Introductory Chemistry: A Foundation - Zumdahl S.S., DeCoste D.J. 2019
Energy
Thermodynamics
Objective
· To understand how energy flow affects internal energy.
The study of energy is called thermodynamics . The law of conservation of energy is often called the first law of thermodynamics and is stated as follows:
The energy of the universe is constant.
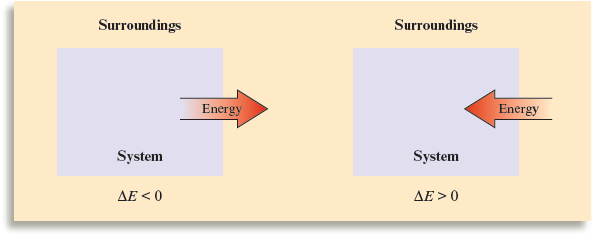
The internal energy , , of a system can be defined most precisely as the sum of the kinetic and potential energies of all “particles” in the system. The internal energy of a system can be changed by a flow of work, heat, or both. That is,
where
(“delta”) means a change in the function that follows
represents heat
represents work
Thermodynamic quantities always consist of two parts: a number, giving the magnitude of the change, and a sign, indicating the direction of the flow. The sign reflects the system’s point of view. For example, when a quantity of energy flows into the system via heat (an endothermic process), is equal to , where the positive sign indicates that the system’s energy is increasing. On the other hand, when energy flows out of the system via heat (an exothermic process), is equal to , where the negative sign indicates that the system’s energy is decreasing.
In this text the same conventions apply to the flow of work. If the system does work on the surroundings (energy flows out of the system), is negative. If the surroundings do work on the system (energy flows into the system), is positive. We define work from the system’s point of view to be consistent for all thermodynamic quantities. That is, in this convention the signs of both and reflect what happens to the system; thus we use .
Critical Thinking
· You are calculating in a chemistry problem. What if you confuse the system and the surroundings? How would this affect the magnitude of the answer you calculate? The sign?