Liquid-State Physical Chemistry: Fundamentals, Modeling, and Applications (2013)
3. Basic Energetics: Intermolecular Interactions
3.4. Dispersion Interaction
Although the motion of electrons in a molecule is rather fast, at any moment the charge distribution of a molecule will have a (nonpermanent) dipole moment. The field associated with this dipole moment induces a (also nonpermanent) dipole moment in a neighboring molecule. Both induced dipoles interact, and this leads to the so-called (London) dispersion interaction, which is relatively weak but omnipresent and (in vacuum) always attractive. The exact derivation is outside the scope of these notes. However, a relatively simple model, proposed by London [19]7) and using the Drude model for atoms, yields good insight in the nature of the interaction. Moreover, it yields the correct expression apart from the exact numerical pre-factor.
In the Drude model, one assumes that an atom or molecule can be considered as a set of particles (electrons) with charge ei and mass mi. Each of these particles is harmonically and isotropically bound to its equilibrium position. The configuration and notation as used for a single electron is sketched in Figure 3.4. By considering the in-line configuration first, the dynamics of this electron are thus given by those of a harmonic oscillator for which we have
![]()
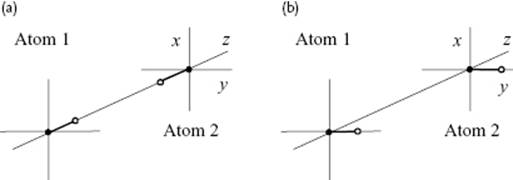
Figure 3.4 Schematic of the momentary interaction between the electrons (o) bonded to the nucleus (•) of two Drude atoms for an in-line orientation (a) and a parallel orientation (b).
From quantum mechanics (see Section 2.2) we know that the energy expression becomes
![]()
where n denotes the quantum number n = 0, 1, 2, 3, … and ω0 the circular frequency of the oscillator. The latter is given by
![]()
where a is the force constant with which the electron is bound to the nucleus and m the electron mass. Without any interaction the total energy is given by
![]()
In the ground state, n = 0, the energy is
![]()
At any moment electron 1 of atom 1 exerts a force on electron 2 of atom 2 and for a distance r, using Eq. (3.8), the potential is given by
(3.13) ![]()
so that the resulting total energy U is
![]()
This equation can be reduced to a sum of two independent harmonic oscillators, but with different frequencies. This is accomplished by the transformation
![]()
![]()
Hence, the solution becomes
![]()
with the new frequencies
![]()
and force constants
![]()
The new ground-state energy becomes
![]()
Expanding the square roots by the binomial theorem up to second order results in
(3.14) ![]()
Finally, we link the force constant a to the polarizability α using the force eE of the electric field E as compensated by the restoring force az, that is, z = eE/a. The induced dipole moment is given by μ = ez = e2E/a as well as μ = αE, so that α = e2/a. The final expression for the in-line interaction energy becomes
(3.15) ![]()
The dispersion energy is thus proportional to the polarizability α squared, a characteristic energy I ≡ ħω0 and the reciprocal sixth power of the distance r.
For the two parallel vibrations, perpendicular to the joining axis, of which the solutions are degenerated, a similar calculation leads to one-quarter of the above mentioned expression for each orientation, so that the total interaction becomes
(3.16) ![]()
For dissimilar molecules, a similar but more complex calculation leads to
(3.17) ![]()
This result is thus exact for the Drude model of a molecule with only one characteristic frequency, apart from the “binomial approximation”. For real molecules, the energies I should be chosen in accordance with the strongest absorption frequencies. In the absence of this information for I the ionization energies I0 are often taken. This seems to work reasonably well for He (I/I0 ≅ 1.20) and H2 (I/I0 ≅ 1.09). For the noble gases Ne, Ar, Kr and Xe, however, I/I0 should be taken about 9/4 in order to match more reliable calculations [2]. A similar factor or even higher was noted for N2, Cl2, and CH4. In general, although estimates for α are fairly reliable (see Chapter 10), estimates for I are less trustworthy.
Problem 3.7
Show that Vint for the in-line configuration and the parallel orientation using the Drude model and the expressions for the dipole interactions is given by
![]()
Problem 3.8
Verify for interacting oscillators the expression for U0′ and Win-line.
Problem 3.9*
Verify for interacting oscillators the expression for Wparallel.
Problem 3.10*
Derive the London dispersion interaction between dissimilar molecules.