Liquid-State Physical Chemistry: Fundamentals, Modeling, and Applications (2013)
11. Mixing Liquids: Molecular Solutions
11.7. The Activity Coefficient for Other Composition Measures*
So far, we have dealt with activity coefficients using the mole fraction x as a measure for composition. In practice, people use also molality m (m moles of solute per 1 kg of solvent) and molarity c (c moles per liter solution). Whichever measure is used, the difference μ − μ∞, where μ∞ is the infinite dilution reference value, should be the same. Denoting with ax and am the activities using the mole fraction and molality scale we must have
(11.111) 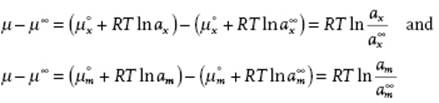
So we obtain
(11.112) ![]()
with γ the activity coefficient. For the molality scale we have (see Chapter 2)
(11.113) ![]()
where M1 is the molar mass of the solvent. For infinite dilution
![]()
Moreover, for infinite dilution,
![]()
In total, we thus obtain
(11.114) ![]()
A similar calculation for the molarity scale yields γc/γx = x2ρ/c2M1 with ρ the mass density and c2 the molarity of the solute.
Problem 11.17
Calculate the relationship between the activity coefficients on the mole fraction scale and the molarity scale.