Introductory Chemistry: A Foundation - Zumdahl S.S., DeCoste D.J. 2019
Energy
Chapter Review
Key Terms
· energy (10.1)
· potential energy (10.1)
· kinetic energy (10.1)
· law of conservation of energy (10.1)
· work (10.1)
· state function (10.1)
· temperature (10.2)
· heat (10.2)
· system (10.3)
· surroundings (10.3)
· exothermic (10.3)
· endothermic (10.3)
· thermodynamics (10.4)
· first law of thermodynamics (10.4)
· internal energy (10.4)
· calorie (10.5)
· joule (10.5)
· specific heat capacity (10.5)
· enthalpy (10.6)
· calorimeter (10.6)
· Hess’s law (10.7)
· fossil fuels (10.9)
· petroleum (10.9)
· natural gas (10.9)
· coal (10.9)
· greenhouse effect (10.9)
· energy spread (10.10)
· matter spread (10.10)
· entropy (10.10)
· second law of thermodynamics (10.10)
· spontaneous process (10.10)
For Review
· Energy is conserved.
· The law of conservation of energy states that energy is neither created nor destroyed in any process.
· In a process, energy can be changed from one form to another, but the amount of energy remains constant.
· Thermodynamics is the study of energy and its changes.
· Energy is classified as one of the following:
o Kinetic energy—energy due to the motion of an object
o Potential energy—energy due to the position or composition of an object
· Some functions, called state functions, depend only on the beginning and final states of the system, not on the specific pathway followed.
o Energy is a state function.
o Heat and work are not state functions.
· Temperature indicates the vigor of the random motions of the components of that substance.
· Thermal energy is the sum of the energy produced by the random motions of the components.
· Heat is a flow of energy between two objects due to a temperature difference between the objects.
o An exothermic process is one in which energy as heat flows out of the system into the surroundings.
o An endothermic process is one in which energy as heat flows into the system from the surroundings.
o The common units for heat are calories and joules.
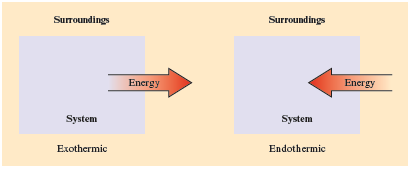
· Internal energy is the sum of the kinetic and potential energy associated with an object.
o Internal energy can be changed by two types of energy flow:
§ Heat
§ Work
o
· Specific heat capacity is the energy required to change the temperature of a mass of g of a substance by .
· For a process carried out at constant pressure, the change in enthalpy of that process is equal to the heat.
Characteristics of
o If a reaction is reversed, the sign of is also reversed.
o The magnitude of is directly proportional to the quantities of reactants and products in a reaction. If the coefficients in a balanced reaction are multiplied by an integer, the value of is multiplied by the same integer.
· Hess’s law enables the calculation of the heat for a given reaction from known heats of related reactions.
· A calorimeter is a device used to measure the heat associated with a given chemical reaction.
· Although energy is conserved in every process, the quality (usefulness) of the energy decreases in every real process.
· Natural processes occur in the direction that leads to an increase in the disorder (increase in entropy) of the universe.
o The principal driving forces for natural processes can be described in terms of “matter spread” and “energy spread.”
· Our world has many sources of energy. The use of this energy affects the environment in various ways.
Active Learning Questions
These questions are designed to be considered by groups of students in class. Often these questions work well for introducing a particular topic in class.
· 1.
Look at Fig. 10.1 in your text. Ball A has stopped moving. However, energy must be conserved. So what happened to the energy of ball A?
· 2.
A friend of yours reads that the process of water freezing is exothermic. This friend tells you that this can’t be true because exothermic implies “hot,” and ice is cold. Is the process of water freezing exothermic? If so, explain this process so your friend can understand it. If not, explain why not.
· 3.
You place hot metal into a beaker of cold water.
a. Eventually what is true about the temperature of the metal compared to that of the water? Explain why this is true.
b. Label this process as endothermic or exothermic if we consider the system to be
I. the metal. Explain.
II. ii. the water. Explain.
· 4.
What does it mean when the heat for a process is reported with a negative sign?
