Industrial Chemistry: For Advanced Students - Mark A. Benvenuto 2015
Lead
Lead metal is one of only a few metals known to have been used in ancient times. The atomic symbol for it, Pb, derives from the Latin word, plumbum. The English word plumber is also derived from this word, since metal plumbing and materials to direct water were historically often made from lead.
17.1 Sources
Lead is rarely found in its reduced state in nature. Rather, several oxide and sulfide ores exist, all of which contain varying amounts of lead. Ores — often mixed ores —with as little as 5% of the element can generally be refined, so that the lead can be reduced and used. Table 17.1 shows the major types of lead ores.
Table 17.1: Sources of lead (Handbook of Mineralogy, 2014).
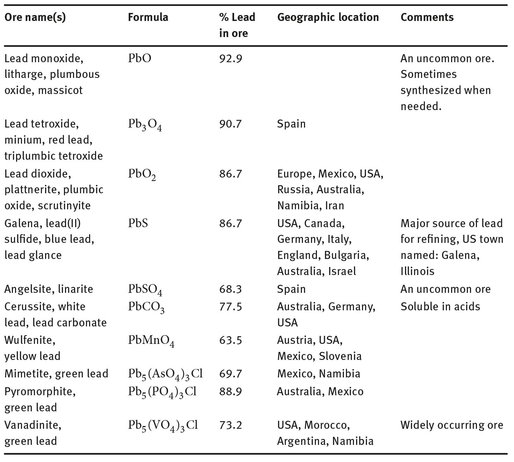
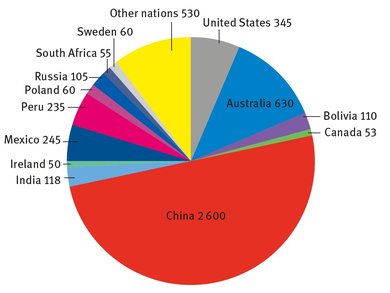
Fig. 17.1: Global lead production (USGS, 2013).
The United States Geologic Survey (USGS) does track lead production in its Mineral Commodity Summaries each year (USGS, 2013). A global breakdown of lead production is shown in Figure 17.1, in terms of thousands of metric tons. This figure does not take into account recycled lead metal, only that which has been refined from ores.
17.2 Extraction chemistry
The extraction of lead from lead ores is not always easy to represent with simple chemical reactions, because lead ores occur with lead co-mingled with several other metal elements, often metals such as silver. In such cases, lead is considered the co-product of silver, with silver being the more expensive metal.
One example of this co-production is the lead (and silver) of Bolivia. The famous Potosi mines, which fueled the royal coffers of Spain with silver from the 1500s until the waning of Spain’s New World empire in the 1800s, produced enough silver metal that South American silver had displaced European silver in European coinage by the 1580s. The mines still function today long after Bolivia’s independence, but now lead is the primary metal that is extracted, and silver the secondary.
Lead is separated from silver by what is called the Parkes process. The process utilizes molten lead that contains silver as impurities. Zinc is added to the melt, because it is not miscible with lead, but does become a molten alloy with the silver. The reaction chemistry can be simplified to the following:
![]()
The zinc—silver alloy forms a separate layer from the lead, and is separated from it, leaving a pure lead behind. The zinc—silver alloy is then heated, vaporizing the zinc. The reaction chemistry for this can be presented as follows:
![]()
The process works well because zinc and lead are essentially immiscible and because zinc is quite inexpensive. This process also works to remove gold as an impurity in a predominantly lead product. This was patented (number 13,118) by Alexander Parkes in 1850, and thus is a very mature chemical technique that is still used today.
17.3 Uses
The general public thinks of automotive batteries as the main use of lead metal, but there are several others as well (USGS, 2013; EUROBAT, 2014; Battery Council International, 2014). They include: ballast, diving weights, counterweights, and radiation shielding materials. The US Department of Defense Strategic and Critical Materials 2013 Report on Stockpile Requirements list major uses of lead as: “storage batteries, ammunition, broadcast and wireless communications equipment” (US Department of Defense Strategic and Critical Materials, 2013).
Leaded glass
What is often referred to as lead glass generally contains between 20% and 40% lead oxide (PbO) in the final material. Calcium is often present in glass, and is replaced by the lead oxide to make this product. The process of making this type of glass is over 300 years old, and has been used over that time with only minor modifications. The clarity of the finished glass makes it desirable for many products, and the lead oxide added to it actually makes it easier to melt and less energy intensive to form.
17.4 Recycling
Lead metal recycling is a mature industry, with the lead from lead-acid batteries being recycled worldwide (America’s Battery Recyclers, 2014). The sulfuric acid used in such batteries is so inexpensive that the vast majority of it is neutralized and discarded, but the plastic housing and the lead metal are routinely recycled.
Bibliography
America’s Battery Recyclers. Website. (Accessed 20 May 2014, as: http://www.americasbatteryrecyclers.com/association.html).
Association of European Automotive and Industrial Battery Manufacturers | EUROBAT. Website. (Accessed 20 May 2014, as: http://www.eurobat.org/).
Battery Council International. Website. (Accessed 20 May 2014, as: http://batterycouncil.org/).
Handbook of Mineralogy. Mineral Data Publishing. Website. (Accessed 23 May 2014, pdf, as: http://www.handbookofmineralogy.org/).
US Department of Defense Strategic and Critical Materials 2013 Report on Stockpile Requirements. Downloadable document.
USGS Mineral Commodities Summary 2013. Website. (Accessed 20 May, 2014, as: http://minerals.usgs.gov/minerals/pubs/mcs/2013/mcs2013.pdf).