Industrial Chemistry: For Advanced Students - Mark A. Benvenuto 2015
Bromine
Bromine, a halogen that when isolated takes the form of a toxic red—brown liquid with a strong odor (the name is derived from the Greek word for “stench”), is a relatively rare element, but one that has found several uses in the chemical industry. It does not have a particularly long history, having only been isolated in 1825—1826. But it quickly became a commodity chemical, with Dow Chemical extracting it before the turn of the twentieth century (First Commercial Production of Bromine, 2014; Dow Chemical Company, 2014).
21.1 Mining and sources
Bromine is neither found widely in different minerals nor found in nature as a free element. Because the bromide ion is quite soluble in water, bromides can be concentrated from brine solutions, and then extracted from them. Bromide exists in seawater in concentrations as high as 65 ppm; it also occurs at much higher concentrations in certain brines at various locations throughout the world. Figure 21.1 shows the most recent data for bromine production by country. The United States is excluded from the figure because the two companies that manufacture it in the country did not wish to disclose proprietary data (USGS, 2013).
The total worldwide annual output is almost 600,000 metric tons. Both Israel and Jordan extract bromine as bromide from their shared body of water and boundary, the Dead Sea. The salinity of this small, endorheic sea is roughly nine times greater than that of the oceans. The United States extracts bromine from wells in the southern counties of the state of Arkansas (Albermarle Chemical Company, 2014) as well as from Michigan (Dow Chemical Company, 2014). Japan and China extract it from seawater, as do most other nations, through large evaporation ponds or pools. In all cases, some concentration of the existing brine must be accomplished to make the recovery of bromine economically profitable.
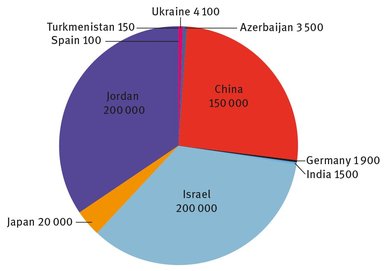
Fig. 21.1: Global bromine production, in metric tons (USGS, 2013).
21.2 Extraction chemistry
The isolation of bromine from bromide-containing brines is the major way by which the element is isolated. Although there are methods which start with hydrobromic acid (HBr) or sodium bromide (NaBr), these are small in scope, and not utilized on an industrial scale.
Bromine refining and isolation depends on the availability of chlorine, which is much more common halide and halogen. The brines must be concentrated, but not dried, and then have chlorine gas flushed through them along with air. Chlorine is thus reduced to the anion, while bromide is oxidized to the free element. The reaction chemistry can be shown very simply, as depicted in Figure 21.2.
![]()
Fig. 21.2: Bromine isolation.
The earliest commercial production of bromine was an electrolytic process pioneered by Herbert Dow, who with the help of his process found the Dow Chemical Company in Michigan, USA in 1897. At the same time when Mr. Dow was producing bromine, he was also producing and using chlorine, as both elements were found in the brine wells that served as the point source for bromine (First Commercial Production of Bromine, 2014).
Large amounts of elemental chlorine are produced in what is called the chlor-alkali process, which can operate in three somewhat different configurations. Sodium hydroxide is often considered the main product of that process, chlorine is the second, and hydrogen gas is a third isolable product. Chlorine is also produced in Downs cells, as a co-product in the production of elemental sodium, which was discussed in Chapter 16.
21.3 Uses
Bromine finds a very diverse suite of uses in industry and in consumer end-use products. Curiously, the ease with which the carbon—bromine bond can be broken is what has made organo-bromides very useful materials in flame retardants. The resulting radical combines with those formed in combustions, and thus shuts the combustion down. Unfortunately, in the atmosphere, these bromine radicals can react with ozone, with the overall affect being a weakening of the ozone layer. For that reason, continued efforts are made to find replacement materials that do not contain bromine for use as flame retardants and fumigants.
21.3.1 Flame retardants
While there are several major uses for bromine, the series of end-products that occupy the largest market share are flame retardants. The bromine—carbon bond in brominated organic compounds is weaker than a hydrogen—carbon covalent bond, and when broken, tends to form a radical. As mentioned, these radicals are able to suppress those produced during combustion. Thus, brominated compounds find use in flame and fire retardants. There are a large number of these compounds, but some of the common ones are:
· — Decabromodiphenyl ethane
· — Decabromodiphenyl ether (decaBDE)
· — Brominated polystyrene
· — Hexabromocyclododecane
· — Tetrabromobisphenol A
· — Tetrabromophthalic anhydride
These and other poly-brominated compounds can be produced through direct addition of bromine or HBr to the organic starting molecule (Albermarle Chemical Company, 2014). Small amounts of them are often added to large volume plastics until the desired level of flame retardant ability is met. For example, hexabromocyclododecane is usually added to polystyrene in less than 10% by volume, because its flame retarding ability is quite high.
Lewis structures of these flame retardants are shown in Figure 21.3.
The United States Environmental Protection Agency and corporate producers of brominated flame retardants have agreed that decaBDE will no longer be manufactured in the United States by the end of 2013. The reason was concern over breakdown products and their effects on human health (USGS, 2013).
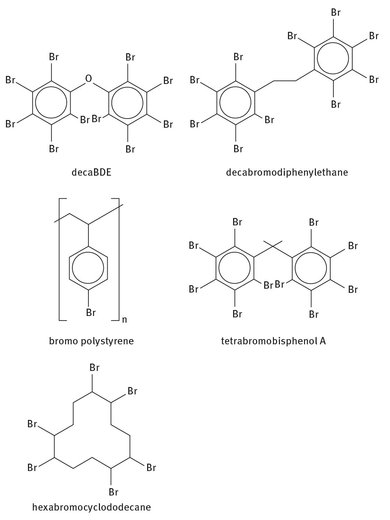
Fig. 21.3: Lewis structures of several flame retardants.