· 5.
You place g of a hot metal in g of cold water. Which substance (metal or water) undergoes a larger temperature change? Why is this?
· 6.
Explain why aluminum cans make good storage containers for soft drinks. Styrofoam cups can be used to keep coffee hot and cola cold. How can this be?
· 7.
In Section 10.7, two characteristics of enthalpy changes for reactions are listed. What are these characteristics? Explain why these characteristics are true.
· 8.
What is the difference between quality and quantity of energy? Are both conserved? Is either conserved?
· 9.
What is meant by the term driving forces? Why are matter spread and energy spread considered to be driving forces?
· 10.
Give an example of a process in which matter spread is a driving force and an example of a process in which energy spread is a driving force, and explain each. These examples should be different from the ones given in the text.
· 11.
Explain in your own words what is meant by the term entropy. Explain how both matter spread and energy spread are related to the concept of entropy.
· 12.
Consider the processes
a. Which process is favored by energy spread? Explain.
b. Which process is favored by matter spread? Explain.
c. How does temperature affect which process is favored? Explain.
· 13.
What if energy was not conserved? How would this affect our lives?
· 14.
The internal energy of a system is said to be the sum of the kinetic and potential energies of all the particles in the system. Section 10.1 discusses potential energy and kinetic energy in terms of a ball on a hill. Explain potential energy and kinetic energy for a chemical reaction.
· 15.
Hydrogen gas and oxygen gas react violently to form water.
a. Which is lower in energy: a mixture of hydrogen gases, or water? Explain.
b. Sketch an energy-level diagram (like Fig. 10.5) for this reaction and explain it.
· 16.
Consider four -g samples of water, each in a separate beaker at . Into each beaker you drop g of a different metal that has been heated to . Assuming no heat loss to the surroundings, which water sample will have the highest final temperature? Explain your answer.
a. The water to which you have added aluminum .
b. The water to which you have added iron .
c. The water to which you have added copper .
d. The water to which you have added lead .
e. Because the masses of the metals are the same, the final temperatures would be the same.
· 17.
For each of the following situations a, b, and c, use the following choices i, ii, and iii to complete the statement “The final temperature of the water should be”
i. Between and
ii.
iii. Between and
d. A -g sample of water at is added to a -g sample of water at .
e. A -g sample of water at is added to a -g sample of water at .
f. You have a Styrofoam cup with g of water at . You add a -g iron ball at to the water.
· 18.
How is Hess’s law a restatement of the first law of thermodynamics?
· 19.
Does the entropy of the system increase or decrease for each of the following? Explain.
. the evaporation of alcohol
a. the freezing of water
b. dissolving in water
· 20.
Predict the sign of for each of the following changes.
. 
a.
b.
c.
Questions and Problems: 10.1 The Nature of Energy
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 1.
represents the ability to do work or to produce heat.
· 2.
What is meant by potential energy? Give an example of an object or material that possesses potential energy.
· 3.
What is the kinetic energy of a particle of mass moving through space with velocity ?
· 4.
The total energy of the universe is .
· 5.
What is meant by a state function? Give an example.
· 6.
In Fig. 10.1, what kind of energy does ball A possess initially when at rest at the top of the hill? What kind of energies are involved as ball A moves down the hill? What kind of energy does ball A possess when it reaches the bottom of the hill and stops moving after hitting ball B? Where did the energy gained by ball B, allowing it to move up the hill, come from?
Questions and Problems: 10.2 Temperature and Heat
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 7.
Students often confuse what is meant by heat and temperature. Define each. How are the two concepts related?
· 8.
If you spilled a cup of freshly brewed hot tea on yourself, you would be burned. If you spilled the same quantity of iced tea on yourself, you would not be burned. Explain.
· 9.
The random motions of the components of an object constitute the of that object.
· 10.
How are the temperature of an object and the thermal energy of an object related?
Questions and Problems: 10.3 Exothermic and Endothermic Processes
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 11.
In studying heat flows for chemical processes, what do we mean by the terms system and surroundings?
· 12.
When a chemical system evolves energy, where does the energy go?
· 13.
