes from fossil sources. Nearly 10 million tons of this vitamin are produced annually, with over half being consumed in animal feed.
12.1.5 Vitamin B5
Vitamin B5 is also called pantothenic acid, and is another water-soluble vitamin. It is found in most meats in at least small amounts, and is also found in whole grains. The Lewis structure and a representative synthesis are shown in Figure 12.6. Because of its presence in so many different plant and animal sources, diseases caused by its deficiency are rare.
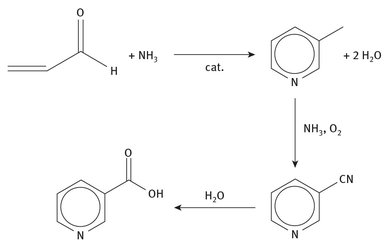
Fig. 12.5: Production of vitamin B3
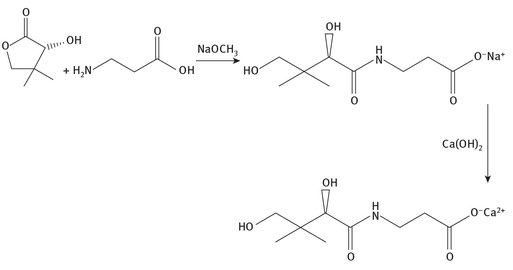
Fig. 12.6: Lewis structure and synthesis for pantothenic acid.
Several thousand tons of vitamin B5 are produced annually, much for use in animal feed. In multivitamin supplements designed for human consumption, vitamin B5 is often included as calcium pantothenate, its calcium salt. This is because the salt of the vitamin is stable over longer periods of time.
12.1.6 Vitamin B6
Vitamin B6 or pyridoxine is another water-soluble vitamin, in this case one which exists in seven different forms. The active form, pyridoxal phosphate (often abbreviated PLP) is shown in Figure 12.7.
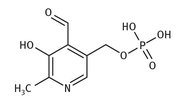
Fig. 12.7: Lewis structure of vitamin B6
Vitamin B6 is another material that is synthesized on a large scale using microbial and enzymatic processes. Some firms, such as Daiichi and Takeda, have developed different methods for the large-scale production of this vitamin (Daiichi, 2014; Takeda, 2014).
12.1.7 Vitamin B7
Vitamin B7 is another water-soluble vitamin, one that is also called biotin, or coenzyme R, or occasionally vitamin H. The Lewis structure is shown in Figure 12.8.
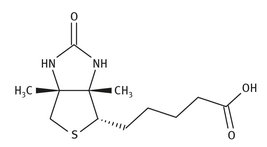
Fig. 12.8: Lewis structure of biotin.
The current synthesis of biotin, which produces the correct isomer, has not changed greatly from that developed by Hoffmann—La Roche in 1949, sometimes called the Sternbach—Goldberg synthesis. Fumaric acid is the starting material for this multistep process.
12.1.8 Vitamin B9
Vitamin B9 is another water-soluble vitamin, which is also called folic acid, folacin, or pteroyl-glutamic acid (and which has in the past been called vitamin M), all of which are much more useable terms than its IUPAC name: (2S)-2-[(4-{[(2-amino-4-hydroxypteridin-6-yl) methyl]amino}phenyl)formamido]pentanedioic acid. The Lewis structure is shown in Figure 12.9, as is one of its one-pot syntheses from three smaller starting materials.
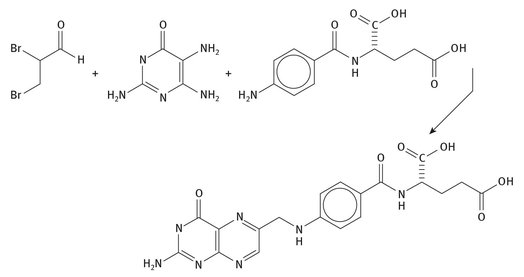
Fig. 12.9: Lewis structure and synthesis of folic acid.
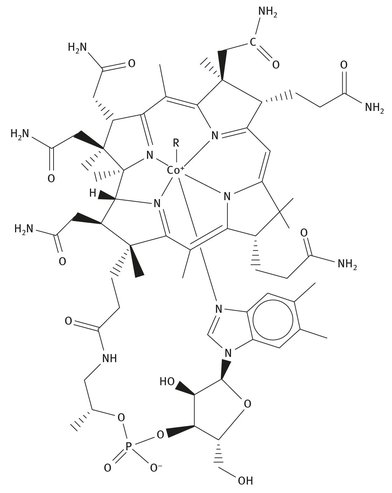
Fig. 12.10: Lewis structure of cobalamin.
