Liquid-State Physical Chemistry: Fundamentals, Modeling, and Applications (2013)
11. Mixing Liquids: Molecular Solutions
11.2. Ideal and Real Solutions
Perfect solutions behave as perfect gas mixtures over the complete concentration range. In practice, perfect solution behavior is obeyed only for a limited range of concentrations, and we label these cases as ideal solutions. For perfect and ideal behavior, a number of consequences follow of which we describe Raoult's and Henry's laws. Thereafter, we discuss how to deal with real solutions.
11.2.1 Raoult's and Henry's Laws
For ideal solutions we have (within the range of validity) the expression
(11.20) ![]()
while for the perfect gas the expression
(11.21) ![]()
holds. Using the equilibrium condition μ(sol) = μ(vap), we easily obtain
(11.22) ![]()
Solving for Pα leads to
(11.23) ![]()
The parameter KH,α is independent of composition. For the solute we have
(11.24) ![]()
known as Henry's law (1803!), and stating that the partial pressure P2 of solute 2 is proportional to the mole fraction in the liquid phase with a proportionality constant dependent on the difference in chemical potential, that is, in interaction, between solvent 1 and solute 2. For the solvent, the limiting situation is x1 = 1 for which ![]() . Hence approximately
. Hence approximately
(11.25) ![]()
known as Raoult's law (1886–1887), and stating that the partial pressure P1 of solvent 1 is directly proportional to the vapor pressure ![]() of the pure solvent. Obviously, for perfect solutions Raoult's law is valid for the complete composition range.
of the pure solvent. Obviously, for perfect solutions Raoult's law is valid for the complete composition range.
Some data for Henry's constant are given in Table 11.1. Example 11.1 shows how to use these concepts.
Table 11.1 Henry's constant KH for gases dissolved in H2O and C6H6 (in brackets) at 25 °C.a)
|
Gas |
KH (GPa) |
|
CH4 |
4.185 (0.0569) |
|
C2H2 |
0.135 |
|
C2H4 |
1.155 |
|
C2H6 |
3.06 |
|
Air |
7.295 |
|
N2 |
8.7650 (0.239) |
|
O2 |
44380 |
|
H2 |
7.16 (0.367) |
|
He |
12.66 |
|
CO |
5.79 (0.163) |
|
CO2 |
1.670 (0.0144) |
|
H2 S |
0.055 |
a) Data from Refs [21, 22].
Example 11.1
Assuming that sparkling water contains only H2O (1) and CO2 (2), we want to determine for a sealed can the compositions of the vapor and liquid phases and the pressure exerted at 10 °C, knowing that Henry's constant for CO2 in H2O at 10 °C is about 990 bar. There is only one degree of freedom according to the phase rule. We use the mole fraction x2 of CO2 in the liquid and assume it is 0.01. We denote the mole fraction in the gas phase by y. For the solute (2) we have Henry's law, while for the solvent (1) Raoult's law applies.
![]()
With KH,2 = 990 bar and ![]() (steam tables at 10 °C or Antoine's equation), P = 9.912 bar. By Raoult's law,
(steam tables at 10 °C or Antoine's equation), P = 9.912 bar. By Raoult's law, ![]() . Therefore, y2 = 1 − y1 = 0.9988. As expected, the vapor is nearly pure CO2.
. Therefore, y2 = 1 − y1 = 0.9988. As expected, the vapor is nearly pure CO2.
11.2.2 Deviations
To describe deviations from perfect solution behavior we have a few options. The most well-known option is introducing the activity a = γ x, where γ is the activity coefficient. The activity coefficient γα = γα(T,x) is introduced “to keep up appearances,” that is, to keep the same formal expression for μα as for the perfect solution. Hence, ![]() becomes
becomes ![]() , with activity aα = γαxα. Obviously, γideal = 1 always. There are two conventions for the activity coefficient.
, with activity aα = γαxα. Obviously, γideal = 1 always. There are two conventions for the activity coefficient.