The combustion of methane, , is an exothermic process. Therefore, the products of this reaction must possess (higher/lower) total potential energy than do the reactants.
· 14.
Are the following processes exothermic or endothermic?
a. When solid is dissolved in water, the solution gets colder.
b. Natural gas is burned in a furnace.
c. When concentrated sulfuric acid is added to water, the solution gets very hot.
d. Water is boiled in a teakettle.
Questions and Problems: 10.4 Thermodynamics
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 15.
What do we mean by thermodynamics? What is the first law of thermodynamics?
· 16.
The energy, , of a system represents the sum of the kinetic and potential energies of all particles within the system.
· 17.
Calculate for each of the following cases.
a. ,
b. ,
c. ,
· 18.
If for a process is a positive number, then the system is (gaining/losing) energy.
· 19.
For an endothermic process, will have a (positive/negative) sign.
· 20.
A system absorbs kJ of heat, and kJ of work is done on it. Calculate .
Questions and Problems: 10.5 Measuring Energy Changes
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 21.
How is the calorie defined? How does a Calorie differ from a calorie? How is the joule related to the calorie?
· 22.
Write the conversion factors that would be necessary to perform each of the following conversions:
a. an energy given in calories to its equivalent in joules
b. an energy given in joules to its equivalent in calories
c. an energy given in calories to its equivalent in kilocalories
d. an energy given in kilojoules to its equivalent in joules
Problems
![]() directs you to the Chemistry in Focus feature in the chapter
directs you to the Chemistry in Focus feature in the chapter
· 23.
If kJ of heat is needed to raise the temperature of a sample of metal from to , how many kilojoules of heat will be required to raise the temperature of the same sample of metal from to ?
· 24.
If it takes J of energy to warm a -g sample of water, how much energy would be required to warm g of water by the same amount?
· 25.
Convert the following numbers of calories or kilocalories into joules and kilojoules (Remember: kilo means .)
a. kcal
b. cal
c. cal
d. kcal
· 26.
Convert the following numbers of calories into kilocalories. (Remember: kilo means .)
a. cal
b. cal
c. cal
d. cal
· 27.
Convert the following numbers of kilojoules into kilocalories. (Remember: kilo means .)
a. kJ
b. kJ
c. kJ
d. kJ
· 28.
Convert the following numbers of calories or kilocalories into joules or kilojoules.
a. cal
b. cal
c. kcal
d. cal
· 29.
Perform the indicated conversions.
a. cal into kilojoules
b. kJ into joules
c. kcal into joules
d. kJ into kilocalories
· 30.
Perform the indicated conversions.
a. kcal into calories
b. kJ into calories
c. J into kilocalories
d. cal into kilojoules
· 31.
If kJ of heat is applied to a -g block of metal, the temperature of the metal increases by Calculate the specific heat capacity of the metal in .
· 32.
What quantity of heat energy must have been applied to a block of aluminum weighing g if the temperature of the block of aluminum increased by ? (See Table 10.1.)
· 33.
If J of heat energy is applied to a block of silver weighing g, by how many degrees will the temperature of the silver increase? (See Table 10.1.)
· 34.
If J of heat energy is applied to a -g sample of mercury, by how many degrees will the temperature of the sample of mercury increase? (See Table 10.1.)
· 35.
What quantity of heat is required to raise the temperature of g of gold from to ? (See Table 10.1.)
· 36.
![]() The “Chemistry in Focus” segment Coffee: Hot and Quick(lime) discusses self-heating cups of coffee using the chemical reaction between quicklime, , and water. Is this reaction endothermic or exothermic?
The “Chemistry in Focus” segment Coffee: Hot and Quick(lime) discusses self-heating cups of coffee using the chemical reaction between quicklime, , and water. Is this reaction endothermic or exothermic?
· 37.
![]() The “Chemistry in Focus” segment Nature Has Hot Plants discusses thermogenic, or heat-producing, plants. For some plants, enough heat is generated to increase the temperature of the blossom by . About how much heat is required to increase the temperature of L of water by ?
