Introductory Chemistry: A Foundation - Zumdahl S.S., DeCoste D.J. 2019
Chemical Composition
Molar Mass
Objectives
· To understand the definition of molar mass.
· To learn to convert between moles and mass of a given sample of a chemical compound.
A chemical compound is, fundamentally, a collection of atoms. For example, methane (the major component of natural gas) consists of molecules each containing one carbon atom and four hydrogen atoms . How can we calculate the mass of mole of methane? That is, what is the mass of molecules? Because each molecule contains one carbon atom and four hydrogen atoms, mole of molecules consists of mole of carbon atoms and moles of hydrogen atoms (Fig. 8.3). The mass of mole of methane can be found by summing the masses of carbon and hydrogen present:
Figure 8.3.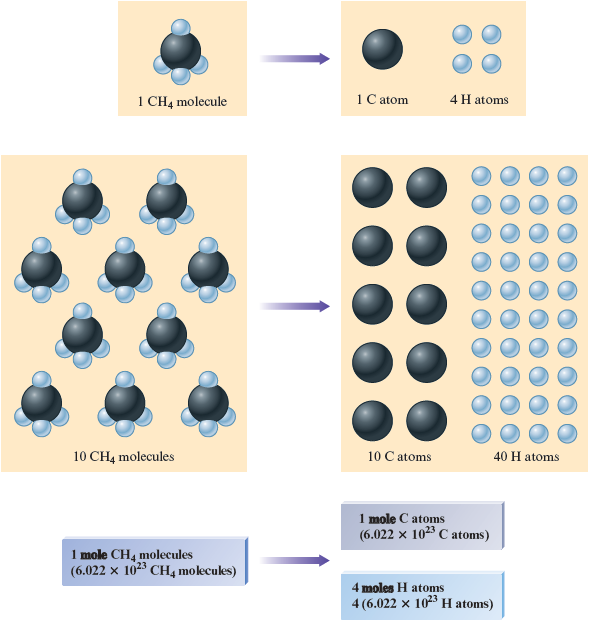
Various numbers of methane molecules showing their constituent atoms.
The quantity g is called the molar mass for methane: the mass of mole of molecules. The molar mass * of any substance is the mass (in grams) of mole of the substance. The molar mass is obtained by summing the masses of the component atoms.
Interactive Example 8.5. Calculating Molar Mass
Calculate the molar mass of sulfur dioxide, a gas produced when sulfur-containing fuels are burned. Unless “scrubbed” from the exhaust, sulfur dioxide can react with moisture in the atmosphere to produce acid rain.
Solution
Where Are We Going?
We want to determine the molar mass of sulfur dioxide in units of g/mol.
What Do We Know?
· The formula for sulfur dioxide is , which means that mole of molecules contains mole of sulfur atoms and moles of oxygen atoms.
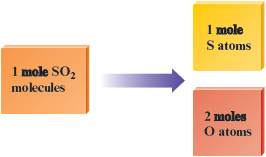
· We know the atomic masses of sulfur ( g/mol) and oxygen ( g/mol).
How Do We Get There?
We need to find the mass of mole of molecules, which is the molar mass of .
The molar mass of is g. It represents the mass of mole of molecules.
Reality Check The answer is greater than the atomic masses of sulfur and oxygen. The units (g/mol) are correct, and the answer is reported to the correct number of significant figures (to two decimal places).
Self-Check: Exercise 8.4
· Polyvinyl chloride (called PVC), which is widely used for floor coverings (“vinyl”) and for plastic pipes in plumbing systems, is made from a molecule with the formula . Calculate the molar mass of this substance.
See Problems 8.27, 8.28, 8.29, and 8.30.
Some substances exist as a collection of ions rather than as separate molecules. For example, ordinary table salt, sodium chloride , is composed of an array of and ions. There are no molecules present. In some books the term formula weight is used instead of molar mass for ionic compounds. However, in this book we will apply the term molar mass to both ionic and molecular substances.
To calculate the molar mass for sodium chloride, we must realize that mole of contains mole of ions and mole of ions.
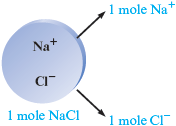
Therefore, the molar mass (in grams) for sodium chloride represents the sum of the mass of mole of sodium ions and the mass of mole of chloride ions.
The molar mass of is g. It represents the mass of mole of sodium chloride.
Interactive Example 8.6. Calculating Mass from Moles
Calcium carbonate, (also called calcite), is the principal mineral found in limestone, marble, chalk, pearls, and the shells of marine animals such as clams.
a. Calculate the molar mass of calcium carbonate.
b. A certain sample of calcium carbonate contains moles. What is the mass in grams of this sample?
Solution
a. Where Are We Going?
We want to determine the molar mass of calcium carbonate in units of g/mol.
What Do We Know?
· The formula for calcium carbonate is . One mole of contains mole of , mole of , and moles of .