21.3.2 Halons
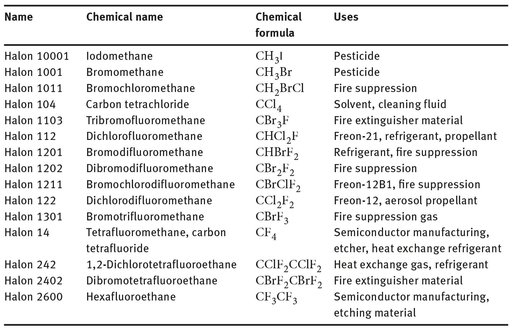
The term “halons” indicates a series of short-chain alkanes that contain one or more types of halogen, often including bromine. Several of them have not only been used as flame retardants, but they also find use as dry-cleaning fluids and in other agricultural applications. Table 21.1 lists several of the halons that have been made on a commercial scale, as well as their uses.
It can be seen that of the 15 halons listed here, eight have at least one bromine atom in their formula. While many of these make effective fire suppression materials or pesticides, many of the halons have been banned for public use in the past 10 years, because they and their breakdown products are known to be ozone-depleting materials.
Table 21.1: Halons and their uses.
21.3.3 Fumigants
The compound 1,2-dibromoethane, more commonly known as ethylene bromide (an analog to 1,2-dichloroethane, historically also called Dutch liquid) is easily prepared from ethylene, and has found wide use as a fumigant for various insects. The reaction chemistry for its formation can be seen in Figure 21.4.
![]()
Fig. 21.4: 1,2-Dibromoethane production.
This compound also found use in leaded fuels, where the bromine combined with lead to create lead(II) bromide. This was phased out when leaded automobile fuels were replaced with the current unleaded fuels.
This compound was also used as an insecticide in various crop applications, but has generally been replaced by other, less toxic insecticides.
Methyl bromide found a specific niche use as a pesticide in the production of strawberries, enabling maximum strawberry growth. It too has been phased out, in part because improper, liberal spraying of it is believed to have caused three fatalities among farm workers in southern California. Its production requires methanol and hydrobromic acid, and can be written fairly simply, as shown in Figure 21.5.
![]()
Fig. 21.5: Methyl bromide production.
Several other bromine-containing pesticides that have been produced on an industrial scale have been listed in Table 21.1.
21.3.4 Drilling fluids
Bromine is combined with metals such as zinc, sodium, or calcium because the resulting solutions made from the salts are dense and useful as oil drilling solutions. InIn general, the additives in drilling fluids keep the drill from overheating or help keep the cuttings and materials suspended by raising the viscosity of the fluid.
21.3.5 Vegetable oil
Brominated vegetable oil (sometimes abbreviated BVO) is a food additive, mentioned in Chapter 11, which is added in small amounts to soft drinks. When added at the level of a few parts per million, BVO helps solubilize citrus flavors in soft drinks. Several countries have banned the production of soft drinks using BVO as an ingredient, citing health problems by consumers who ingest large amounts of such beverages. Consumers do know when BVO is in a product however, because it is listed in the ingredients. Figure 21.6 shows a bottle of Mountain Dew® with BVO listed as the second to the last ingredient. This means that it is the second to the smallest amount of ingredient present in the beverage.
Overall, the use of bromine for BVO is much smaller than that for other applications, such as flame retardants.

Fig. 21.6: Bottled Mountain Dew with brominated vegetable oil as ingredient.
21.4 Reuse and recycling
The recycling of bromine is possible in large enough industrial processes. The USGS Mineral Commodity Summaries 2013 makes the interesting statement:
Some bromide solutions were recycled to obtain elemental bromine and to prevent the solutions from being disposed of as hazardous waste. Hydrogen bromide is emitted as a byproduct in many organic reactions. Plastics containing bromine flame retardants can be incinerated as solid or-organic waste, and the bromine can be recovered (USGS, 2013).
While this does not explain the chemistry involved in the recycling processes, it implies that the driver for such recycling programs is economic. Specifically, there are cost savings when brominated material does not have to be disposed of — and paid for — as hazardous waste.
Bromine-containing materials also find use as a sorbent material in coal-fired power plants, in scrubbing mercury out of the effluent stacks (VGB PowerTech, 2014). While this is not a form of bromine recycling, it is an example of bromine being used toto capture another material that is considered dangerous to human and environmental health when released. Abatement of mercury emission from power plants continues to be an area of concern.
Bibliography
Albermarle Chemical Company. Website. (Accessed, 30 May 2014, as: http://www.albemarle.com/Products-and-Markets/Fine-Chemistry/Performance-Chemicals/Bromine-and-Derivatives-160.html).
Dow Chemical Company. Website. (Accessed 30 May 2014, as: http://www.dow.com).
First Commercial Production of Bromine in the United States. Website. (Accessed 26 January 2014, as: http://www.acs.org/content/dam/acsorg/education/whatischemistry/landmarks/bromineproduction/first-electrolytic-production-of-bromine-historical-resource.pdf), a free download pamphlet.
US Geological Survey, Mineral Commodity Summaries 2013. Website. (Accessed 26 January 2014, as: http://minerals.usgs.gov/minerals/pubs/mcs/2013/mcs2013.pdf).
VGB PowerTech. Website. (Accessed 30 May 2014, as: http://www.vosteen-consulting.de/downloads/2006_03_Vosteen_Consulting_Special_print_VGB_PT03_06_U.pdf).