Introductory Chemistry: A Foundation - Zumdahl S.S., DeCoste D.J. 2019
Chemical Composition
Formulas of Compounds
Objective
· To understand the meaning of empirical formulas of compounds.
Assume that you have mixed two solutions, and a solid product (a precipitate) forms. How can you find out what the solid is? What is its formula? There are several possible approaches you can take to answering these questions. For example, we saw in Chapter 7 that we can usually predict the identity of a precipitate formed when two solutions are mixed in a reaction of this type if we know some facts about the solubilities of ionic compounds.
However, although an experienced chemist can often predict the product expected in a chemical reaction, the only sure way to identify the product is to perform experiments. Usually we compare the physical properties of the product to the properties of known compounds.
Sometimes a chemical reaction gives a product that has never been obtained before. In such a case, a chemist determines what compound has been formed by determining which elements are present and how much of each. These data can be used to obtain the formula of the compound. In Section 8.6 we used the formula of the compound to determine the mass of each element present in a mole of the compound. To obtain the formula of an unknown compound, we do the opposite. That is, we use the measured masses of the elements present to determine the formula.
Recall that the formula of a compound represents the relative numbers of the various types of atoms present. For example, the molecular formula tells us that for each carbon atom there are two oxygen atoms in each molecule of carbon dioxide. So to determine the formula of a substance, we need to count the atoms. As we have seen in this chapter, we can do this by weighing. Suppose we know that a compound contains only the elements carbon, hydrogen, and oxygen, and we weigh out a -g sample for analysis. Using methods we will not discuss here, we find that this -g sample of compound contains g of carbon, g of hydrogen, and g of oxygen. We have just learned how to convert these masses to numbers of atoms by using the atomic mass of each element. We begin by converting to moles.
Carbon
Hydrogen
Oxygen
Let’s review what we have established. We now know that g of the compound contains mole of atoms, mole of atoms, and mole of atoms. Because mole is , these quantities can be converted to actual numbers of atoms.
Carbon
Hydrogen
Oxygen
These are the numbers of the various types of atoms in g of compound. What do these numbers tell us about the formula of the compound? Note the following:
1. The compound contains the same number of and atoms.
2. There are twice as many atoms as atoms or atoms.
We can represent this information by the formula , which expresses the relative numbers of , , and atoms present. Is this the true formula for the compound? In other words, is the compound made up of molecules? It may be. However, it might also be made up of molecules, molecules, molecules, molecules, molecules, and so on. Note that each of these molecules has the required ratio of carbon to hydrogen to oxygen atoms (the ratio shown by experiment to be present in the compound).
When we break a compound down into its separate elements and “count” the atoms present, we learn only the ratio of atoms—we get only the relative numbers of atoms. The formula of a compound that expresses the smallest whole-number ratio of the atoms present is called the empirical formula or simplest formula. A compound that contains the molecules has the same empirical formula as a compound that contains molecules. The empirical formula for both is . The actual formula of a compound—the one that gives the composition of the molecules that are present—is called the molecular formula . The sugar called glucose is made of molecules with the molecular formula (Fig. 8.4). Note from the molecular formula for glucose that the empirical formula is . We can represent the molecular formula as a multiple (by ) of the empirical formula:
Figure 8.4.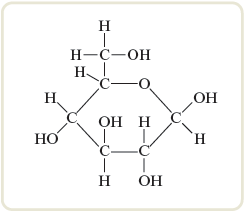
The glucose molecule. The molecular formula is , as can be verified by counting the atoms. The empirical formula for glucose is .
In the next section, we will explore in more detail how to calculate the empirical formula for a compound from the relative masses of the elements present. As we will see in Sections 8.8 and 8.9, we must know the molar mass of a compound to determine its molecular formula.
Interactive Example 8.10. Determining Empirical Formulas
In each of the following cases, the molecular formula for a compound is given. Determine the empirical formula for each compound.
a. . This is the molecular formula for benzene, a liquid commonly used in industry as a starting material for many important products.
b. . This is the molecular formula for a substance commonly called dioxin, a powerful poison that sometimes occurs as a by-product in the production of other chemicals.
c. . This is the molecular formula for one of the reactants used to produce nylon.
Solution
a. ; is the empirical formula. Each subscript in the empirical formula is multiplied by to obtain the molecular formula.
b. ; ; is the empirical formula. Each subscript in the empirical formula is multiplied by to obtain the molecular formula.
c. ; is the empirical formula. Each subscript in the empirical formula is multiplied by to obtain the molecular formula.