· We know the atomic masses of calcium ( g/mol), carbon ( g/mol), and oxygen ( g/mol).
How Do We Get There?
Calcium carbonate is an ionic compound composed of and ions. One mole of calcium carbonate contains mole of and mole of ions. We calculate the molar mass by summing the masses of the components.
Reality Check The answer is greater than the atomic masses of calcium, carbon, and oxygen. The units (g/mol) are correct, and the answer is reported to the correct number of significant figures (to two decimal places).
b. Where Are We Going?
We want to determine the mass of moles of .
What Do We Know?
· From part a, we know that the molar mass of is g/mol.
· We have moles of .
How Do We Get There?
We determine the mass of moles of by using the molar mass.
This can be diagrammed as follows:
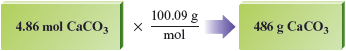
Reality Check We have a bit less than moles of , which has a molar mass of about g/mol. We should expect an answer a bit less than g, so our answer makes sense. The number of significant figures in our answer ( g) is three, as required by the initial number of moles ( moles).
Self-Check: Exercise 8.5
· Calculate the molar mass for sodium sulfate, . A sample of sodium sulfate with a mass of g represents what number of moles of sodium sulfate?
See Problems 8.35, 8.36, 8.37, and 8.38.
In summary, the molar mass of a substance can be obtained by summing the masses of the component atoms. The molar mass (in grams) represents the mass of mole of the substance. Once we know the molar mass of a compound, we can compute the number of moles present in a sample of known mass. The reverse, of course, is also true, as illustrated in Example 8.7.
Interactive Example 8.7. Calculating Moles from Mass
Juglone, a dye known for centuries, is produced from the husks of black walnuts. It is also a natural herbicide (weed killer) that kills off competitive plants around the black walnut tree but does not affect grass and other noncompetitive plants. The formula for juglone is .
a. Calculate the molar mass of juglone.
b. A sample of g of pure juglone was extracted from black walnut husks. How many moles of juglone does this sample represent?

© Melinda Fawver/Shutterstock.com
Black walnuts encased in green husks growing on a tree.
Solution
a. Where Are We Going?
We want to determine the molar mass of juglone in units of g/mol.
What Do We Know?
a. The formula for juglone is . One mole of juglone contains moles of , moles of , and moles of .
b. We know the atomic masses of carbon ( g/mol), hydrogen ( g/mol), and oxygen ( g/mol).
How Do We Get There?
The molar mass is obtained by summing the masses of the component atoms. In mole of juglone there are moles of carbon atoms, moles of hydrogen atoms, and moles of oxygen atoms.
Reality Check Ten moles of carbon would have a mass of about g, and our answer is higher than this. The units (g/mol) are correct, and the answer is reported to the correct number of significant figures (to one decimal place).
b. Where Are We Going?
We want to determine the number of moles of juglone in a sample with mass of g.
What Do We Know?
· From part a, we know that the molar mass of juglone is g/mol.
· We have g of juglone.
How Do We Get There?
The mass of mole of this compound is g, so g is much less than a mole. We can determine the exact fraction of a mole by using the equivalence statement
to derive the appropriate conversion factor:
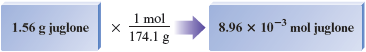
Reality Check The mass of mole of juglone is g, so g is much less than mole. Our answer has units of moles, and the number of significant figures in our answer is three, as required by the initial mass of g.
Interactive Example 8.8. Calculating Number of Molecules
Isopentyl acetate, , the compound responsible for the scent of bananas, can be produced commercially. Interestingly, bees release about of this compound when they sting. This attracts other bees, which then join the attack. How many moles and how many molecules of isopentyl acetate are released in a typical bee sting?
Solution
Where Are We Going?
We want to determine the number of moles and the number of molecules of isopentyl acetate in a sample with mass of g.
What Do We Know?
· The formula for isopentyl acetate is .
· We know the atomic masses of carbon ( g/mol), hydrogen ( g/mol), and oxygen ( g/mol).
· The mass of isopentyl acetate is g.
· There are molecules in mole.
How Do We Get There?
We are given a mass of isopentyl acetate and want the number of molecules, so we must first compute the molar mass.
This means that mole of isopentyl acetate ( molecules) has a mass of g.
Next we determine the number of moles of isopentyl acetate in , which is g. To do this, we use the equivalence statement
which yields the conversion factor we need:
Using the equivalence statement units, we can determine the number of molecules:
This very large number of molecules is released in each bee sting.
Reality Check The mass of isopentyl acetate released in each sting is much less than the mass of mole of , so the number of moles should be less than mole, and it is ( mol). The number of molecules should be much less than , and it is ( molecules).
Our answers have the proper units, and the number of significant figures in our answer is one, as required by the initial mass.
Self-Check: Exercise 8.6
· The substance Teflon, the slippery coating on many frying pans, is made from the molecule. Calculate the number of units present in g of Teflon.
See Problems 8.39 and 8.40.