The “Chemistry in Focus” segment Nature Has Hot Plants discusses thermogenic, or heat-producing, plants. For some plants, enough heat is generated to increase the temperature of the blossom by . About how much heat is required to increase the temperature of L of water by ?
· 38.
![]() In the “Chemistry in Focus” segment Firewalking: Magic or Science?, it is claimed that one reason people can walk on hot coals is that human tissue is mainly composed of water. Because of this, a large amount of heat must be transferred from the coals to change the temperature of the feet significantly. How much heat must be transferred to g of water to change its temperature by ?
In the “Chemistry in Focus” segment Firewalking: Magic or Science?, it is claimed that one reason people can walk on hot coals is that human tissue is mainly composed of water. Because of this, a large amount of heat must be transferred from the coals to change the temperature of the feet significantly. How much heat must be transferred to g of water to change its temperature by ?
Questions and Problems: 10.6 Thermochemistry (Enthalpy)
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 39.
The enthalpy change for a reaction that occurs at constant pressure is (higher than/lower than/the same as) the heat for that reaction.
· 40.
A is a device used to determine the heat associated with a chemical reaction.
Problems
· 41.
The enthalpy change for the reaction of hydrogen gas with fluorine gas to produce hydrogen fluoride is kJ for the equation as written:
a. What is the enthalpy change per mole of hydrogen fluoride produced?
b. Is the reaction exothermic or endothermic as written?
c. What would be the enthalpy change for the reverse of the given equation (that is, for the decomposition of HF into its constituent elements)?
· 42.
For the reaction , per mole of formed.
a. Calculate the quantity of heat released when g of sulfur is burned in oxygen.
b. Calculate the quantity of heat released when mole of sulfur is burned in air.
c. What quantity of energy is required to break up mole of into its constituent elements?
· 43.
![]() The “Chemistry in Focus” segment Burning Calories discusses calories in food. If a food is said to contain calories per serving, determine this value in terms of joules.
The “Chemistry in Focus” segment Burning Calories discusses calories in food. If a food is said to contain calories per serving, determine this value in terms of joules.
Questions and Problems: 10.7 Hess’s Law
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 44.
When ethanol (grain alcohol, ) is burned in oxygen, approximately kJ of heat energy is released per mole of ethanol.
a. What quantity of heat is released for each gram of ethanol burned?
b. What is for the reaction as written?
c. How much heat is released when sufficient ethanol is burned so as to produce mole of water vapor?
Problems
· 45.
Given the following hypothetical data:
Calculate for the reaction
· 46.
Given the following data:
Calculate for the reaction .
· 47.
Given the following data:
Calculate for the reaction .
· 48.
Given the following data:
Calculate for the reaction
Questions and Problems: 10.8 Quality versus Quantity of Energy
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 49.
Consider the gasoline in your car’s gas tank. What happens to the energy stored in the gasoline when you drive your car? Although the total energy in the universe remains constant, can the energy stored in the gasoline be reused once it is dispersed to the environment?
· 50.
Although the total energy of the universe will remain constant, why will energy no longer be useful once everything in the universe is at the same temperature?
· 51.
Why are petroleum products especially useful as sources of energy?
· 52.
Why is the “quality” of energy decreasing in the universe?
Questions and Problems: 10.9 Energy and Our World
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 53.
Where did the energy stored in wood, coal, petroleum, and natural gas originally come from?
· 54.
What does petroleum consist of? What are some “fractions” into which petroleum is refined? How are these fractions related to the sizes of the molecules involved?
· 55.
What does natural gas consist of? Where is natural gas commonly found?
· 56.
What was tetraethyl lead used for in the petroleum industry? Why is it no longer commonly used?
· 57.
What are the four “stages” of coal formation? How do the four types of coal differ?
· 58.
What is the “greenhouse effect”? Why is a certain level of greenhouse gases beneficial, but too high a level dangerous to life on earth? What is the most common greenhouse gas?
Questions and Problems: 10.10 Energy as a Driving Force
Questions and Problems with answers below also have full solutions in the Student Solutions Guide.
Questions
· 59.
A is some factor that tends to make a process occur.
· 60.
What does it mean to say that “energy spread” and “matter spread” are driving forces in chemical reactions?
· 61.
