Liquid-State Physical Chemistry: Fundamentals, Modeling, and Applications (2013)
12. Mixing Liquids: Ionic Solutions
12.4. Strong and Weak Electrolytes
For strong electrolytes Bjerrum first made the proposition, now generally accepted, that complete dissociation is present [14]. NaCl in water provides an example. Generally, an electrolyte ![]() dissociates into ν = ν+ + ν− ions according to
dissociates into ν = ν+ + ν− ions according to
(12.13) ![]()
where z+ and z− are the charges of M and X, respectively. Electroneutrality requires that
(12.14) ![]()
The chemical potential of the electrolyte with activities a+ and a− is
(12.15) ![]()
with the activity aQ defined by ![]() . As a reminder, the molality scale for component i is defined as mi/m0, with m0 = 1 kg mol−1. Frequently, m0 is omitted and we do so here (compare ln P, which actually means ln (P/P°) with, say, the reference pressure P° = 1 bar). The activity coefficient5) (on molality basis) is defined as γi = ai/mi, and for the salt the activity becomes
. As a reminder, the molality scale for component i is defined as mi/m0, with m0 = 1 kg mol−1. Frequently, m0 is omitted and we do so here (compare ln P, which actually means ln (P/P°) with, say, the reference pressure P° = 1 bar). The activity coefficient5) (on molality basis) is defined as γi = ai/mi, and for the salt the activity becomes
(12.16) ![]()
using the mean activity coefficient ![]() , the mean stoichiometric coefficient
, the mean stoichiometric coefficient ![]() and the fact that for an electrolyte of molality mQ we have m+ = ν+mQ and m− = ν−mQ. For a symmetric electrolyte, the above expression reduces to aQ = (γ±mQ)2. For mixed electrolytes the full expression should be used. Similar equations can be written down using molarity c and mole fraction x instead of molality m. The mole fraction is, however, infrequently used in this connection.
and the fact that for an electrolyte of molality mQ we have m+ = ν+mQ and m− = ν−mQ. For a symmetric electrolyte, the above expression reduces to aQ = (γ±mQ)2. For mixed electrolytes the full expression should be used. Similar equations can be written down using molarity c and mole fraction x instead of molality m. The mole fraction is, however, infrequently used in this connection.
For weak electrolytes, Arrhenius proposed in 1887 that dissociation is incomplete but that dissociation increases with increasing dilution. An example is a solution of acetic acid in water for which the dissociation equilibrium is
(12.17) ![]()
For the general electrolyte, Eq. (12.13), the molarity of positive ions for a fully dissociated solution is ν+mQ with mQ the molarity Q. With association this becomes m+ + αν+mQ with α the degree of dissociation. Because the total molarity of M+ ions, whether dissociated or not, is ν+mQ, we have
(12.18) ![]()
where mIP denotes the molarity of the ion pairs. The total molarity of negative ions, whether dissociated or not, is ν−mQ so that
(12.19) ![]()
Since γ± is given by ![]() and the chemical potential reads
and the chemical potential reads
(12.20) ![]()
we find, using Eq. (12.15),
(12.21) ![]()
For ν+ = ν− = 1, the activity coefficient reduces to (αγ±)ν. For the acetic acid example mentioned before, the dissociation constant K is thus given by
(12.22) ![]()
For a solution of HA of molality m we have ![]() and
and
(12.23) ![]()
where the last step only can be made if we take γ± = γHA = 1.
Equation (12.23) is the dilution law, due to Ostwald in 1888, which expresses quantitatively that the lower the concentration, the larger the degree of dissociation. For acetic acid at 25 °C at infinite dilution (i.e., the limiting value), K ≅ 1.75 × 10−5 mol kg−1, and does not deviate more than a few percent from this value up to 10−1 mol kg−1. From the overall reaction, Eq. (12.17), and the expression for K, Eq. (12.22), the chemical potential becomes ![]() , where
, where ![]() . An accurate empirical description for the dissociation constant K reads
. An accurate empirical description for the dissociation constant K reads
(12.24) ![]()
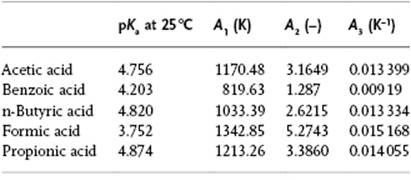
In practice, one uses pKa values, defined by pKa ≡ −logK = A1/T − A2 + A3T. Values of the constants A1, A2 and A3 for a few typical acids are listed in Table 12.7.
Table 12.7 Constants for the pKa expression values (mol kg−1) for some acids in H2O.
Van't Hoff found that the osmotic pressure of electrolyte solutions was always significantly higher than was predicted by colligative theory. For example, for the osmotic pressure equation Van't Hoff proposed Π = icRT, where i is now known as the van't Hoff factor. In principle this factor is equal to, but dependent on concentration, in practice usually less than the number of ions formed from one mole of compound. If, for a symmetric electrolyte 1 mole provides ν ions upon complete dissociation, the number of really dissociated molecules is να and the number of undissociated molecules is 1 − α. Hence, the total number of ions in solution i = 1 − α + να or α = (i − 1)/(ν − 1). The value for α obtained from colligative properties is only in approximate agreement with values determined via conductivity measurements.
Problem 12.10
Show that, if −R ln Ka = A/T − C + DT, the Ka value shows a maximum given by R ln Ka = C − 2(AD)1/2 at temperature Tmax = (A/D)1/2.
Problem 12.11
Calculate ΔH, ΔS and ΔCP from ΔG for the dissociation of a weak electrolyte.