12.1.9 Vitamin B12
Vitamin B12, also called cobalamin, is produced by different types of bacteria, and is not simply found in plant and animal sources. Its structure, shown in Figure 12.10,is the most complex of all the vitamins, and it was not discovered until 1954, by Professor Dorothy Hodgkin, who was awarded the Nobel Prize in Chemistry in 1964 for “her determinations by X-ray techniques of the structures of important biochemical substances” (NobelPrize.org, 2014).
The industrial scale production of vitamin B12 is still accomplished via a fermentation synthesis that is bacterially based. The organism Psuedomonas dentrificans is often used for large-scale production of this vitamin.
12.1.10 Vitamin C
Vitamin C is a water-soluble vitamin. Most people are familiar with the idea that citric fruits have significant amounts of vitamin C, and that it can easily be consumed by drinking orange juice. Oranges, lemons, and limes, in addition to several other plant sources, are high in this vitamin C. Its structure is shown in Figure 12.11.
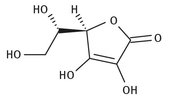
Fig. 12.11: Lewis structure of vitamin C.
The synthesis of vitamin C has been an industrial process for decades, and is often called the Reichstein process. BASF and Merck have been long-term producers of the vitamin. While it is a series of chemical steps, the use of acetobacter, as indicated in Figure 12.12, is crucial for ensuring the correct stereochemistry for all later steps. The final portion of vitamin C synthesis shown in Figure 12.12 is the equilibrium conversion between the open and closed structures of the active molecule.
12.1.11 Vitamin D
Vitamin D is a fat-soluble vitamin that can exist in several different forms, sometimes called vitamers. The broad term “vitamin D” generally refers to the forms that are more specifically called vitamin D2 or vitamin D3. The vitamin D2 structure is shown in Figure 12.13.
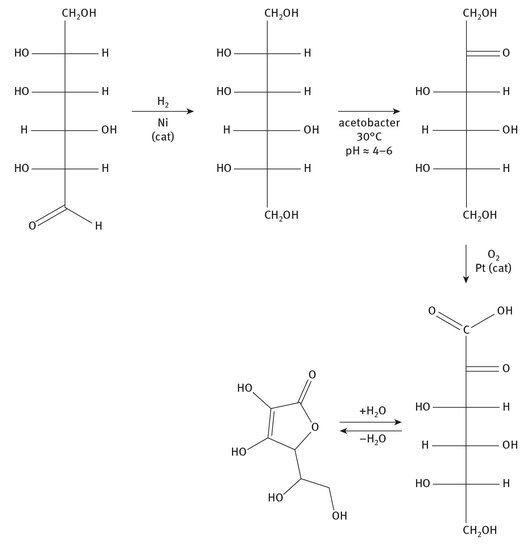
Fig. 12.12: Vitamin C synthesis.
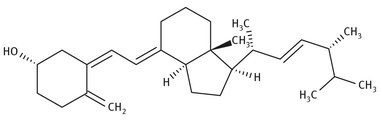
Fig. 12.13: Lewis structure of vitamin D2.
Vitamin D3 is synthesized in human skin from the action of ultraviolet light on a precursor molecule, 7-dehydrocholesterol. This molecule can in turn be extracted from lanolin (sometimes called “wool grease” or “wool wax”) if needed.
12.1.12 Vitamin E
The term vitamin E refers to a class of fat-soluble molecules that are all generally tocotrienols and tocopherols. The Lewis structure of the active form is shown in Figure 12.14, the form designated as alpha-tocopherol.
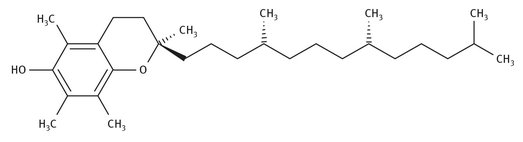
Fig. 12.14: Lewis structure of vitamin E.
Vitamin E can be divided into two groups of molecules, four of which are tocotrienols and four of which are tocopherols. There are three stereocenters in the molecules, and the R, R, R configuration of the tocopherols is the naturally occurring one.
There are different methods for vitamin E production, based on the company that produces it. Archer Daniels Midland claims at its website that “ADM has the broadest vitamin E product line, with alcohols, acetates and succinates in a wide range of manufacturer-friendly forms for capsule, tablet, and compounding applications. We offer powder formulations specifically designed for better flow and compressibility” (Archer Daniels MidlandI, 2014). Clearly, the production of what is called vitamin E is not that of a single molecule.
12.1.13 Vitamin K
Vitamin K has been known to be important to blood coagulation since its discovery in 1929. The term refers to a group of fat-soluble vitamins that are all structurally very similar. Figure 12.15 shows the structure of what is often called vitamin K1, which occurs naturally in numerous leafy, green vegetables. The differences in the vitamin K often involve the length of the aliphatic chain. Large-scale production today usually involves the reaction of phytol (or another long chain alcohol) with menadiol. Both Merck and Roche have been active in producing vitamin K.
