Introductory Chemistry: A Foundation - Zumdahl S.S., DeCoste D.J. 2019
Modern Atomic Theory
Electromagnetic Radiation
Objective
· To explore the nature of electromagnetic radiation.
If you hold your hand a few inches from a brightly glowing light bulb, what do you feel? Your hand gets warm. The “light” from the bulb somehow transmits energy to your hand. The same thing happens if you move close to the glowing embers of wood in a fireplace—you receive energy that makes you feel warm. The energy you feel from the sun is a similar example.
In all three of these instances, energy is being transmitted from one place to another by light—more properly called electromagnetic radiation . Many kinds of electromagnetic radiation exist, including the X rays used to make images of bones, the “white” light from a light bulb, the microwaves used to cook hot dogs and other food, and the radio waves that transmit voices and music. How do these various types of electromagnetic radiation differ from one another? To answer this question we need to talk about waves. To explore the characteristics of waves, let’s think about ocean waves. In Fig. 11.2 a seagull is shown floating on the ocean and being raised and lowered by the motion of the water surface as waves pass by. Notice that the gull just moves up and down as the waves pass—it is not moved forward. A particular wave is characterized by three properties: wavelength, frequency, and speed.
Figure 11.2.
A seagull floating on the ocean moves up and down as waves pass.
The wavelength (symbolized by the Greek letter lambda, ) is the distance between two consecutive wave peaks (Fig. 11.3). The frequency of the wave (symbolized by the Greek letter nu, ) indicates how many wave peaks pass a certain point per given time period. This idea can best be understood by thinking about how many times the seagull in Fig. 11.2 goes up and down per minute. The speed of a wave indicates how fast a given peak travels through the water.
Figure 11.3.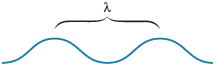
The wavelength of a wave is the distance between peaks.
Although it is more difficult to picture than water waves, light (electromagnetic radiation) also travels as waves. The various types of electromagnetic radiation (X rays, microwaves, and so on) differ in their wavelengths. The classes of electromagnetic radiation are shown in Fig. 11.4. Notice that X rays have very short wavelengths, whereas radio waves have very long wavelengths.
Figure 11.4.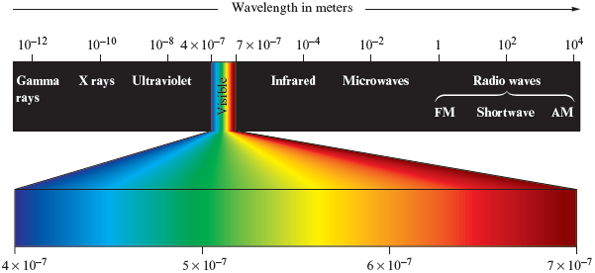
The different wavelengths of electromagnetic radiation.
Radiation provides an important means of energy transfer. For example, the energy from the sun reaches the earth mainly in the forms of visible and ultraviolet radiation. The glowing coals of a fireplace transmit heat energy by infrared radiation. In a microwave oven, the water molecules in food absorb microwave radiation, which increases their motions; this energy is then transferred to other types of molecules by collisions, increasing the food’s temperature.
Chemistry in Focus Light as a Sex Attractant
Parrots, which are renowned for their vibrant colors, apparently have a secret weapon that enhances their colorful appearance—a phenomenon called fluorescence. Fluorescence occurs when a substance absorbs ultraviolet (UV) light, which is invisible to the human eye, and converts it to visible light. This phenomenon is widely used in interior lighting in which long tubes are coated with a fluorescent substance. The fluorescent coating absorbs UV light (produced in the interior of the tube) and emits intense white light, which consists of all wavelengths of visible light.
Interestingly, scientists have shown that parrots have fluorescent feathers that are used to attract the opposite sex. Note in the accompanying photos that a budgerigar parrot has certain feathers that produce fluorescence. Kathryn E. Arnold of the University of Queensland in Australia examined the skins of Australian parrots from museum collections and found that the feathers that showed fluorescence were always display feathers—ones that were fluffed or waggled during courtship. To test her theory that fluorescence is a significant aspect of parrot romance, Arnold studied the behavior of a parrot toward birds of the opposite sex. In some cases, the potential mate had a UV-blocking substance applied to its feathers, blocking its fluorescence. Arnold’s study revealed that parrots always preferred partners that showed fluorescence over those in which the fluorescence was blocked. Perhaps on your next date you might consider wearing a shirt with some fluorescent decoration!
See Problem 11.7
Thus we visualize electromagnetic radiation (“light”) as a wave that carries energy through space. Sometimes, however, light doesn’t behave as though it were a wave. That is, electromagnetic radiation can sometimes have properties that are characteristic of particles. (You will learn more about this idea in later courses.) Another way to think of a beam of light traveling through space, then, is as a stream of tiny packets of energy called photons .
What is the exact nature of light? Does it consist of waves or is it a stream of particles of energy? It seems to be both (Fig. 11.5). This situation is often referred to as the wave—particle nature of light.