Liquid-State Physical Chemistry: Fundamentals, Modeling, and Applications (2013)
5. The Transition from Microscopic to Macroscopic: Statistical Thermodynamics
5.2. Perfect Gases
The concept of a perfect gas is well known, and its behavior is succinctly described for one mole of gas by the equation of state
![]()
where P is pressure, Vm the molar volume, R the gas constant, and T the (absolute) temperature. Originally, this equation was obtained empirically.
Let us apply the statistical mechanics ideas to the perfect gas. The simplest model that we have for a perfect gas is that of particles without internal structure and without any mutual interaction contained in a box. For evaluation of the partition function, we first need the partition function for a single particle, and in order to obtain that result we need an expression for the energy of a particle. Thereafter, we have to extend the result to many particles.
5.2.1 Single Particle
From quantum mechanics we know that for a particle of mass m in a 3D box with edge l and volume V = l3, we have for the energy levels
(5.25) ![]()
|
ħ |
= |
h/2π = Planck's constant/2π and |
|
n |
= |
(n1, n2, n3) with (n1, n2, n3) ∈ (±1, ±2, ±3, … ) |
The momentum p = ħk of such a particle is given by k = 2πn/2l, where k is the wave vector. The single particle partition function z is thus
![]()
The spacing of energy levels is close so that the summation may be replaced by an integration leading to the standard integrals (see Appendix B)
(5.26) ![]()
(5.27) ![]()
(5.28) ![]()
with the thermal wavelength Λ defined by Λ ≡ (h2/2πmkT)1/2.
5.2.2 Many Particles
In many cases, such as in the case of the ideal gas, the total energy Ej in state j can be written as the sum of the energies of particles εi, that is, the interaction energy between particles is negligible as compared with to the total energy, and we have
(5.29) ![]()
where the indices i = 1, 2, … , N now indicate each of the N particles in state j. The partition function becomes
(5.30) 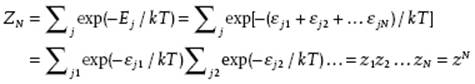
The total partition function ZN is thus the product of the single particle partition functions z, that is, ZN = zN.
If the gas consists of molecules instead of atoms, we must realize that molecules have an internal motion with an associated energy. To a good first approximation, the total energy can be taken as the sum of energies of the contributing mechanisms. For polyatomic molecules these mechanisms would be (overall) translation, (overall and internal) rotation, (internal) vibration and electronic transitions. So, we have
![]()
where each of the indices α, β, … indicates a mechanism. In this case we have
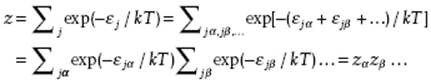
Thus, the total single partition function z is thus also the product of the partition functions zα for each of the mechanisms.
There is one more aspect that we have to discuss for systems with N particles, namely that particles are indistinguishable, leading to a correction for the N-particle partition function. A simple consideration often given is that for each of the N particles we could choose another one, leading to overcounting by N! and this leads to extra factor 1/N! in the partition function9). The real reason originates from the quantum nature of molecules, indeed related to indistinguishability, and, after a somewhat complex, approximate reasoning leading to the factor 1/N!. Here, we accept that result (see Justification 5.3) and replace the expression for partition function ZN of N independent particles Z = zN by
(5.31) ![]()
where z is the total single particle partition function. If we apply this approximation, we usually say that we use Boltzmann statistics. We will see in the next paragraph that the factor N! does not influence the pressure P, that is, the EoS, but does influence the entropy S (see Problem 5.8).
5.2.3 Pressure and Energy
The Helmholtz function F is related to the partition function Z via
(5.32) ![]()
So, let us try to calculate the mechanical equation of state (EoS) for an ideal gas. The mechanical EoS expresses the pressure P = −∂F/∂V as a function of the independent variables. Here we find, using the Stirling approximation10) for lnN!,
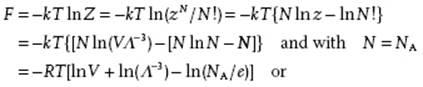
(5.33) ![]()
In this last expression we recognize the well-known perfect gas law.
Because from the Helmholtz function F all other thermodynamic expressions can be derived, the same is true for the partition function as the two are related via the expression F = −kT lnZ. For the entropy S and energy U we find, respectively,
(5.34) ![]()
We can also calculate U directly from Z using to the Gibbs–Helmholtz expression U = ∂(F/T)/∂(1/T) = kT2 ∂lnZ/∂T. The total energy U is entirely kinetic and reads
(5.35) ![]()
and hence the capacity CV is given by CV = (∂U/∂T)V = 3R/2. Since we have the general relation ![]() , we obtain CP = 5R/2.
, we obtain CP = 5R/2.
Problem 5.7
Show that the internal energy U for an ideal gas for the ideal gas consisting of N structureless particles reads U = 3NkT/2. Also show that CV = 3R/2, and hence that CP = 5R/2.
Problem 5.8
Show that the entropy S for an ideal gas is given by the so-called Sackur–Tetrode equation
![]()
Similarly, show that the chemical potential μ = kT ln(N/z).
Problem 5.9: N! again*
According to quantum mechanics a system wave function should be either antisymmetric (FD particles) or symmetric (BE particles) in its coordinates (see Section 2.3). For weakly interacting particles in a system we may assume the existence of single-particle states. Show that for N particles in a system with q single-particle states (q ≥ N, generally q >> N), the number of system states for FD particles is WFD = q!/(q − N)!N!, while for BE particles WBE = (q + N − 1)!/N!(q − 1)! ≅ (q + N)!/N!(q)!. Also show that for q >> N, both WFD and WBE reduces to the number of Boltzmann states WBo = qN/N!.
Problem 5.10*
Show that for the perfect gas, using ![]() , the fluctuations are given by
, the fluctuations are given by ![]() and thus vanish in the thermodynamic limit.
and thus vanish in the thermodynamic limit.