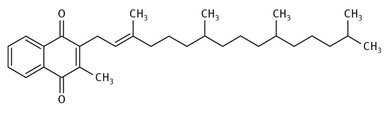
Fig. 12.15: Structure of vitamin K.
12.1.14 Vitamin F
The term “vitamin F” was used in the early part of the 20th century to describe what are now known to be two different fatty acids, called essential fatty acids because the human body does not synthesize them, but must ingest them. These two materials are alpha-linolenic acid and linoleic acid. The structure of each is shown in Figure 12.16.
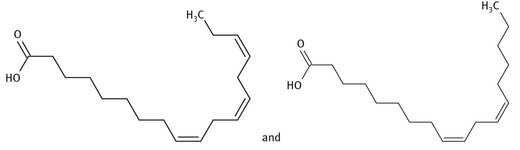
Fig. 12.16: Lewis structures of alpha-linolenic acid and linoleic acid.
The term vitamin F has fallen into disuse, however. These two molecules are now simply referred to as essential fatty acids, or EFAs. They are found in a variety of seeds, with kiwi and flax seeds being high in them.
12.2 Vitamin uses
Beyond simple requirements in human diets, and use as food additives in animal feed, vitamins find the following uses, listed in Table 12.1. Because lack of any vitamin in the human diet causes some disease of deficiency, vitamins fall under the US Food and Drug Administration designation of “generally recognized as safe,” (GRAS) and their addition to foods is usually not considered a problem.
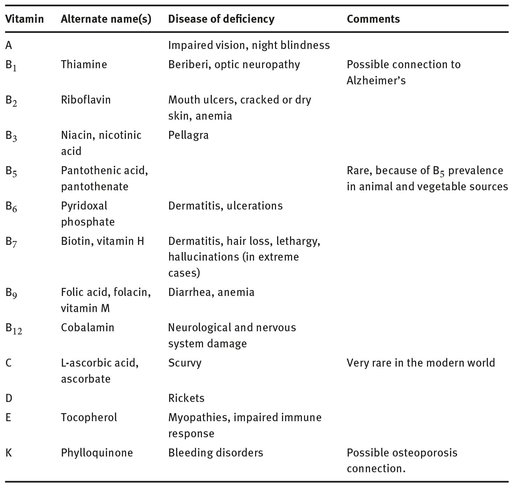
The role some vitamins play in human health continues to be an active area of study. Certain diseases of vitamin deficiency are well known to the general public, such as scurvy from a lack of vitamin C in the diet. Table 12.2 lists each vitamin and the disease to which a deficiency of it leads. But the uses and roles other vitamins play in human health, and how different dosages affect people, continue to be the subject of researchers’ interests.
Table 12.1: Vitamin use.
Vitamin |
Use |
Comments |
A |
Skin cream additive |
|
Riboflavin, B2 |
Orange food coloring |
Has the European number E101. |
B5 |
Shampoos |
Appears to have no real effect on hair care |
B6 |
Antinausea during early pregnancy |
|
B6 with magnesium |
Treating autism |
An alternative medicine choice, degree of effectiveness is still controversial |
B7 |
Protein assays |
|
B12 |
Medical treatment for cyanide poisoning |
|
C |
Food additive |
Despite being prevalent in citrus fruits, vitamin C is added to numerous foods. |
Table 12.2: Vitamins and diseases of deficiency.
12.3 Recycling
Vitamins are consumed in some way, and thus there is no form of vitamin recycling either industrially or at the consumer level. In the past decade, some localities have instituted voluntary drug and medication return programs, which can include vitamin pills. The idea behind such programs is usually to prevent the introduction of medications, vitamins, and other such substances into local waters. Sewage treatment plants have traditionally not been designed to be able to remove these materials from water.
Bibliography
Archer Daniels Midland. Website. (Accessed 14 June 2014, as: http://origin.adm.com/en-US/products/food/vitamin_e/Pages/default.aspx).
BASF. Website. (Accessed 3 June 2014 as: http://www.basf.com).
Bayer. Website. (Accessed 14 June 2014, as: http://www.bayer.com/).
Daiichi. Website. (Accessed 14 June 2014, as: http://www.daiichi-fcj.co.jp/eng/).
Hoffmann-La Roche. Website. (Accessed 14 June 2014, as: http://www.roche.com).
Natural Products Association. Website. (Accessed 3 June 2014, as: http://www.npainfo.org/).
NobelPrize.org. The Official Website of the Nobel Prize. Website. (Accessed 10 June 2014, as: http://www.nobelprize.org/nobel_prizes/chemistry/laureates/1964/index.html).
Takeda. Website. (Accessed 14 June 2014, as: http://www.takeda.com/).
US Patent. Process for the production of yeast having high vitamin b1 potency, US 2328025 A.
Vitamins.org. Website. (Accessed 3 June 2014, as: http://www.vitamins.org).