Industrial Chemistry: For Advanced Students - Mark A. Benvenuto 2015
Hydrogen peroxide
Hydrogen peroxide is the simplest of all the peroxides, and is produced each year on a scale of roughly 2 million tons. It has been considered a useful chemical in several different processes for over a century. Its production today is through an ingenious process, generally called the anthraquinone process, although it is sometimes referred to as the Riedl—Pfleiderer process. The process dates back to the 1930s and was pioneered at BASF (The Manufacture of Hydrogen Peroxide, 2014; BASF/Dow/Solvay, 2014).
13.1 Production
The anthraquinone process is currently the only industrial-scale process for the production of hydrogen peroxide (BASF/Dow/Solvay, 2014). The reaction chemistry is fascinating, but can be reduced to the deceptively simple, single reaction:
![]()
The reason this simplicity is considered deceptive is the means by which the hydrogen and oxygen are reacted and joined: the anthraquinone. Figure 13.1 shows the catalytic and cyclic nature of hydrogen peroxide’s production.
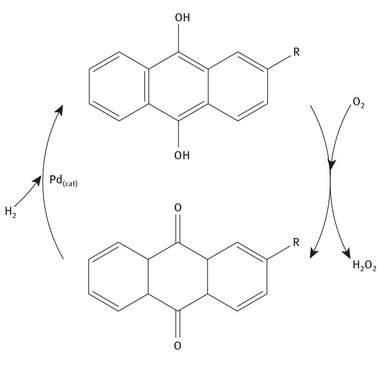
Fig. 13.1: The anthraquinone process.
The R group on the anthraquinone can be different alkyl groups, but ethyl is the most commonly used, with tert-butyl being another proven group. Putting the entire process into somewhat more detail and explaining the chemical reactions includes the following four major steps:
1. Hydrogenation. Molecular hydrogen is introduced to the anthraquinone with a palladium catalyst, producing anthrahydroquinone.
2. Catalyst separation. Because the palladium catalyst is denser than all other components involved in the process, it is filtered out, recovered and reused.
3. Air input. This oxidation step is the point at which oxygen from air is introduced to the process. Air is the least expensive component in the process and produces the oxidation.
4. Product separation. The hydrogen peroxide and anthraquinone are separated using an extraction column and the anthraquinone is reused.
There are times when more anthraquinone is added to the system, but for the most part it is recycled. Its purpose is to transport the hydrogen. At the BASF website, the process is explained as follows:
The manufacturing process involves the catalysis of the reaction of H2 (obtained from processing Maui gas) with atmospheric O2 to give H2O2. Anthraquinone (Q) is used as a H2 carrier. The major uses of peroxide in New Zealand utilize its strongly oxidizing nature to oxidize various chemical groups.
During the hydrogenation step of hydrogen peroxide production, the working solution containing the dissolved anthraquinone is hydrogenated using hydrogen gas in a slurry-type hydrogenator using alumina loaded with a small amount of palladium catalyst. The anthraquinone working solution is heated in the presence of either activated alumina or activated magnesia, thereby regenerating its hydrogen peroxide synthesizing capacity (BASF/Dow/Solvay, 2014; BASF, 2014).
So, not only is palladium used catalytically and recycled, but also alumina or magnesia must be used as a support.
Because of the size of hydrogen peroxide production facilities, they must be located near a source of water, which must itself be further purified. Also, they can be co-located with a facility that produces hydrogen gas.
13.2 Uses of hydrogen peroxide
The general public tends to think of hydrogen peroxide as a treatment for minor scrapes and cuts, and think of the bottle of 1% H2O2 solution in a household medicine cabinet as the primary use for the material. While this is indeed one use for hydrogen peroxide, its profile of uses is quite wide.
Hydrogen peroxide is always miscible in water in any proportion, and is often sold as solutions of various concentrations. US Peroxide lists the following industrial applications of this material:
· — Refinery and petrochemical reduced sulfur compound treatment
· — Refinery and petrochemical supplemental dissolved oxygen
· — Refinery and petrochemical high strength wastewater pretreatment
· — Nitrogen oxides (NOx) abatement
· — Cooling and process water system
· — Antimicrobial applications
· — Gas scrubbing
· — Iron/metals removal
· — Filamentous bulking control
· — Inorganic toxic pollutant destruction, sulfide oxidation
· — Inorganic toxic pollutant destruction, cyanide treatment
· — Inorganic toxic pollutant destruction, dechlorination
· — Inorganic toxic pollutant destruction, arsenic removal
· — Organic toxic pollutant destruction formaldehyde oxidation
· — Organic toxic pollutant destruction mercaptan. control (US Peroxide, 2014).
Clearly, the number of applications for hydrogen peroxide is great and the industries that utilize it vary widely.
13.3 Recycling and reuse
The process of manufacturing hydrogen peroxide is a very efficient one in terms of recycling materials that are used. As mentioned, the anthraquinone and the solvents are routinely recycled, as is the metal catalyst.
Since virtually all hydrogen peroxide is used in some reaction to produce other chemicals or user-end products, none of it is recycled.
Bibliography
BASF. Website. (Accessed 8 February 2014, as: http://www.basf.com/group/corporate/en/brand/DD_6_AND_DD_PG).
BASF/Dow/Solvay HPPO Plant, Belgium. Website. (Accessed 6 February 2014, as: http://www.chemicals-technology.com/projects/basf-hppo/).
The Manufacture of Hydrogen Peroxide. Website. (Accessed 6 February 2014 as: http://nzic.org.nz/ChemProcesses/production/1E.pdf).
US Peroxide. Website. (Accessed 19 May 2014, as: http://www.h2o2.com/industrial/applications.aspx?pid=83&name=Industrial-Applications).