· Convention I: γα → 1 for xα → 1 and for xα → 0. This convention is usually employed if the (liquid) solute is fully soluble in the solvent. Because, in a dilute solution of component β in α, molecules of type α are mainly surrounded by molecules of type α, component α behaves largely as if the liquid was pure. The reference state is thus the chemical potential of the pure liquid ![]() . This convention is also referred to as Raoult's law convention.
. This convention is also referred to as Raoult's law convention.
· Convention II: γ1 → 1 for x1 → 1 and γ2 → 1 for x2 → 0. This convention is usually employed if the (solid or liquid) solute is only partially soluble in the solvent. For the solvent this convention is identical to convention I. Because, in a dilute solution of component 2 in 1, molecules of type 2 do not interact with each other but mainly with molecules of type 1, the activity coefficient approaches a constant value, characteristic for the 1–2 interaction. Since we would like to have γ2 → 1 for x2 → 0, the reference state for the solute is taken as that of the pure solute in a hypothetical liquid state extrapolated to infinite dilution using Henry's law. Accordingly, this convention is referred to as Henry's law convention.
From the Gibbs–Duhem equation x1dμ1 + x2dμ2 = 0, we obtain (using μ = μ* + RT lnγ x) x1 d lnγ1 + x2 d lnγ2 = 0. Similarly, as for Eq. (11.7), some manipulation results in
(11.26) ![]()
where γ1,0 is the limiting value of γ1 for x1 → 0. Since for x1 → 0, γ1 → 1, the lower limit in the second integral should be taken as zero. This relation provides a check on the experimental data for γ1 and γ2 or allows one to calculate γ2 if γ1 is known.
Another way to introduce deviations from ideal behavior is by introducing the osmotic coefficients g and ϕ, defined for the solvent by
(11.27) ![]()
where the molality m2 = x2/x1M1 and the mole fraction x1 = (1 + m2M1)−1 are used (why g and ϕ are addressed as osmotic coefficients will become clear in the next section). Since for high dilution we should regain ideal behavior, we require that for x1 → 1, g → 1, and thus ϕ → 1. We have the relations lnγ1 = (g − 1)lnx1 and ϕ = −(m2M1)−1g ln (1 + m2M1)−1 ≅ g (1 − ½m2M1 + …).
We also can define excess functions by
(11.28) ![]()
The partial quantities Zα are directly related to the excess quantities ZE. For example, for G we have Zα = Gα = μα and substitution of Eq. (11.7) in ![]() , dividing by x1 = 1 − x2 and differentiating with respect to x2 yields
, dividing by x1 = 1 − x2 and differentiating with respect to x2 yields
(11.29) ![]()
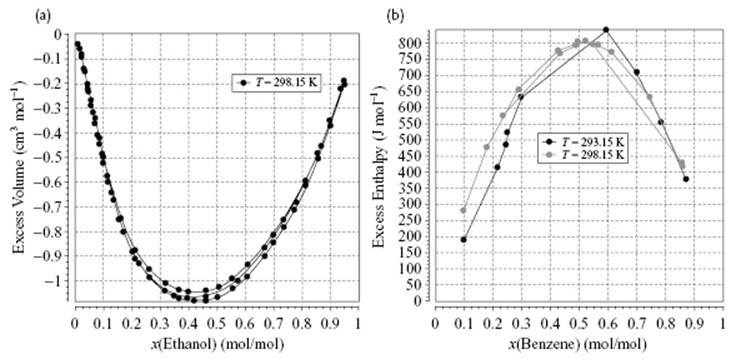
It is a matter of convenience whether excess or partial quantities are used. Figure 11.3 shows two examples of excess functions.
Figure 11.3 (a) Excess volume VE for a mixture of H2O–C2H5OH; (b) Excess enthalpy HE for a mixture of benzene in cyclohexane. Images obtained from the Dortmund Data Base.
Problem 11.7
Verify Eq. (11.26) and (11.27).