If a reaction occurs readily but has an endothermic heat of reaction, what must be the driving force for the reaction?
· 62.
Does a double-displacement reaction such as
result in a matter spread or in a concentration of matter?
· 63.
What do we mean by entropy? Why does the entropy of the universe increase during a spontaneous process?
· 64.
A chunk of ice at room temperature melts, even though the process is endothermic. Why?
Additional Problems
· 65.
Which of the following is an endothermic process?
a. combustion of gasoline in a car engine
b. sublimation of carbon dioxide;
c. water condensing on a cold pipe
d. freezing juice to make popsicles
· 66.
Which of the following statements is(are) true?
a. Energy is changed in form in a process, but it is not produced or consumed.
b. Heat is a flow of energy between two objects due to a temperature difference in the two objects.
c. In an exothermic reaction, energy as heat flows out of the system into its surroundings.
d. In an endothermic process, energy as heat flows from the surroundings into the system.
· 67.
Perform the indicated conversions.
a. cal into joules
b. J into calories
c. kJ into joules
d. cal into kilojoules
· 68.
Calculate the amount of energy required (in calories) to heat g of water from to .
· 69.
It takes kJ of energy to heat a certain sample of pure silver from to . Calculate the mass of the sample of silver.
· 70.
What quantity of heat energy would have to be applied to a -g block of iron in order to raise the temperature of the iron sample by ? (See Table 10.1.)
· 71.
The specific heat capacity of gold is . Calculate the specific heat capacity of gold in .
· 72.
Calculate the amount of energy required (in joules) to heat kg of water from to .
· 73.
If J of heat is applied to -g samples of each of the substances listed in Table 10.1, which substance’s temperature will increase the most? Which substance’s temperature will increase the least?
· 74.
A -g sample of water at is poured into a -g sample of water at . What will be the final temperature of the water?
· 75.
A -g sample of pure iron at is dropped into g of water at . What is the final temperature of the water—iron mixture?
· 76.
If kJ of heat is applied to a -g block of metal, the temperature increases by . Calculate the specific heat capacity of the metal in .
· 77.
For each of the substances listed in Table 10.1, calculate the quantity of heat required to heat g of the substance by .
· 78.
A system releases kJ of heat and has a calculated of kJ. How much work was done on the system?
· 79.
Calculate for each of the following.
a. ,
b. ,
c. ,
d. In which of these cases do the surroundings do work on the system?
· 80.
Calculate the enthalpy change when g of propane is burned with excess oxygen according to the reaction
· 81.
The overall reaction in commercial heat packs can be represented as
a. How much heat is released when moles of iron is reacted with excess ?
b. How much heat is released when mole of is produced?
c. How much heat is released when g iron is reacted with excess ?
d. How much heat is released when g and g are reacted?
· 82.
Consider the following equations:
Suppose the first equation is reversed and multiplied by , the second and third equations are divided by , and the three adjusted equations are added. What is the net reaction and what is the overall heat of this reaction?
· 83.
It has been determined that the body can generate kJ of energy during one hour of strenuous exercise. Perspiration is the body’s mechanism for eliminating this heat. How many grams and how many liters of water would have to be evaporated through perspiration to rid the body of the heat generated during two hours of exercise? (The heat of vaporization of water is kJ/mol.)
· 84.
Which of the following processes is(are) exothermic?
a. combustion of methane (e.g., Bunsen burner)
b. melting of ice
c. evaporation of acetone (e.g., fingernail polish remover)
d. steam condensing on a cold surface
ChemWork Problems
These multiconcept problems (and additional ones) are found interactively online with the same type of assistance a student would get from an instructor.
· 85.
Which of the following reactions is/are endothermic?
a.
b.
c.
d.
e.
· 86.
The specific heat capacity of graphite is . Calculate the energy required to raise the temperature of moles of graphite by .
· 87.
A swimming pool, m by m, is filled with water to a depth of m at a temperature of . How much energy is required to raise the temperature of the water to ?
· 88.
Consider the reaction
Calculate the amount of heat released when g of diborane is combusted.
· 89.
Calculate for the reaction
given the following data:
Equation |
|